Advanced Liquid Phase Synthesis of Etelcalcetide via Cbz Protection for Commercial Scale-up
The pharmaceutical landscape for calcimimetics has been significantly advanced by the disclosure of patent CN111925418B, which details a robust liquid phase synthesis method for Etelcalcetide, also known as the ittaka peptide. This novel therapeutic agent functions by binding to and activating calcium-sensing receptors on the parathyroid gland, effectively inhibiting parathyroid hormone secretion. While previous methodologies relied heavily on solid-phase synthesis or less efficient liquid routes, this patent introduces a paradigm shift by leveraging Cbz (benzyloxycarbonyl) protection strategies combined with precise disulfide bond engineering. The core innovation lies in the ability to control purity while avoiding peptide deletion sequences, a common pitfall in complex polypeptide manufacturing. By integrating Pd/C hydrogenation for deprotection, the process not only simplifies purification but also enhances the economic feasibility of large-scale production. This technical breakthrough offers a compelling value proposition for stakeholders seeking a reliable peptide intermediate supplier capable of delivering high-purity active pharmaceutical ingredients.
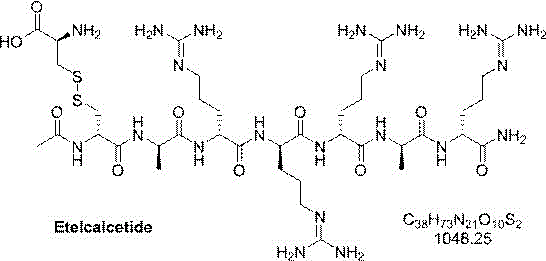
The structural complexity of Etelcalcetide, featuring multiple arginine residues and a critical disulfide bridge between D-cysteine and L-cysteine, demands a synthesis route that minimizes racemization and side reactions. The patent outlines a sequence where the N-terminal is acetylated and the C-terminal is amidated, requiring meticulous stepwise assembly. Unlike traditional approaches that struggle with the steric hindrance of poly-arginine sequences, this method utilizes specific activation agents like Oxymapure and EDCI to drive coupling efficiency. The result is a streamlined pathway that addresses the historical challenges of low crude purity and difficult downstream processing, positioning this technology as a cornerstone for cost reduction in pharmaceutical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Etelcalcetide has been plagued by inefficiencies inherent to both solid-phase and early liquid-phase techniques. Solid-phase methods, such as those described in WO 2017/114240, often suffer from low crude peptide purity, typically hovering around 81%, necessitating multiple rounds of expensive preparative HPLC purification. The reliance on resin-bound synthesis introduces limitations in scalability, as the physical capacity of the resin restricts batch sizes, and the cleavage process often generates significant amounts of hazardous waste. Furthermore, the construction of the disulfide bond on solid support is prone to misconnection, leading to complex impurity profiles that are difficult to separate. Early liquid-phase attempts, referenced in WO 2017/114238, improved some aspects but resulted in abysmal overall yields of approximately 11%, primarily due to the lengthy synthetic sequences and cumulative losses at each protection and deprotection step. These conventional methods create substantial bottlenecks for supply chain heads concerned with consistent availability and procurement managers focused on minimizing the cost of goods sold.
The Novel Approach
The methodology presented in CN111925418B fundamentally reengineers the synthesis workflow by adopting a full liquid-phase strategy anchored by Cbz protection. This approach circumvents the resin capacity limits of solid-phase synthesis, allowing for true solution-phase chemistry that is inherently more scalable. A critical differentiator is the use of Pd/C hydrogenation for removing the Cbz group, a mild and selective process that avoids the harsh acidic conditions associated with Boc deprotection or the basic conditions of Fmoc removal. This gentler deprotection preserves the integrity of the sensitive peptide backbone and reduces the formation of deletion peptides. Moreover, the patent introduces a specialized disulfide bond formation step using Boc-L-Cys(S-S-Py)-OtBu, which acts as a pre-activated intermediate. This ensures that the disulfide bridge forms accurately between the specific D-cysteine and L-cysteine residues, drastically reducing the formation of scrambled disulfide impurities. The cumulative effect of these innovations is a dramatic improvement in overall yield, rising from 11% in prior art to approximately 25%, alongside a final product purity exceeding 99.5% after purification.
Mechanistic Insights into Cbz-Protection and Disulfide Engineering
The chemical elegance of this synthesis lies in the strategic selection of the Cbz protecting group and its subsequent removal via heterogeneous catalysis. In standard peptide synthesis, the choice of protecting group dictates the entire downstream workflow. By employing Cbz-D-Arg(Pbf)-OH and related Cbz-amino acids as building blocks, the synthesis leverages the stability of the carbamate linkage during coupling while ensuring it can be cleanly removed under neutral conditions. The deprotection mechanism involves the adsorption of the Cbz-peptide onto the surface of the palladium on carbon catalyst, where hydrogen gas facilitates the hydrogenolysis of the benzylic C-O bond. This reaction releases the free amine, toluene, and carbon dioxide. Crucially, because the catalyst is a solid suspended in the liquid reaction medium, it can be recovered via simple filtration. The patent explicitly notes that the filtered Pd/C cake can be washed and reused in subsequent hydrogenation cycles without significant loss of catalytic activity. This recyclability is a major technical advantage, as it amortizes the cost of the noble metal catalyst over multiple batches, directly impacting the economic viability of the process.

Furthermore, the mechanism for disulfide bond formation is meticulously controlled to prevent the thermodynamic scrambling that often plagues cysteine-containing peptides. Instead of oxidizing two free thiols, which can lead to a mixture of homodimers and heterodimers, the process utilizes a pyridyl-disulfide activated intermediate, Boc-L-Cys(S-S-Py)-OtBu. When this reagent reacts with the free thiol of the D-cysteine residue on the growing peptide chain, it undergoes a thiol-disulfide exchange reaction. This reaction is highly specific and driven by the release of the pyridine-2-thione leaving group, which is stable and does not interfere with the peptide. This "pre-formed" strategy ensures that the L-cysteine is incorporated with the correct stereochemistry and oxidation state from the outset. The subsequent global deprotection using a TFA/Tips/Water cocktail removes the remaining acid-labile groups (Pbf, Boc, OtBu) simultaneously, releasing the final active Etelcalcetide. This mechanistic precision is what allows the process to achieve high purity levels, making it an ideal candidate for commercial scale-up of complex peptide intermediates.
How to Synthesize Etelcalcetide Efficiently
The synthesis protocol detailed in the patent provides a clear roadmap for replicating this high-yield process in a GMP environment. The procedure begins with the sequential coupling of Cbz-protected amino acids, starting from the C-terminus, utilizing activation agents like EDCI and Oxymapure to ensure high coupling efficiency. Following the assembly of the heptapeptide backbone, the side-chain protecting group on the cysteine is selectively removed to expose the reactive thiol.
- Perform liquid phase sequence synthesis using Cbz-protected amino acids to build the heptapeptide backbone, utilizing Pd/C hydrogenation for efficient deprotection.
- Remove the side-chain protecting group (Mmt or Trt) from the cysteine residue using TFA/DCM to expose the thiol group for bonding.
- React the exposed thiol with Boc-L-Cys(S-S-Py)-OtBu to form the disulfide bond accurately, followed by final global deprotection and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this specific liquid-phase synthesis method offers tangible strategic benefits beyond mere technical metrics. The primary advantage stems from the elimination of solid-phase resins, which are not only expensive consumables but also generate significant volumes of hazardous organic waste that require costly disposal. By moving to a solution-phase process, the manufacturing footprint is reduced, and the reliance on specialized resin suppliers is eliminated, thereby enhancing supply chain resilience. Additionally, the ability to recycle the Pd/C catalyst represents a direct reduction in raw material costs, as palladium is a high-value precious metal. The simplified purification profile, resulting from higher crude purity, means that fewer cycles of preparative HPLC are required, which reduces solvent consumption and extends the lifespan of expensive chromatography columns. These factors collectively contribute to substantial cost savings in pharmaceutical manufacturing, allowing for more competitive pricing of the final API.
- Cost Reduction in Manufacturing: The implementation of Cbz protection coupled with Pd/C hydrogenation creates a highly efficient cycle where the catalyst is not consumed but rather acts as a reusable tool. This contrasts sharply with stoichiometric reagents used in other deprotection methods. Furthermore, the avoidance of expensive solid-support resins removes a significant line item from the bill of materials. The higher overall yield of 25% compared to the 11% of previous liquid methods means that less starting material is required to produce the same amount of final product, effectively nearly doubling the material efficiency. This efficiency gain translates directly into a lower cost per gram of the active ingredient, providing a strong buffer against fluctuations in the prices of protected amino acids.
- Enhanced Supply Chain Reliability: Liquid phase synthesis is inherently more scalable than solid phase methods, which are often limited by the swelling properties and loading capacity of the resin. In a liquid system, reaction volumes can be increased simply by utilizing larger reactors, facilitating a smoother transition from pilot scale to multi-ton commercial production. This scalability ensures that supply chain heads can secure consistent volumes of Etelcalcetide to meet market demand without the bottlenecks associated with resin procurement. Moreover, the use of widely available reagents like Cbz-amino acids and standard activation agents reduces the risk of supply disruption compared to specialized, proprietary resins. The robustness of the process, evidenced by the high purity of intermediates, also reduces the risk of batch failures, ensuring a steady flow of product through the pipeline.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this process aligns well with green chemistry principles. The reduction in solvent usage, particularly the avoidance of the large volumes of DMF and DCM often required for resin washing in solid-phase synthesis, lowers the environmental burden. The ability to crystallize intermediates rather than relying solely on chromatography for purification further reduces solvent waste. The TFA/Tips/Water cleavage system is a standard industry practice, but its efficiency here minimizes the duration of exposure and the volume of acidic waste generated. These factors simplify the environmental permitting process for new manufacturing sites and reduce the operational costs associated with waste treatment facilities, making the technology attractive for production in regions with strict environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific advantages and procedural details outlined in patent CN111925418B, providing clarity for technical teams evaluating this route for adoption.
Q: Why is Cbz protection preferred over Fmoc for this specific peptide synthesis?
A: Cbz protection allows for deprotection via Pd/C catalytic hydrogenation, which avoids the harsh acidic conditions required for Boc removal or the base-labile conditions for Fmoc. This results in fewer side reactions, easier purification through simple filtration, and the ability to reuse the expensive palladium catalyst, significantly lowering production costs.
Q: How does this method improve the yield compared to prior art?
A: By utilizing a pre-formed disulfide bond intermediate (Boc-L-Cys(S-S-Py)-OtBu) rather than forming the bond on the resin or in a less controlled manner, the process minimizes misconnected disulfide impurities. This strategic control increases the overall yield from approximately 11% in previous liquid methods to around 25%.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the liquid phase approach described eliminates the need for expensive solid-phase resins and reduces solvent consumption significantly. The ability to crystallize intermediates and reuse the Pd/C catalyst makes the process highly scalable and economically viable for metric-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Etelcalcetide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex peptides like Etelcalcetide requires more than just a patent; it demands a partner with deep process engineering expertise. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. We understand the critical importance of maintaining stringent purity specifications throughout the synthesis, utilizing our rigorous QC labs to monitor every coupling and deprotection step. Our facility is equipped to handle the specific requirements of liquid phase peptide synthesis, including the safe handling of hydrogenation processes and the precise control of crystallization parameters necessary to achieve the high purity levels described in the patent.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be integrated into your supply chain. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this Cbz-based liquid phase method. We encourage you to request specific COA data and route feasibility assessments to validate the performance of our manufacturing capabilities against your project requirements. Let us collaborate to bring this vital calcimimetic therapy to patients efficiently and cost-effectively.
