Advanced Enantioselective Oxidation for High-Purity Proton Pump Inhibitor Intermediates
Advanced Enantioselective Oxidation for High-Purity Proton Pump Inhibitor Intermediates
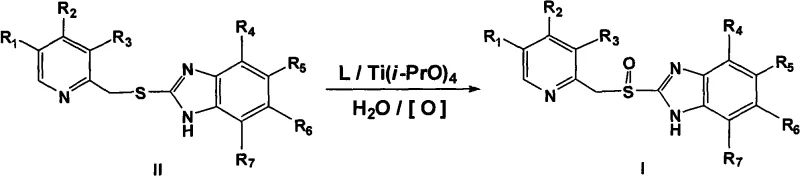
The pharmaceutical industry continuously seeks more efficient pathways for synthesizing chiral drugs, particularly proton pump inhibitors (PPIs) like omeprazole, lansoprazole, and pantoprazole. A pivotal advancement in this domain is detailed in patent CN101538264A, which discloses a novel method for preparing optically pure substituted [(pyridyl methylene) sulfinyl]-1H-benzimidazole sulfoxide compounds. This technology addresses the critical challenge of enantioselective synthesis by directly oxidizing prochiral thioethers into corresponding optically pure sulfoxides. Unlike traditional methods that often rely on the separation of racemic mixtures—a process inherently limited to a maximum 50% yield—this innovative approach utilizes a complex compound catalyst formed by accessible and stable (+)- or (-)-tartaric acid diamide ligands and titanium. For R&D directors and procurement specialists, this represents a significant leap forward in process chemistry, offering a route that is not only chemically elegant but also economically viable for large-scale manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrialization of chiral PPIs has faced substantial hurdles regarding efficiency and cost. Conventional methods often involve the oxidation of sulfides to produce racemic sulfoxides, followed by cumbersome resolution steps to isolate the active enantiomer. Techniques such as chromatographic separation or enzymatic resolution, while effective, introduce multiple processing stages that increase operational costs and extend lead times. Furthermore, older oxidation protocols frequently utilized expensive or unstable chiral reagents, such as diethyl tartrate, which can degrade under alkaline conditions, leading to inconsistent batch quality. The generation of sulfone byproducts is another persistent issue in traditional oxidation, requiring additional purification steps that erode overall yield. These inefficiencies create bottlenecks in the supply chain, making it difficult for manufacturers to meet the growing global demand for high-purity generic and branded PPIs without incurring prohibitive expenses.
The Novel Approach
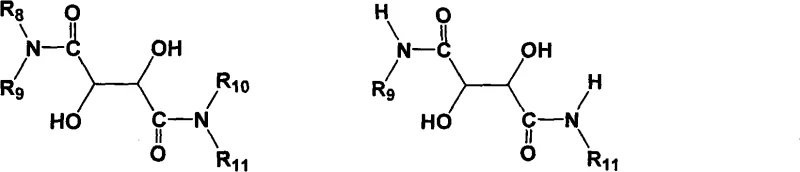
The methodology described in the patent fundamentally shifts the paradigm by employing a highly selective catalytic system that bypasses the need for resolution. By utilizing chiral D- or L-tartrate diamide bidentate ligands in conjunction with titanium(IV) reagents, the process achieves direct asymmetric oxidation. This means the chirality is induced during the bond-forming step itself, rather than corrected afterwards. The use of mild and cheap oxidizing agents, such as cumene hydroperoxide, further enhances the safety profile and cost-effectiveness of the reaction. Crucially, this method suppresses the formation of sulfone byproducts, ensuring a cleaner reaction profile that simplifies downstream processing. The ligands themselves are derived from tartaric acid, a renewable and abundant resource, and exhibit superior chemical stability compared to earlier generations of tartrate esters. This stability allows for potential recovery and reuse of the chiral inducer, adding another layer of economic benefit to the process.
Mechanistic Insights into Titanium-Tartrate Catalyzed Oxidation
At the heart of this technology lies the sophisticated interaction between the titanium center and the chiral ligand. The catalyst system is generated in situ by reacting a metal titanium reagent, preferably titanium tetraisopropoxide, with the chiral tartrate diamide ligand. The structure of the ligand is paramount; the patent highlights the efficacy of tartrate diamide compounds where the amide substituents can vary from hydrogen to alkyl, aryl, or even polymer-supported groups. This structural flexibility allows for fine-tuning the steric environment around the titanium atom, which is critical for discriminating between the two faces of the prochiral sulfur atom during oxidation. The presence of water in the reaction mixture, typically in a molar ratio of 0.1 to 1.2 relative to the thioether, plays a vital role in activating the catalyst species without decomposing it. This delicate balance ensures that the active oxidizing species is generated efficiently, facilitating the transfer of oxygen to the sulfur atom with high stereocontrol.
Impurity control is a major focus for any GMP-compliant manufacturing process, and this catalytic system excels in minimizing side reactions. The specific geometry of the titanium-ligand complex directs the oxidant, typically an alkyl hydroperoxide, to approach the sulfur atom from a specific trajectory. This directed approach not only ensures high enantiomeric excess (ee), reaching up to 95% ee in optimized embodiments, but also prevents over-oxidation to the sulfone stage. In many conventional oxidations, the sulfoxide product is more reactive than the starting sulfide, leading to sulfone accumulation. However, the electronic and steric properties of this novel catalyst system appear to deactivate the product towards further oxidation once the sulfoxide is formed. This intrinsic selectivity reduces the burden on purification teams and ensures that the final API intermediate meets stringent regulatory specifications for impurities.
How to Synthesize Chiral Sulfoxide Compounds Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize both yield and optical purity. The process generally involves dissolving the thioether precursor in an organic solvent such as toluene, followed by the sequential addition of the chiral ligand and the titanium reagent at elevated temperatures to ensure complex formation. After cooling the mixture to the reaction temperature, which can range from -78°C to 35°C depending on the specific substrate, the oxidant is introduced. The reaction is allowed to proceed for a defined period, typically between 3 to 48 hours, after which standard workup procedures involving aqueous extraction and pH adjustment isolate the product. While the general principles are straightforward, the specific stoichiometry and choice of additives can significantly influence the outcome. For a comprehensive understanding of the operational parameters, please refer to the standardized synthesis guide below.
- Prepare the catalyst system by mixing chiral tartrate diamide ligands with titanium tetraisopropoxide in an organic solvent like toluene.
- Add the prochiral thioether precursor and water to the reaction mixture under controlled temperature conditions ranging from -78°C to 35°C.
- Introduce the oxidizing agent, preferably cumene hydroperoxide, and maintain stirring for 3 to 48 hours to achieve high enantiomeric excess.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology translates into tangible strategic advantages beyond mere chemical elegance. The shift towards a direct asymmetric synthesis model fundamentally alters the cost structure of producing PPI intermediates. By eliminating the resolution step, manufacturers effectively double the theoretical yield from the same amount of starting material, which drastically reduces the raw material consumption per kilogram of final product. Furthermore, the reliance on stable, commercially available ligands derived from tartaric acid mitigates the risk of supply disruptions associated with exotic or proprietary chiral auxiliaries. The ability to operate at near-ambient temperatures (0°C to 35°C) in many embodiments also lowers energy consumption compared to cryogenic processes, contributing to a smaller carbon footprint and reduced utility costs.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of expensive resolution agents and the reduction of waste. Since the reaction produces minimal sulfone byproducts, the need for extensive chromatographic purification is diminished, allowing for simpler crystallization or extraction techniques. This streamlining of the downstream process significantly lowers the cost of goods sold (COGS). Additionally, the potential for recovering and reusing the chiral ligand from the reaction medium adds a circular economy aspect to the manufacturing process, further enhancing long-term cost efficiency without compromising on the quality of the chiral induction.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of robust and stable catalyst components. Unlike earlier tartrate ester systems that were sensitive to moisture and alkaline conditions, the tartrate diamide ligands described here offer superior stability, ensuring consistent performance across different batches and storage conditions. This reliability reduces the risk of batch failures and production delays. Moreover, the versatility of the system allows it to be applied to a range of PPIs including omeprazole, lansoprazole, and pantoprazole, enabling manufacturers to utilize a single platform technology for multiple products, thereby simplifying inventory management and supplier qualification processes.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, the use of mild oxidants like cumene hydroperoxide is advantageous. These reagents are safer to handle on a multi-ton scale compared to more hazardous peracids or chlorinated oxidants. The reaction generates fewer hazardous waste streams, aligning with increasingly strict environmental regulations globally. The process has been demonstrated to work effectively in common solvents like toluene, which are well-understood in industrial settings, facilitating a smoother transition from laboratory scale to commercial production. This ease of scale-up ensures that supply can be ramped up quickly to meet market surges without the need for specialized or hard-to-source equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this enantioselective oxidation technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into existing production lines.
Q: What represents the primary advantage of this novel oxidation method over traditional resolution techniques?
A: The primary advantage is the direct asymmetric oxidation which eliminates the need for separating racemic mixtures, thereby theoretically doubling the maximum yield compared to resolution methods while utilizing stable and accessible chiral ligands.
Q: Which oxidizing agents are compatible with this titanium-catalyzed system?
A: The system is designed to work effectively with mild and inexpensive alkyl peroxides, specifically cumene hydroperoxide, which minimizes the formation of unwanted sulfone byproducts often seen with harsher oxidants.
Q: Can this process be scaled for industrial production of pantoprazole and lansoprazole?
A: Yes, the patent explicitly demonstrates the synthesis of S-pantoprazole and S-lansoprazole with significant enantiomeric excess, indicating the robustness of the catalyst system for scaling up complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Omeprazole Intermediate Supplier
The technological breakthroughs detailed in patent CN101538264A underscore the evolving landscape of chiral synthesis, offering a pathway to higher purity and greater efficiency. At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating such advanced laboratory methodologies into robust commercial realities. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, ensuring that every batch of chiral sulfoxide intermediate meets the exacting standards required by global regulatory bodies. We are committed to leveraging our technical expertise to optimize these catalytic systems for your specific production needs.
We invite you to collaborate with us to explore the full potential of this novel synthesis route for your supply chain. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this technology can reduce your overall manufacturing spend. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a competitive edge in the market with reliable, high-quality pharmaceutical intermediates.
