Advanced One-Step Synthesis of 1,2,5-Polysubstituted Imidazoles for Commercial Pharmaceutical Manufacturing
Introduction to Next-Generation Imidazole Synthesis
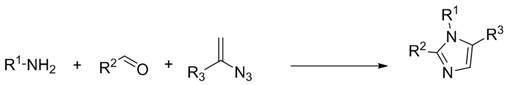
The pharmaceutical and fine chemical industries continuously seek robust methodologies for constructing nitrogen-containing heterocycles, particularly the imidazole scaffold, which serves as a critical pharmacophore in numerous bioactive molecules. Patent CN112441981B introduces a groundbreaking advancement in this domain by disclosing a highly efficient synthesis method for 1,2,5-polysubstituted imidazole derivatives. This technology addresses long-standing challenges in organic synthesis by enabling the direct construction of these complex heterocyclic systems through a streamlined one-step multicomponent reaction. The significance of this innovation lies in its ability to bypass traditional, labor-intensive synthetic routes, thereby offering a pathway to generate diverse chemical libraries with exceptional speed and precision. For research and development teams focused on drug discovery, particularly in the realms of antifungal and antiprotozoal agents similar to metronidazole, this methodology represents a pivotal shift towards more sustainable and economically viable manufacturing processes.
Furthermore, the versatility of this synthetic approach allows for the incorporation of a wide array of functional groups at the R1, R2, and R3 positions, including phenyl, naphthyl, and various heteroaryl moieties. This structural diversity is paramount for medicinal chemists aiming to optimize the physicochemical properties and biological activity of lead compounds. By leveraging the specific catalytic system described in the patent, manufacturers can achieve high yields and superior regioselectivity, ensuring that the final product meets the stringent purity specifications required for active pharmaceutical ingredient (API) production. As we delve deeper into the technical specifics, it becomes evident that this process is not merely a laboratory curiosity but a scalable solution designed to enhance the reliability of the global supply chain for critical pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polysubstituted imidazoles has been plagued by significant inefficiencies that hinder both research throughput and commercial scalability. Traditional methods often rely on multi-step sequences involving the condensation of 1,2-dicarbonyl compounds with amines and aldehydes, which frequently suffer from poor regioselectivity and the formation of difficult-to-separate isomeric mixtures. These conventional pathways typically require harsh reaction conditions, including extreme temperatures or the use of corrosive reagents, which pose safety risks and increase the complexity of waste management. Moreover, the starting materials for these older methods, such as specific alpha-halo ketones or pre-functionalized amidines, are often expensive, commercially scarce, or require tedious preparation themselves. This reliance on complex precursors drastically inflates the cost of goods sold (COGS) and extends the lead time for obtaining key intermediates, creating bottlenecks in the drug development pipeline.
The Novel Approach
In stark contrast, the methodology outlined in patent CN112441981B revolutionizes the field by employing a direct three-component coupling reaction. This novel approach utilizes readily available styryl azides, aromatic aldehydes, and aromatic amines as the fundamental building blocks, effectively collapsing what was once a multi-step synthesis into a single operational unit. The reaction proceeds with high atom economy, meaning that a greater proportion of the starting material atoms are incorporated into the final product, minimizing waste generation. By utilizing a dual-catalyst system involving rhodium acetate and a specific phosphoric acid, the process achieves remarkable control over the reaction trajectory, ensuring that the desired 1,2,5-substitution pattern is formed exclusively. This elimination of side products simplifies downstream processing and purification, directly translating to reduced operational costs and higher overall process efficiency for industrial partners seeking a reliable pharmaceutical intermediate supplier.
Mechanistic Insights into Rhodium-Catalyzed Cyclization
At the heart of this transformative synthesis lies a sophisticated catalytic cycle driven by the synergistic action of rhodium(II) acetate and a chiral binaphthyl-derived phosphoric acid. The mechanism initiates with the activation of the styryl azide by the rhodium catalyst, leading to the formation of a highly reactive rhodium-nitrenoid species. This electrophilic intermediate is crucial as it facilitates the subsequent insertion into the carbon-hydrogen bonds or addition across unsaturated systems with high precision. The presence of the phosphoric acid co-catalyst, as depicted in the structural formula below, plays a dual role: it acts as a Brønsted acid to activate the carbonyl component of the aldehyde and potentially stabilizes the transition state through hydrogen bonding interactions. This cooperative catalysis ensures that the cyclization occurs with exceptional regioselectivity, directing the formation of the imidazole ring specifically at the 1,2,5-positions rather than alternative isomers.
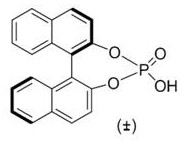
Understanding the impurity profile is critical for R&D directors overseeing process validation. The high selectivity of this rhodium-catalyzed system inherently suppresses the formation of common byproducts associated with non-catalyzed thermal decomposition of azides. The specific stoichiometry, maintaining a molar ratio of styryl azide to aldehyde and amine between 2.0:1.0:1.0 and 3.0:1.5:1.5, is optimized to drive the reaction to completion while minimizing the accumulation of unreacted starting materials. Furthermore, the choice of solvent, typically toluene or 1,2-dichloroethane, provides an ideal medium for solubilizing the organic substrates while withstanding the reaction temperature of 90-110°C without degrading. This mechanistic robustness ensures that the resulting 1,2,5-polysubstituted imidazole derivatives possess a clean impurity spectrum, facilitating easier purification via standard column chromatography and ensuring compliance with rigorous quality control standards required for high-purity OLED material or API intermediate applications.
How to Synthesize 1,2,5-Polysubstituted Imidazole Efficiently
Implementing this synthesis route in a practical setting requires careful attention to the specific reaction parameters defined in the patent to ensure optimal yield and reproducibility. The process is designed to be operationally simple, requiring standard laboratory glassware and heating equipment, which lowers the barrier to entry for contract manufacturing organizations. The key to success lies in the precise preparation of the reaction mixture, where the order of addition and the thorough mixing of the dual-catalyst system are paramount. Operators must maintain the reaction temperature within the specified range of 90-110°C, preferably at 100°C, for a duration of 12 to 18 hours to allow the multicomponent coupling to reach full conversion. Following the reaction, the workup procedure is straightforward, involving solvent removal under reduced pressure followed by purification.
- Combine styryl azide, aromatic aldehyde, aromatic amine, rhodium acetate catalyst, and phosphoric acid co-catalyst in a reaction vessel.
- Add an organic solvent such as toluene or 1,2-dichloroethane to form a homogeneous mixed solution.
- Heat the mixture to 90-110°C for 12-18 hours, then purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis method offers compelling strategic advantages that extend beyond mere technical feasibility. The primary value proposition is the drastic simplification of the supply chain for raw materials. Unlike traditional methods that depend on specialized, hard-to-source precursors, this process utilizes commodity chemicals such as styryl azides, benzaldehydes, and anilines, which are produced on a massive global scale. This abundance ensures a stable supply base, mitigating the risk of production delays caused by raw material shortages. Additionally, the reduction in synthetic steps directly correlates to a significant reduction in manufacturing costs. By eliminating intermediate isolation and purification stages, the facility saves on solvent consumption, energy usage, and labor hours, resulting in substantial cost savings that can be passed down to the end customer or reinvested into further R&D initiatives.
- Cost Reduction in Manufacturing: The economic impact of this one-pot synthesis is profound due to the elimination of multiple unit operations. Traditional multi-step syntheses incur cumulative losses in yield at each stage; by consolidating the process into a single reaction vessel, the overall yield is maximized, and material waste is minimized. Furthermore, the catalyst loading is exceptionally low, with rhodium acetate required at only 2 mol% and phosphoric acid at 10 mol%, which reduces the expense associated with precious metal catalysts. This efficiency translates to a lower cost per kilogram of the final high-purity pharmaceutical intermediate, enhancing the competitiveness of the final drug product in the marketplace.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of robust and widely available starting materials. The generic nature of the substrates means that procurement teams are not locked into single-source suppliers for exotic reagents. The reaction conditions are also relatively mild, operating at atmospheric pressure and moderate temperatures, which reduces the need for specialized high-pressure reactors or cryogenic cooling systems. This operational simplicity allows for flexible manufacturing scheduling and faster turnaround times, effectively reducing lead time for high-purity pharmaceutical intermediates and ensuring consistent delivery to downstream API manufacturers.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, the high atom economy of this reaction aligns perfectly with green chemistry principles. The minimization of byproducts reduces the burden on waste treatment facilities and lowers the environmental footprint of the manufacturing process. The use of common organic solvents like toluene allows for established recycling protocols, further enhancing sustainability. Scalability is inherent in the design; the reaction does not exhibit exothermic spikes that are difficult to manage at large scales, making the commercial scale-up of complex polymer additives or pharmaceutical intermediates feasible from pilot plant to multi-ton production without significant process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity for stakeholders evaluating the feasibility of this route for their specific projects. Understanding these nuances is essential for making informed decisions about process adoption and partnership opportunities.
Q: What are the key advantages of this imidazole synthesis method over traditional routes?
A: This method utilizes a one-pot multicomponent reaction that eliminates the need for complex multi-step sequences. It offers high atom economy, excellent regioselectivity, and operates under relatively mild thermal conditions (90-110°C) with low catalyst loading.
Q: What is the substrate scope for this rhodium-catalyzed reaction?
A: The process demonstrates wide substrate universality, accommodating various substituted aromatic amines, aldehydes, and styryl azides, including those with halogen, alkyl, alkoxy, and heterocyclic groups like thiophene and furan.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the use of cheap and readily available raw materials, combined with simple operational safety and standard purification techniques like column chromatography, makes it highly scalable for industrial pharmaceutical intermediate manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2,5-Polysubstituted Imidazole Supplier
As the global demand for complex heterocyclic building blocks continues to rise, partnering with an experienced CDMO is essential for translating innovative patent technologies into commercial reality. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the laboratory bench to full-scale manufacturing. Our team of expert chemists is well-versed in handling sensitive catalytic systems, including rhodium-mediated transformations, and our facilities are equipped with stringent purity specifications and rigorous QC labs to guarantee that every batch of 1,2,5-polysubstituted imidazole meets the highest industry standards. We understand the critical nature of timeline and quality in the pharmaceutical sector and are committed to delivering excellence at every stage of the production lifecycle.
We invite you to leverage our technical expertise to optimize your supply chain and reduce your overall manufacturing costs. By collaborating with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage potential partners to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a stable, cost-effective supply of these vital intermediates, empowering your organization to focus on what matters most: developing life-saving therapies and advancing chemical innovation.
