Advanced One-Step Synthesis of 1,2,5-Polysubstituted Imidazole Derivatives for Commercial Scale-Up
Introduction to Next-Generation Imidazole Synthesis
The landscape of heterocyclic chemistry is continuously evolving, driven by the demand for more efficient and sustainable manufacturing processes for critical pharmacophores. As detailed in the recent patent CN112441981A, a groundbreaking methodology has been established for the synthesis of 1,2,5-polysubstituted imidazole derivatives, a structural motif ubiquitous in high-value therapeutic agents. This innovation addresses long-standing challenges in organic synthesis by enabling the direct construction of the imidazole core through a highly efficient multicomponent reaction. The significance of this technology cannot be overstated for the global pharmaceutical supply chain, as imidazole derivatives serve as essential intermediates for a wide array of bioactive molecules, including antifungal and antiprotozoal drugs like metronidazole. By leveraging a novel catalytic system, this approach transforms the production paradigm from laborious multi-step sequences into a streamlined, one-pot operation.
The strategic value of this patent lies in its ability to access complex chemical space with unprecedented simplicity. Traditional routes to polysubstituted imidazoles often suffer from limited substrate scope, requiring pre-functionalized starting materials that are expensive and difficult to source. In contrast, the disclosed method utilizes readily available styryl azides, aromatic aldehydes, and aromatic amines as building blocks. This shift not only democratizes access to diverse imidazole libraries for drug discovery but also lays a robust foundation for commercial manufacturing. For R&D directors and procurement strategists, understanding the nuances of this technology is crucial for securing a competitive edge in the development of next-generation active pharmaceutical ingredients (APIs) and fine chemicals.
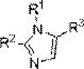
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,2,5-polysubstituted imidazoles has been plagued by significant inefficiencies that hinder both research throughput and industrial scalability. Conventional methodologies typically rely on the condensation of 1,2-dicarbonyl compounds with amines and aldehydes, a process that often necessitates harsh reaction conditions, such as high temperatures and strong acidic or basic environments. These aggressive conditions frequently lead to poor regioselectivity, resulting in complex mixtures of isomers that are notoriously difficult to separate, thereby drastically reducing the overall yield and purity of the desired product. Furthermore, the requirement for specialized, pre-oxidized precursors increases the step count and the associated cost of goods sold (COGS), making the final intermediates prohibitively expensive for large-scale applications.
Another critical bottleneck in traditional synthesis is the limited tolerance for functional groups. Many classical protocols fail when substrates contain sensitive moieties such as halogens, ethers, or heterocycles, which are often essential for the biological activity of the final drug molecule. This lack of universality forces chemists to employ protecting group strategies, adding further steps, waste, and time to the synthesis timeline. From a supply chain perspective, the reliance on multiple discrete steps introduces multiple points of failure, increasing the risk of batch-to-batch variability and extending lead times. Consequently, there has been a persistent industry demand for a more robust, selective, and operationally simple method to construct this vital heterocyclic scaffold without compromising on quality or cost.
The Novel Approach
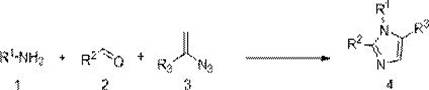
The innovative strategy presented in patent CN112441981A represents a paradigm shift by introducing a rhodium-catalyzed multicomponent reaction that directly assembles the imidazole ring from simple, commercially available feedstocks. This one-step protocol merges styryl azide, aromatic aldehyde, and aromatic amine in a single reaction vessel, achieving high atom economy and exceptional regioselectivity. The use of a dual-catalyst system comprising rhodium acetate and phosphoric acid allows the reaction to proceed under relatively mild thermal conditions, typically between 90°C and 110°C, which preserves the integrity of sensitive functional groups. This operational simplicity eliminates the need for intermediate isolation and purification, significantly compressing the manufacturing timeline and reducing solvent consumption.
Beyond mere convenience, this novel approach offers superior control over the chemical outcome. The catalytic system ensures that the substitution pattern is strictly controlled at the 1, 2, and 5 positions of the imidazole ring, minimizing the formation of unwanted regioisomers. This high fidelity is critical for pharmaceutical applications where impurity profiles are tightly regulated. Moreover, the method demonstrates remarkable substrate universality, successfully accommodating a broad spectrum of electronic and steric variations on the aromatic rings, including electron-withdrawing halogens and electron-donating alkoxy groups. For manufacturers, this translates to a versatile platform technology capable of producing a wide library of derivatives using a standardized process, thereby simplifying inventory management and enhancing supply chain resilience against raw material fluctuations.
Mechanistic Insights into Rhodium-Catalyzed Multicomponent Cyclization
The success of this synthesis hinges on the sophisticated interplay between the rhodium catalyst and the reactive intermediates generated in situ. The mechanism initiates with the decomposition of the styryl azide by the rhodium acetate catalyst to form a reactive rhodium-carbene species. This highly electrophilic intermediate is the key driver of the reaction, facilitating the subsequent insertion and cyclization steps that construct the imidazole core. The presence of phosphoric acid as a co-catalyst plays a pivotal role in proton transfer events, stabilizing transition states and promoting the elimination of nitrogen gas, which serves as the thermodynamic driving force for the reaction. This synergistic catalysis ensures that the reaction pathway is energetically favorable, allowing it to proceed efficiently even with low catalyst loadings.
From an impurity control perspective, the mechanistic precision of this catalytic cycle is a major advantage. Because the reaction proceeds through a well-defined organometallic pathway, side reactions such as polymerization of the azide or non-specific condensation of the aldehyde are effectively suppressed. The high regioselectivity observed is a direct result of the steric and electronic environment created by the ligand sphere around the rhodium center, which directs the attack of the amine and aldehyde components to specific positions on the carbene intermediate. This minimizes the generation of difficult-to-remove isomeric impurities, simplifying the downstream purification process. For quality assurance teams, this means that the crude product profile is much cleaner, reducing the burden on analytical laboratories and ensuring that the final API intermediate meets stringent purity specifications with less processing effort.
How to Synthesize 1,2,5-Polysubstituted Imidazole Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize yield and safety. The process is designed to be robust, utilizing common organic solvents and standard heating equipment, which facilitates easy technology transfer. The reaction involves mixing the three key components—styryl azide, aromatic aldehyde, and aromatic amine—along with the catalytic system in a suitable solvent such as toluene or 1,2-dichloroethane. The mixture is then heated to reflux temperatures, typically around 100°C, and maintained for a period of 12 to 18 hours to ensure complete conversion. Following the reaction, the workup is straightforward, involving solvent removal and standard chromatographic purification, making it accessible for both small-scale discovery and larger-scale production campaigns.
- Combine styryl azide, aromatic aldehyde, and aromatic amine in a reaction vessel with rhodium acetate and phosphoric acid catalysts.
- Add organic solvent such as toluene or 1,2-dichloroethane and heat the mixture to 90-110°C for 12-18 hours.
- Remove solvent under reduced pressure and purify the crude product via column chromatography using ethyl acetate and petroleum ether.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic methodology offers tangible economic and logistical benefits that directly impact the bottom line. The primary advantage lies in the drastic simplification of the manufacturing process. By collapsing what was traditionally a multi-step synthesis into a single pot, the method significantly reduces labor costs, energy consumption, and equipment occupancy time. This intensification of the process allows for higher throughput within existing facility footprints, effectively increasing capacity without the need for capital-intensive expansion. Furthermore, the use of commodity chemicals as starting materials mitigates the risk of supply disruptions associated with specialized, custom-synthesized precursors, ensuring a more stable and predictable supply chain for critical intermediates.
- Cost Reduction in Manufacturing: The economic viability of this process is bolstered by the exceptionally low loading of the precious metal catalyst. With rhodium acetate required at only 2% to 5% molar ratio, the cost contribution of this expensive metal is minimized, addressing a major pain point in transition metal-catalyzed processes. Additionally, the high atom economy of the multicomponent reaction means that a greater proportion of the raw material mass is incorporated into the final product, reducing waste disposal costs. The elimination of intermediate isolation steps further cuts down on solvent usage and processing time, leading to substantial overall cost savings in the production of high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The reliance on widely available building blocks such as substituted anilines, benzaldehydes, and styrene derivatives ensures a robust supply base. These commodities are produced globally at massive scales, meaning that procurement teams can source them from multiple vendors to mitigate geopolitical or logistical risks. The simplicity of the reaction conditions also reduces the dependency on specialized equipment or extreme operating parameters, making the process easier to replicate across different manufacturing sites. This flexibility enhances supply continuity, allowing companies to maintain consistent inventory levels and meet tight delivery schedules for their downstream customers in the pharmaceutical and agrochemical sectors.
- Scalability and Environmental Compliance: Scaling this reaction from gram to kilogram or ton scale is facilitated by its thermal stability and lack of hazardous reagents. The reaction operates at moderate temperatures and does not require high-pressure equipment, simplifying the engineering controls needed for large-scale reactors. From an environmental standpoint, the high selectivity and yield reduce the generation of chemical waste, aligning with green chemistry principles and easing the burden on waste treatment facilities. The use of recoverable solvents like toluene further supports sustainability goals, making this process attractive for companies aiming to reduce their carbon footprint while maintaining high production efficiency.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims within the patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this method into their existing portfolios and assess the potential return on investment for process development initiatives.
Q: What are the key advantages of this Rh-catalyzed synthesis over traditional methods?
A: This method offers high atom economy and regioselectivity in a single step, eliminating the need for complex multi-step sequences and harsh conditions typical of conventional imidazole synthesis.
Q: Is the catalyst loading cost-effective for large-scale production?
A: Yes, the process utilizes extremely low catalyst loadings, with rhodium acetate at only 2-5 mol% and phosphoric acid at 10-20 mol%, significantly reducing heavy metal costs and purification burdens.
Q: What is the substrate scope for this reaction?
A: The reaction demonstrates wide universality, tolerating various substituents including halogens, alkyl groups, alkoxy groups, and heterocycles like thiophene and furan on all three coupling partners.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2,5-Polysubstituted Imidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic methodologies like the one described in CN112441981A for accelerating drug development and commercialization. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to market supply is seamless. Our state-of-the-art facilities are equipped to handle complex organometallic chemistry with the highest safety standards, and our rigorous QC labs guarantee that every batch of 1,2,5-polysubstituted imidazole meets stringent purity specifications required by global regulatory bodies. We are committed to delivering high-quality intermediates that empower your R&D teams to focus on innovation rather than supply constraints.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your target molecule, demonstrating how this efficient route can optimize your budget. Please contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your custom synthesis projects. Let us be your strategic partner in navigating the complexities of modern chemical manufacturing.
