Advanced One-Step Synthesis of Polysubstituted 1,2-Benzothiazine Derivatives for Pharmaceutical Applications
Introduction to Novel 1,2-Benzothiazine Synthesis Technology
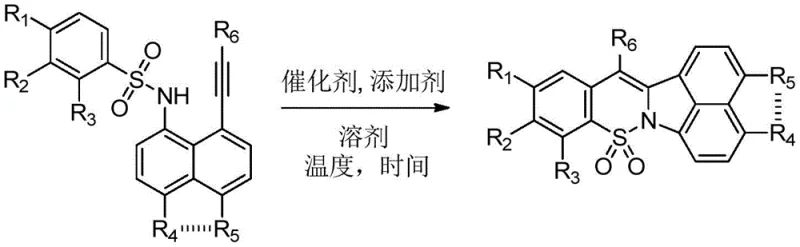
The pharmaceutical industry continuously seeks robust and economical pathways to access heterocyclic scaffolds that serve as the backbone for potent bioactive molecules. Patent CN111362973A introduces a groundbreaking methodology for the synthesis of polysubstituted 1,2-benzothiazine derivatives, a class of compounds renowned for their anti-inflammatory, anti-tumor, and antibacterial properties. This technology addresses critical bottlenecks in the current manufacturing landscape by replacing complex, multi-step sequences with a streamlined, one-step oxidative cyclization protocol. By leveraging inexpensive transition metal catalysis, specifically copper-based systems, this invention offers a viable alternative to legacy processes that rely on costly noble metals or hazardous rearrangement reactions. For R&D directors and procurement strategists, this represents a significant opportunity to optimize the supply chain for oxicam-class drugs and related therapeutic agents.
The core innovation lies in the direct transformation of 8-alkynylnaphthalenesulfonamide precursors into the target benzothiazine core through a highly efficient intramolecular cyclization. This approach not only simplifies the operational workflow but also enhances the overall atom economy of the process. The patent details a broad substrate scope, accommodating various substituents on the aromatic rings and alkyne termini, which underscores the versatility of this method for generating diverse chemical libraries. Such flexibility is paramount for medicinal chemists aiming to explore structure-activity relationships (SAR) without being constrained by synthetic limitations. Furthermore, the use of commodity solvents and mild reaction conditions positions this technology as a frontrunner for sustainable and scalable fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1,2-benzothiazine skeleton has been fraught with synthetic challenges that impede efficient large-scale production. Traditional routes often commence with sodium saccharin, necessitating a sequence of N-alkylation followed by a Gabriel-Colman rearrangement to expand the ring system. This multi-step trajectory is inherently inefficient, suffering from cumulative yield losses at each stage and requiring rigorous purification between steps. Moreover, alternative modern methods reported in literature frequently depend on palladium or ruthenium catalysts, which are not only prohibitively expensive but also pose significant supply chain risks due to their status as critical raw materials. The reliance on these precious metals also introduces stringent regulatory hurdles regarding residual metal limits in final pharmaceutical products, necessitating additional and costly purification steps to ensure compliance.
The Novel Approach
In stark contrast, the methodology disclosed in CN111362973A circumvents these obstacles by employing a direct, copper-catalyzed oxidative cyclization strategy. This novel route utilizes readily available 8-alkynylnaphthalenesulfonamides as starting materials, which undergo a seamless transformation into the desired 1,2-benzothiazine derivatives in a single operational step. The substitution of precious palladium or ruthenium catalysts with abundant and inexpensive copper salts drastically reduces the raw material costs associated with the synthesis. Additionally, the reaction conditions are remarkably benign, utilizing common aprotic solvents like dimethyl sulfoxide (DMSO) and operating at moderate temperatures, which simplifies reactor requirements and energy consumption. This paradigm shift from complex multi-step syntheses to a concise one-pot procedure exemplifies the kind of process intensification that modern chemical manufacturing demands.
Mechanistic Insights into Copper-Catalyzed Oxidative Cyclization
The mechanistic underpinning of this transformation involves a sophisticated interplay between the copper catalyst and the oxidant to facilitate C-N and C-C bond formation concurrently. The reaction likely proceeds through a copper-mediated activation of the alkyne moiety, rendering it susceptible to nucleophilic attack by the adjacent sulfonamide nitrogen. The presence of an oxidant, such as tert-butyl peroxide, is crucial for regenerating the active catalytic species and driving the oxidative aromatization of the intermediate dihydro-species to the final stable benzothiazine system. This oxidative cyclization mechanism avoids the need for pre-functionalized halogenated substrates often required in cross-coupling reactions, thereby reducing the generation of stoichiometric salt waste. Understanding this mechanism allows process chemists to fine-tune reaction parameters, such as the ratio of oxidant to catalyst, to maximize turnover numbers and minimize side reactions.
Impurity control is another critical aspect where this mechanism offers distinct advantages over traditional pathways. In multi-step syntheses involving rearrangements, the formation of regioisomers and polymeric byproducts is common, complicating downstream purification. However, the concerted nature of this copper-catalyzed cyclization tends to favor the formation of the thermodynamically stable 1,2-benzothiazine core with high selectivity. The use of specific additives further suppresses potential side reactions, such as alkyne polymerization or over-oxidation of the sensitive sulfonamide group. For quality assurance teams, this implies a cleaner crude reaction profile, which translates to higher recovery rates during crystallization or chromatography. The ability to predictably manage the impurity profile is essential for meeting the rigorous purity specifications required for pharmaceutical intermediates intended for human consumption.
How to Synthesize Polysubstituted 1,2-Benzothiazine Derivatives Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific procedural guidelines to ensure reproducibility and safety. The process begins with the precise weighing of the 8-alkynylnaphthalenesulfonamide substrate, the copper catalyst, and the peroxide additive, typically maintaining a molar ratio that favors slight excesses of the oxidant to drive the reaction to completion. These components are suspended or dissolved in a polar aprotic solvent, with dimethyl sulfoxide (DMSO) being the preferred medium due to its excellent solvating power and thermal stability. The reaction mixture is then heated to a temperature range of 70°C to 150°C, with 110°C identified as an optimal balance between reaction rate and energy efficiency. Detailed standardized operating procedures for scaling this reaction are provided in the technical guide below.
- Charge a reaction vessel with 8-alkynylnaphthalenesulfonamide, a copper catalyst (e.g., CuI), and an oxidant additive (e.g., tert-butyl peroxide) in a molar ratio of approximately 1: 0.2:2.0.
- Add an aprotic solvent such as dimethyl sulfoxide (DMSO) to dissolve the reactants completely and heat the mixture to a temperature between 70°C and 150°C.
- Stir the reaction for up to 24 hours, then cool, quench with water, extract with ethyl acetate, and purify the crude product via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers profound benefits for procurement managers and supply chain directors tasked with optimizing cost structures and ensuring material availability. The primary driver of value creation is the drastic reduction in catalyst costs; shifting from palladium or ruthenium to copper represents a magnitude of savings in raw material expenditure, especially when scaled to metric ton quantities. Furthermore, the simplification of the process from multiple steps to a single pot operation significantly reduces labor costs, solvent consumption, and waste disposal fees. These operational efficiencies compound to lower the overall cost of goods sold (COGS), providing a competitive edge in the pricing of high-purity pharmaceutical intermediates. The robustness of the method also mitigates the risk of batch failures, ensuring a more reliable supply stream for downstream API manufacturers.
- Cost Reduction in Manufacturing: The elimination of expensive noble metal catalysts is the most immediate financial benefit of this technology. Copper salts are orders of magnitude cheaper than palladium complexes, and their usage in catalytic amounts further diminishes the cost burden. Additionally, the one-step nature of the reaction removes the need for intermediate isolation and purification stages, which are typically resource-intensive. This consolidation of unit operations leads to substantial savings in utility consumption, including heating, cooling, and vacuum distillation. Consequently, the total manufacturing cost per kilogram of the final 1,2-benzothiazine derivative is significantly reduced, allowing for more aggressive pricing strategies in the global market without compromising margin integrity.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available commodity chemicals as reagents and solvents. Unlike specialized ligands or rare earth metals that may suffer from geopolitical supply disruptions, copper iodide and tert-butyl peroxide are produced in vast quantities globally. This ubiquity ensures that production schedules are not held hostage by raw material shortages. Moreover, the simplicity of the reaction setup means that the process can be easily transferred between different manufacturing sites or contract manufacturing organizations (CMOs) without extensive re-validation. This flexibility is crucial for maintaining business continuity and meeting tight delivery windows for key pharmaceutical clients who rely on just-in-time inventory models.
- Scalability and Environmental Compliance: The environmental footprint of this process is markedly smaller than that of conventional methods, aligning with increasingly strict global regulations on industrial emissions and waste. The avoidance of halogenated solvents in favor of DMSO, coupled with the high atom economy of the cyclization, results in less hazardous waste generation. From a scalability standpoint, the reaction does not require exotic high-pressure equipment or cryogenic conditions, making it amenable to standard stainless steel reactors found in most fine chemical plants. This ease of scale-up facilitates the rapid transition from gram-scale laboratory optimization to multi-ton commercial production, ensuring that supply can quickly ramp up to meet market demand for anti-inflammatory drug precursors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical aspects of the technology. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The responses cover catalyst selection, substrate compatibility, and the specific advantages over legacy technologies.
Q: What are the primary advantages of this copper-catalyzed method over traditional saccharin-based routes?
A: Unlike traditional multi-step methods starting from sodium saccharin which require harsh Gabriel-Colman rearrangements, this novel approach utilizes a direct one-step oxidative cyclization. This significantly reduces processing time, minimizes waste generation, and eliminates the need for expensive palladium or ruthenium catalysts, thereby lowering overall production costs.
Q: Which catalysts and additives are preferred for optimal yield in this synthesis?
A: The patent identifies Copper(I) Iodide (CuI) as the preferred catalyst due to its high efficiency and low cost compared to silver or palladium alternatives. For the oxidant additive, tert-butyl peroxide (TBHP) is highlighted as the optimal choice to drive the oxidative cyclization effectively under moderate thermal conditions.
Q: Is this synthetic route scalable for industrial manufacturing of oxicam precursors?
A: Yes, the method is explicitly designed for scalability. It employs readily available commodity chemicals like DMSO and simple copper salts, operates under standard heating conditions (70-150°C), and avoids sensitive reagents, making it highly suitable for large-scale commercial production of anti-inflammatory drug intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2-Benzothiazine Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic methodology described in CN111362973A for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate this innovative laboratory protocol into a robust, commercial-scale manufacturing process. Our facilities are equipped to handle complex organic syntheses, ranging from initial process development at the 100 kgs level to full-scale commercial production exceeding 100 MT annually. We are committed to delivering products that meet stringent purity specifications, utilizing our rigorous QC labs to ensure that every batch of 1,2-benzothiazine derivative adheres to the highest quality standards required by the global pharmaceutical industry.
We invite potential partners to engage with our technical team to explore how this cost-effective synthesis can enhance your supply chain efficiency. By collaborating with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact our technical procurement team to request specific COA data for our reference standards and to discuss route feasibility assessments for your target molecules. Together, we can leverage this advanced copper-catalyzed technology to secure a sustainable and economical supply of critical anti-inflammatory intermediates for the global market.
