Transforming Biodiesel Byproducts: A Technical Analysis of Continuous Epichlorohydrin Synthesis via Reactive Distillation
The global chemical landscape is undergoing a significant paradigm shift driven by the surplus of renewable feedstocks, specifically glycerol derived from biodiesel production. Patent CN101337950A introduces a groundbreaking methodology for the continuous preparation of epichlorohydrin, a critical intermediate for epoxy resins and pharmaceuticals, by leveraging this abundant byproduct. Unlike traditional batch processes that struggle with equilibrium limitations and energy inefficiency, this invention employs a sophisticated coupling of continuous catalytic chlorination and reactive distillation. The technology utilizes a homogeneous multi-component catalytic system comprising carboxylic acids and Lewis acids to facilitate the substitution of hydroxyl groups with chlorine atoms under mild thermal conditions. This approach not only addresses the environmental burden of glycerol disposal but also establishes a sustainable, closed-loop manufacturing pathway that aligns with modern green chemistry principles. For industrial stakeholders, this represents a viable alternative to petroleum-dependent routes, offering a robust framework for cost reduction in epoxy resin manufacturing while maintaining rigorous quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of epichlorohydrin has been dominated by the high-temperature chlorination of propylene, a process fraught with significant operational and environmental challenges. This legacy technology requires extreme reaction temperatures and pressures, leading to severe equipment corrosion due to the aggressive nature of chlorine gas and hydrogen chloride byproducts. Furthermore, the propylene route is inherently energy-intensive, demanding substantial inputs for heating and subsequent separation of complex chlorinated mixtures. The process generates large volumes of wastewater containing calcium chloride and organic chlorides, imposing heavy burdens on effluent treatment facilities and escalating operational expenditures. Additionally, the reliance on fossil-fuel-derived propylene exposes supply chains to volatile petrochemical markets, creating uncertainty in long-term procurement planning. The batch nature of many older glycerol conversion methods also suffers from poor heat transfer and limited conversion rates, often necessitating multiple recycling loops that degrade overall process efficiency.
The Novel Approach
In stark contrast, the methodology disclosed in CN101337950A revolutionizes the synthesis through a continuous reactive distillation architecture that seamlessly integrates reaction and separation. By employing a tailored homogeneous catalyst system, the process achieves high selectivity for 1,3-dichloro-2-propanol and 2,3-dichloro-1-propanol at temperatures ranging from 80°C to 150°C, drastically reducing thermal stress on reactor materials. The continuous removal of water via azeotropic distillation effectively drives the reversible hydrochlorination reaction to completion, maximizing atom economy without the need for excessive reagent excess. This continuous flow design ensures consistent product quality and facilitates real-time monitoring via gas chromatography, enabling precise control over impurity profiles. Moreover, the ability to utilize crude biological glycerol directly eliminates the capital and energy costs associated with feedstock purification, presenting a compelling value proposition for a reliable epichlorohydrin supplier seeking to optimize their raw material intake strategies.
Mechanistic Insights into Homogeneous Multi-component Catalytic Hydrochlorination
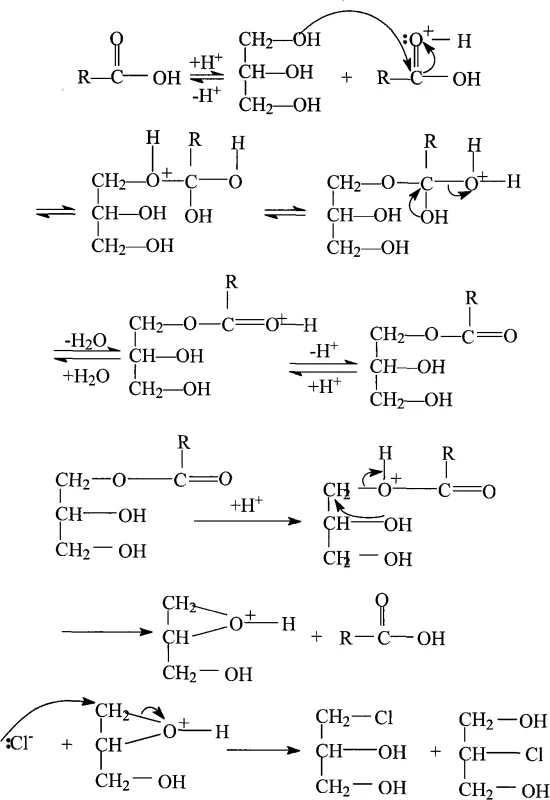
The core innovation of this process lies in its intricate catalytic mechanism, which orchestrates the transformation of polyhydroxy glycerol into chlorinated intermediates with exceptional precision. The reaction initiates with a nucleophilic substitution where the carboxylic acid component of the catalyst protonates the hydroxyl groups of glycerol, rendering them better leaving groups. This activation step is crucial for overcoming the kinetic barriers associated with displacing hydroxyl functionalities in an aqueous environment. Following protonation, the alkyl-oxygen bond cleaves to form a transient three-membered oxonium ion intermediate, a highly reactive species that stabilizes the transition state. The Lewis acid co-catalyst further enhances electrophilicity, coordinating with oxygen atoms to facilitate the departure of water molecules. This synergistic interaction between the Brønsted acid and Lewis acid components creates a highly active catalytic pocket that promotes rapid turnover rates while minimizing side reactions such as polymerization or ether formation.
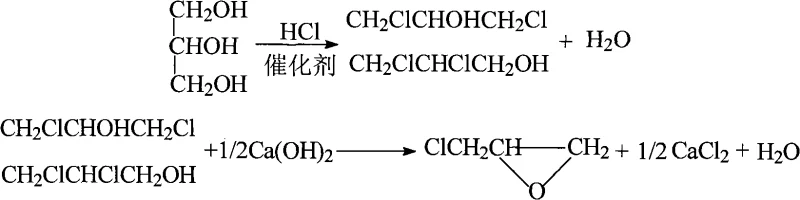
Subsequent to the formation of the oxonium ion, chloride ions generated from the dissociation of hydrogen chloride or metal chlorides perform a nucleophilic attack on the carbon centers. This SN2-type displacement opens the oxonium ring, resulting in the formation of monochlorohydrins which undergo further chlorination to yield the desired dichlorohydrins. The continuous removal of water is thermodynamically essential, as it prevents the hydrolysis of the chlorinated products back to glycerol, thereby locking in the yield. The catalyst system is designed to be recoverable; after the reaction, the organic acid and metal salts remain in the heavy fraction or can be separated via simple distillation, allowing for reuse in subsequent batches. This mechanistic elegance ensures that the process maintains high purity levels, typically exceeding 99.5%, which is critical for downstream applications in high-purity pharmaceutical intermediates and electronic-grade epoxy systems.
How to Synthesize Epichlorohydrin Efficiently
Implementing this continuous synthesis route requires careful attention to reactor design and process parameters to fully realize the benefits of the patented technology. The procedure involves feeding a mixture of glycerol and the multi-component catalyst into the top of a chlorination tower while introducing anhydrous hydrogen chloride gas from the bottom, establishing a counter-current flow that maximizes mass transfer efficiency. Operators must maintain strict temperature control within the 100°C to 110°C optimal range to balance reaction kinetics with selectivity, ensuring that the formation of trichloropropane byproducts is minimized. The overhead vapor, containing water and unreacted HCl, is condensed and separated, with the acid phase recycled to the reactor to enhance atom economy. Once the dichlorohydrin mixture is collected, it is transferred to a saponification unit where it reacts with an alkaline solution, such as calcium hydroxide or sodium hydroxide, to effect ring closure. For detailed operational parameters and safety protocols, please refer to the standardized synthesis guide below.
- Feed biodiesel-derived glycerol mixed with a homogeneous multi-component catalyst (carboxylic acid and Lewis acid) into a chlorination reaction tower while introducing anhydrous hydrogen chloride gas.
- Maintain reaction temperature between 80°C and 150°C to facilitate nucleophilic substitution, continuously removing water via azeotropic distillation to drive equilibrium.
- Collect the dichlorohydrin mixture, degas residual HCl, and react with alkaline solution (e.g., NaOH or Ca(OH)2) in a saponification tower to cyclize into epichlorohydrin.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, adopting this glycerol-based technology offers profound advantages in terms of supply chain resilience and cost structure optimization. The primary driver of value is the decoupling of epichlorohydrin production from the volatile petrochemical sector, replacing propylene with glycerol, a commodity whose price is inversely correlated with biodiesel demand. This shift mitigates exposure to crude oil price fluctuations and provides a more predictable cost baseline for long-term contracting. Furthermore, the continuous nature of the process significantly enhances throughput capabilities, allowing manufacturers to respond rapidly to spikes in market demand without the downtime associated with batch reactor cleaning and reloading. The simplified downstream processing, characterized by easy phase separation and minimal solvent usage, translates directly into reduced utility consumption and lower waste disposal fees. These operational efficiencies collectively contribute to a leaner manufacturing model that improves margin potential while adhering to increasingly stringent environmental regulations.
- Cost Reduction in Manufacturing: The elimination of energy-intensive feedstock purification steps and the ability to recycle catalyst components substantially lower the variable cost per kilogram of product. By utilizing crude glycerol directly, the process bypasses the capital expenditure required for high-vacuum distillation units typically needed to upgrade bio-glycerol to chemical grade. Additionally, the mild reaction conditions reduce the specification requirements for reactor construction materials, avoiding the need for expensive Hastelloy or titanium alloys often mandated by high-temperature chlorination processes. The integrated recovery of hydrogen chloride further minimizes raw material consumption, creating a closed-loop system that maximizes resource utilization and drives down overall production costs.
- Enhanced Supply Chain Reliability: Diversifying the raw material base to include renewable glycerol strengthens supply security against geopolitical disruptions affecting oil and gas supplies. The continuous operation mode ensures a steady, uninterrupted output stream, which is vital for maintaining just-in-time delivery schedules for downstream epoxy resin producers. The robustness of the catalytic system against feedstock impurities means that variations in glycerol quality from different biodiesel plants do not compromise production stability. This flexibility allows procurement teams to source glycerol from a wider geographic range, reducing logistics risks and fostering a more resilient supply network capable of withstanding regional shortages.
- Scalability and Environmental Compliance: The modular design of the reactive distillation columns facilitates straightforward scale-up from pilot plants to commercial-scale facilities, reducing the time-to-market for new production capacity. Environmentally, the process generates significantly less saline wastewater compared to traditional lime-based saponification methods, easing the burden on effluent treatment plants. The reduction in hazardous waste and the potential for recycling process water align with corporate sustainability goals and help manufacturers meet rigorous ISO 14001 standards. This eco-friendly profile not only reduces regulatory compliance costs but also enhances the brand reputation of the final product in markets that prioritize green chemistry and carbon footprint reduction.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this continuous glycerol-to-epichlorohydrin technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for engineering teams evaluating the feasibility of retrofitting existing facilities or designing new greenfield plants. The answers highlight the specific operational windows and material compatibilities that define the success of this catalytic route.
Q: What are the primary advantages of the glycerol route over traditional propylene chlorination?
A: The glycerol route utilizes renewable feedstocks from biodiesel production, operates under milder conditions (100-110°C vs. high-temperature propylene chlorination), and significantly reduces equipment corrosion and hazardous waste generation associated with chlorine gas handling.
Q: How does the reactive distillation process improve yield in this synthesis?
A: By continuously removing the water byproduct through azeotropic distillation during the reaction, the equilibrium is shifted towards the formation of dichlorohydrins according to Le Chatelier's principle, thereby increasing conversion rates without requiring excessive reagent ratios.
Q: Can crude glycerol from biodiesel be used directly without purification?
A: Yes, the patented process is designed to tolerate low-purity biological glycerol with high water content and impurities, eliminating the energy-intensive concentration steps typically required for chemical-grade glycerol.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Epichlorohydrin Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic processes like the one described in CN101337950A to redefine the economics of fine chemical production. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory concepts are translated into robust industrial realities. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications, guaranteeing that every batch of epichlorohydrin meets the exacting standards required for pharmaceutical and electronic applications. We are committed to leveraging our technical expertise to optimize reaction parameters, maximize yield, and minimize environmental impact, delivering a supply solution that is both economically competitive and scientifically superior.
We invite forward-thinking organizations to collaborate with us to explore the full commercial viability of this glycerol-based route. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions about integrating this sustainable technology into your supply chain. Together, we can drive the next generation of green chemical manufacturing, turning renewable resources into high-value intermediates with unmatched efficiency and reliability.
