Advanced Synthesis of Esomeprazole Sodium: Technical Breakthroughs and Commercial Scalability
Advanced Synthesis of Esomeprazole Sodium: Technical Breakthroughs and Commercial Scalability
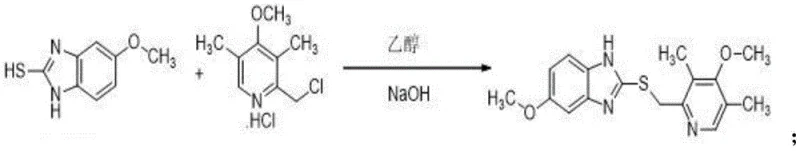
The pharmaceutical industry constantly seeks robust, scalable, and cost-effective routes for producing high-value Active Pharmaceutical Ingredients (APIs) and their intermediates. A significant advancement in this domain is detailed in patent CN113512026A, which discloses a novel synthesis method for Esomeprazole Sodium, the S-isomer of omeprazole widely used for treating gastric acid-related disorders. This technical disclosure outlines a streamlined three-step process that begins with the condensation of 5-methoxy-2-mercaptobenzimidazole and 2-chloromethyl-3,5-dimethyl-4-methoxypyridine hydrochloride. Unlike traditional methods that often struggle with impurity profiles and low yields, this patented approach leverages specific reaction conditions—including precise pH control, temperature regulation, and chiral auxiliaries—to achieve a final product purity exceeding 99%. For R&D directors and procurement specialists, understanding the mechanistic nuances and commercial implications of this route is critical for securing a reliable Esomeprazole Sodium supplier and optimizing manufacturing costs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of esomeprazole has been plagued by challenges related to stereochemical control and impurity management. Conventional routes often involve the oxidation of omeprazole sulfide using non-selective oxidants, which results in a racemic mixture requiring difficult and expensive chiral resolution steps. Furthermore, traditional processes frequently generate a complex array of by-products, including sulfones and various pyridine N-oxides, which are structurally similar to the target molecule and difficult to remove. These impurities not only lower the overall yield but also necessitate rigorous purification protocols, such as preparative HPLC, which are impractical for large-scale commercial production. The accumulation of these inefficiencies leads to inflated production costs, extended lead times, and potential supply chain bottlenecks, making it difficult for manufacturers to meet the stringent quality standards required for high-purity pharmaceutical intermediates.
The Novel Approach
The method described in patent CN113512026A offers a transformative solution by integrating a highly selective asymmetric oxidation step directly into the synthesis workflow. Instead of producing a racemate and separating it later, this route utilizes a chiral titanium-tartrate complex to induce enantioselectivity during the oxidation of the sulfide intermediate. This strategic shift eliminates the need for downstream chiral resolution, significantly simplifying the process flow. Additionally, the patent emphasizes optimized crystallization techniques using solvent systems like ethyl acetate and acetonitrile, which effectively purge impurities without the need for chromatography. By controlling reaction parameters such as the mass ratio of reactants and the addition rate of oxidants, the process minimizes the formation of over-oxidized sulfone by-products. This results in a shorter synthesis route with higher atom economy, directly addressing the pain points of cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Titanium-Tartrate Catalyzed Asymmetric Oxidation
The core of this synthesis lies in the second step, where the achiral omeprazole thioether is converted into the chiral esomeprazole sulfoxide. This transformation is mediated by a chiral catalyst system formed in situ from tetraisopropyl titanate and D-(-)-diethyl tartrate. Mechanistically, the titanium center coordinates with the tartrate ligand to create a chiral environment that directs the approach of the oxidant, cumene hydroperoxide, to the sulfur atom of the thioether. This coordination ensures that the oxygen transfer occurs preferentially from one face of the planar sulfide, yielding the desired S-enantiomer with high optical purity. The patent specifies maintaining the reaction temperature between 45°C and 55°C during the catalyst formation and then lowering it to 20°C for the oxidation, a critical thermal profile that balances reaction kinetics with stereoselectivity. The inclusion of triethylamine as a basic auxiliary further stabilizes the transition state and scavenges acidic by-products, preventing the degradation of the acid-sensitive benzimidazole ring.
Impurity control is another pivotal aspect of this mechanism. The patent data indicates that the R-enantiomer content in the final product is tightly controlled to be less than 0.15%, a specification that far exceeds typical industry standards. This high level of enantiomeric excess is achieved by fine-tuning the molar ratios of the chiral auxiliary to the substrate, specifically using a mass ratio of omeprazole thioether to diethyl tartrate of approximately 1:0.6 to 0.7. Furthermore, the workup procedure involves a careful acidification step using glacial acetic acid to adjust the pH to 8.0-8.5, which facilitates the extraction of the product into methyl isobutyl ketone while leaving polar impurities in the aqueous phase. This meticulous control over the reaction environment ensures that the final Esomeprazole Sodium meets the rigorous purity specifications required for clinical applications, demonstrating the feasibility of commercial scale-up of complex chiral sulfoxides.

How to Synthesize Esomeprazole Sodium Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for producing Esomeprazole Sodium with high efficiency and reproducibility. The process begins with the nucleophilic substitution of the mercaptobenzimidazole, followed by the critical asymmetric oxidation, and concludes with a salification and recrystallization sequence. Each step is designed to maximize yield while minimizing waste, making it an ideal candidate for green chemistry initiatives in the pharmaceutical sector. The detailed operational parameters, including solvent choices and temperature gradients, offer a robust framework for process engineers to adapt this laboratory-scale method to industrial reactors. For a comprehensive understanding of the specific operational procedures and safety considerations, the detailed standardized synthesis steps are provided in the guide below.
- Condense 5-methoxy-2-mercaptobenzimidazole with 2-chloromethyl-3,5-dimethyl-4-methoxypyridine hydrochloride in ethanol/NaOH to form Omeprazole thioether.
- Perform asymmetric oxidation on the thioether using D-(-)-diethyl tartrate, tetraisopropyl titanate, and cumene hydroperoxide to generate Esomeprazole.
- React the Esomeprazole solution with sodium hydroxide, followed by crystallization in acetone/acetonitrile to obtain refined Esomeprazole Sodium.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis method presents compelling economic and logistical benefits. The primary advantage stems from the significant simplification of the purification process. By replacing expensive and time-consuming chromatographic separations with efficient crystallization steps, the method drastically reduces the consumption of silica gel and organic solvents associated with column chromatography. This reduction in material usage translates directly into lower variable costs per kilogram of product. Moreover, the use of common industrial solvents such as ethanol, toluene, and methyl isobutyl ketone ensures that raw materials are readily available from multiple suppliers, mitigating the risk of supply disruptions. The high yield reported in the patent, reaching up to 75% for the final step, further enhances the economic viability by maximizing the output from a fixed amount of starting materials.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts that require complex removal steps, combined with the high selectivity of the asymmetric oxidation, leads to substantial cost savings. The process avoids the need for specialized chiral columns or resolution agents, which are often the most expensive components in chiral drug synthesis. Additionally, the ability to recycle solvents like ethyl acetate and acetonitrile further optimizes the cost structure. The streamlined workflow reduces labor hours and energy consumption, contributing to a more competitive pricing model for the final API intermediate.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals rather than bespoke reagents enhances the resilience of the supply chain. Starting materials like 5-methoxy-2-mercaptobenzimidazole are well-established in the market, ensuring stable pricing and availability. The robustness of the reaction conditions, which tolerate minor variations in temperature and mixing without significant loss of quality, reduces the rate of batch failures. This reliability is crucial for maintaining continuous production schedules and meeting the just-in-time delivery requirements of global pharmaceutical clients.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by the successful execution of reactions in multi-kilogram batches within the patent examples. The waste stream is primarily composed of aqueous salts and recoverable organic solvents, simplifying wastewater treatment and reducing the environmental footprint. Compliance with environmental regulations is easier to achieve when the process generates fewer hazardous by-products and utilizes greener solvent systems. This alignment with sustainability goals is increasingly important for multinational corporations seeking responsible partners for reducing lead time for high-purity gastric inhibitors.

Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of Esomeprazole Sodium as described in the patent literature. These insights are derived from the specific experimental data and beneficial effects reported, providing clarity on the method's capabilities and limitations. Understanding these details helps stakeholders make informed decisions about integrating this technology into their existing manufacturing portfolios.
Q: What is the key advantage of this synthesis method regarding purity?
A: The patented method achieves a purity of greater than 99.5% for the final Esomeprazole Sodium product by strictly controlling the R-enantiomer content to below 0.15% through optimized asymmetric oxidation conditions.
Q: How does this process improve supply chain reliability?
A: By utilizing readily available starting materials like 5-methoxy-2-mercaptobenzimidazole and avoiding complex chromatographic separations in favor of crystallization, the process ensures consistent batch-to-batch quality and shorter production cycles.
Q: Is this method suitable for large-scale commercial production?
A: Yes, the process uses standard industrial solvents such as ethanol, toluene, and methyl isobutyl ketone, and operates at moderate temperatures (35-55°C), making it highly scalable from pilot plants to multi-ton annual production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Esomeprazole Sodium Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of quality and consistency in the production of life-saving medications. Our team of expert chemists has extensively evaluated the synthesis route disclosed in CN113512026A and confirmed its potential for industrial application. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to manufacturing plant is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the critical chiral purity metrics required for Esomeprazole Sodium. We are committed to delivering products that not only meet but exceed the pharmacopeial standards set by major regulatory bodies worldwide.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our expertise in chiral synthesis and process optimization, we can help you secure a stable supply of high-quality intermediates. Please contact us to request specific COA data and route feasibility assessments for your next project. Let us be your partner in driving innovation and efficiency in the pharmaceutical industry.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
