Advanced Catalytic Synthesis of Photoinitiator 369 Intermediates for Commercial Scale-Up and Cost Efficiency
The global demand for high-performance ultraviolet (UV) curing materials continues to surge, driving the need for efficient and safe synthesis routes for critical photoinitiators. Patent CN108358871B, published on May 22, 2020, introduces a groundbreaking synthetic method for 2-benzyl-2-dimethylamino-1-(4-morpholinophenyl) butanone, widely known in the industry as Photoinitiator 369. This specific compound serves as a vital component in UV adhesives, coatings, and inks, where its ability to generate free radicals upon light exposure dictates the curing speed and final material properties. The disclosed technology addresses long-standing inefficiencies in existing manufacturing protocols by optimizing reaction conditions and reagent usage. By shifting from hazardous high-pressure processes to a mild, catalytic-driven sequence, this innovation offers a compelling value proposition for manufacturers seeking to enhance both operational safety and economic viability in the production of specialty fine chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Photoinitiator 369 has been plagued by significant technical and economic hurdles, primarily revolving around the choice of starting materials and reaction severity. One prevalent conventional route, depicted in the reaction scheme below, utilizes chlorobenzene as the foundational raw material. While chlorobenzene is inexpensive, the subsequent morpholine substitution step in this pathway necessitates extreme conditions, specifically high temperature and high pressure. These harsh parameters not only escalate energy consumption and equipment maintenance costs but also pose severe safety risks due to the potential for runaway reactions. Furthermore, the selectivity under such aggressive conditions is often compromised, leading to the formation of numerous byproducts that complicate downstream purification and depress the overall yield.
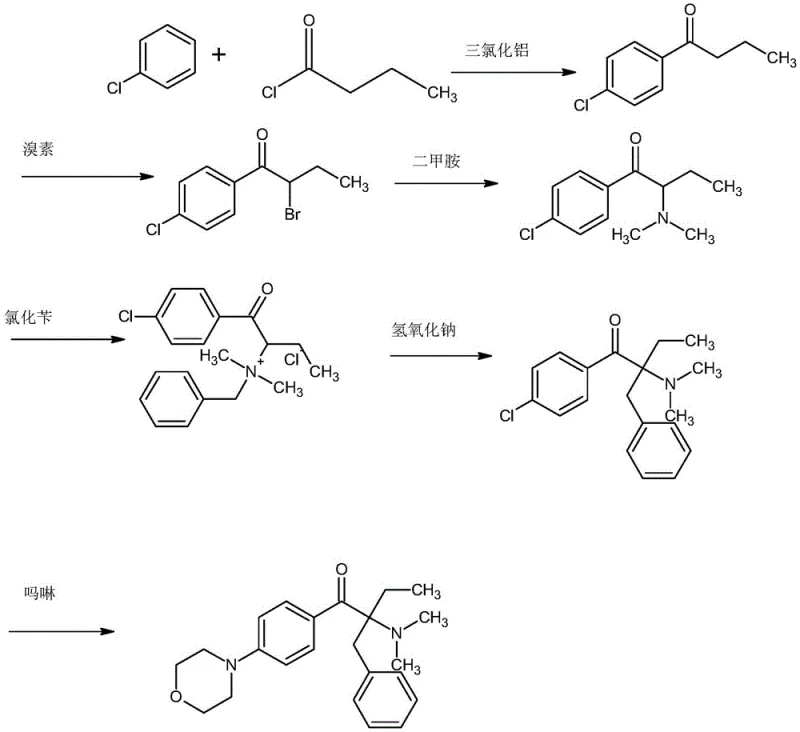
Alternatively, another established method employs fluorobenzene to circumvent the high-pressure requirement. Although this approach allows for milder morpholine substitution conditions, it introduces different bottlenecks. Fluorobenzene is a substantially more expensive raw material compared to chlorobenzene, directly inflating the Bill of Materials (BOM). Moreover, the rearrangement step in the fluorobenzene route typically suffers from suboptimal yields, often hovering around 70%, which negates the benefits of the milder substitution step. Consequently, procurement managers and supply chain directors face a dilemma: choose the cheaper raw material with high operational risk or the safer process with prohibitive material costs and mediocre yields. Neither option represents an ideal solution for large-scale, cost-sensitive commercial manufacturing.
The Novel Approach
The innovative methodology outlined in patent CN108358871B effectively resolves these dichotomies by re-engineering the synthetic sequence to leverage the cost advantages of chlorobenzene while eliminating the need for dangerous high-pressure equipment. As illustrated in the comprehensive reaction pathway below, the new process strategically reorders the synthetic steps. It initiates with a standard Friedel-Crafts acylation but introduces a crucial modification in the bromination stage by employing a catalytic oxidant. This is followed by dimethylamine substitution and quaternization before the morpholine substitution occurs. Crucially, the morpholine substitution is performed on the quaternary ammonium salt intermediate rather than the aryl chloride, utilizing a specialized composite catalyst. This sequence allows the reaction to proceed under mild thermal conditions without high pressure, thereby drastically reducing capital expenditure requirements for reactor specifications and enhancing operator safety.
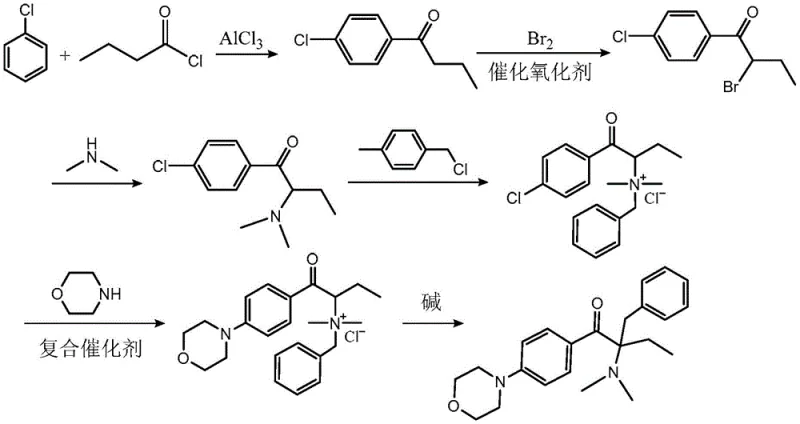
Furthermore, this novel approach achieves a remarkable improvement in total yield, reaching approximately 76.6% in optimized examples, which stands in stark contrast to the roughly 64.8% yield of the traditional chlorobenzene route and 64.5% of the fluorobenzene route. The strategic placement of the rearrangement step as the final transformation ensures that the sensitive morpholine moiety is introduced efficiently before the structural reorganization occurs. This logical progression minimizes side reactions and maximizes the conversion of valuable intermediates into the final high-purity product. For a reliable photoinitiator intermediate supplier, adopting this route translates directly into a more robust supply chain capable of meeting high-volume demands with consistent quality and reduced waste generation.
Mechanistic Insights into Catalytic Bromination and Composite Catalyst Substitution
A deeper mechanistic analysis reveals that the efficiency gains in this process are driven by two key catalytic interventions. First, the bromination of the ketone intermediate (Compound I) is enhanced by the addition of a catalytic oxidant, such as hydrogen peroxide or peracetic acid. In traditional bromination, a stoichiometric or excess amount of elemental bromine is often required to drive the reaction to completion, leading to significant halogen waste and the generation of corrosive hydrogen bromide byproducts. In this patented method, the oxidant facilitates the regeneration of active brominating species or activates the substrate, allowing the amount of elemental bromine to be reduced by 30-50%. This improvement in atom economy is not merely an environmental benefit; it significantly lowers the cost of goods sold (COGS) by reducing the consumption of one of the more expensive and hazardous reagents in the synthesis.
Secondly, the substitution of the chlorine atom on the aromatic ring with morpholine is mediated by a composite catalyst system comprising a copper source (such as copper powder or cuprous chloride) and a ligand like EDTA. In the absence of this catalyst, nucleophilic aromatic substitution on a chlorobenzene derivative would typically require extreme temperatures and pressures to overcome the high activation energy barrier. The copper-ligand complex likely operates through a coordination mechanism that activates the carbon-chlorine bond, facilitating the displacement by the morpholine nucleophile under much milder conditions (80-130°C). This catalytic cycle not only improves the reaction rate but also enhances regioselectivity, ensuring that the morpholine attaches exclusively at the para-position relative to the carbonyl group. This high selectivity is critical for maintaining the purity profile required for electronic-grade or pharmaceutical-grade applications, minimizing the burden on purification teams.
How to Synthesize 2-benzyl-2-dimethylamino-1-(4-morpholinophenyl) butanone Efficiently
The execution of this synthesis requires precise control over reaction parameters to maximize the benefits of the catalytic systems described. The process begins with the preparation of the ketone backbone via Friedel-Crafts acylation, followed by the catalytic bromination which must be carefully monitored to prevent over-bromination. The subsequent amination and quaternization steps build the necessary nitrogen functionality, setting the stage for the pivotal morpholine substitution. Operators must ensure the composite catalyst is properly activated and that the reaction temperature is maintained within the specified window to achieve full conversion without degrading the sensitive quaternary ammonium intermediate. The final rearrangement step, induced by a base such as potassium carbonate, completes the molecular architecture. Detailed standardized operating procedures regarding reagent addition rates, temperature ramps, and workup protocols are essential for reproducibility.
- Perform Friedel-Crafts acylation of chlorobenzene with n-butyryl chloride using aluminum trichloride to form Compound I.
- Execute catalytic bromination of Compound I using a catalytic oxidant and reduced bromine to obtain Compound II.
- Conduct dimethylamine substitution, followed by quaternization with benzyl chloride, composite catalyst-mediated morpholine substitution, and final alkaline rearrangement.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers transformative advantages that extend beyond simple yield improvements. The shift away from high-pressure processing equipment represents a substantial reduction in capital expenditure (CAPEX) and operational risk. Facilities can utilize standard glass-lined or stainless steel reactors rated for atmospheric or low pressure, rather than investing in specialized autoclaves required for the conventional chlorobenzene route. This flexibility allows for faster scale-up and the utilization of existing infrastructure, significantly reducing lead time for high-purity photoinitiator intermediates. Additionally, the elimination of high-pressure steps simplifies regulatory compliance and safety audits, further streamlining the path to commercial production.
- Cost Reduction in Manufacturing: The economic benefits of this process are multifaceted, stemming from both raw material optimization and operational efficiency. By reducing the consumption of elemental bromine by nearly half through catalytic oxidation, the direct material costs are significantly lowered. Furthermore, the use of chlorobenzene instead of fluorobenzene leverages a much cheaper and more widely available starting material, insulating the supply chain from the price volatility associated with fluorinated aromatics. The higher total yield of over 76% means that less raw material is wasted per kilogram of final product, effectively spreading fixed costs over a larger output volume and driving down the unit cost of production.
- Enhanced Supply Chain Reliability: Reliability in the supply of critical UV curing components is paramount for downstream customers in the coatings and electronics industries. This synthetic route enhances reliability by simplifying the process flow and removing bottleneck steps that are prone to failure or batch-to-batch variability. The mild reaction conditions reduce the likelihood of thermal runaways or equipment failures that can halt production lines. Moreover, the use of common, non-proprietary reagents like chlorobenzene and morpholine ensures that raw material sourcing remains stable and resilient against market shocks, providing a secure foundation for long-term supply contracts.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this method aligns perfectly with modern green chemistry principles. The reduction in bromine usage decreases the load on waste treatment facilities, lowering the cost and complexity of effluent management. The avoidance of high-pressure operations reduces the energy footprint of the manufacturing process. As global regulations on industrial emissions and safety tighten, having a process that inherently generates less hazardous waste and operates under safer conditions provides a competitive edge. This scalability ensures that production can be ramped up from pilot batches to multi-ton commercial quantities without encountering the engineering challenges typical of high-pressure chemistries.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on how this method outperforms legacy processes. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer or for procurement specialists assessing the long-term viability of this supply source.
Q: How does the new catalytic bromination method improve atom economy?
A: The patented process utilizes a catalytic oxidant (such as hydrogen peroxide) during the bromination step, which reduces the required amount of elemental bromine by 30-50%, significantly improving atom utilization and reducing hazardous waste.
Q: What are the safety advantages over conventional chlorobenzene routes?
A: Unlike traditional methods that require high-temperature and high-pressure conditions for morpholine substitution, this novel route performs the substitution under mild conditions using a composite copper catalyst, eliminating the need for dangerous high-pressure reactors.
Q: Why is this route more cost-effective than the fluorobenzene method?
A: While the fluorobenzene route avoids high pressure, it suffers from lower yields in the rearrangement step and higher raw material costs. This new method combines the low cost of chlorobenzene with a high-yield rearrangement sequence, achieving a total yield of approximately 76.6%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-benzyl-2-dimethylamino-1-(4-morpholinophenyl) butanone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields observed in patent examples can be reliably replicated on an industrial scale. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of Photoinitiator 369 intermediate meets the exacting standards required for high-performance UV curing applications. Our commitment to quality assurance ensures consistency that your downstream formulation processes depend upon.
We invite you to collaborate with us to leverage this advanced synthetic technology for your supply chain needs. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. We encourage potential partners to contact us directly to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data. Let us help you secure a stable, cost-effective, and high-quality supply of this critical photoinitiator intermediate, empowering your business to lead in the competitive UV curing market.
