Advanced Eltrombopag Synthesis: Safe Scalable Process for Global Pharmaceutical Supply Chains
Advanced Eltrombopag Synthesis: Safe Scalable Process for Global Pharmaceutical Supply Chains
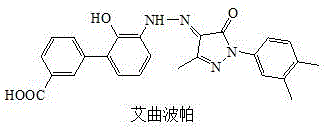
The pharmaceutical landscape for thrombopoietin receptor agonists continues to evolve, driven by the critical need for safer and more efficient manufacturing processes for complex small molecules. Eltrombopag, a pivotal oral nonpeptidic thrombopoietin receptor agonist approved for treating chronic Immune Thrombocytopenic Purpura (ITP), represents a high-value target for generic and specialty chemical manufacturers. Recent intellectual property developments, specifically patent CN114507186A, disclose a refined preparation method that addresses longstanding safety and scalability challenges inherent in earlier synthetic routes. This technical insight report analyzes the strategic advantages of this novel methodology, focusing on its potential to enhance supply chain reliability and reduce operational risks for global procurement teams. By shifting away from hazardous direct hydrogenation, this process offers a robust pathway for the commercial scale-up of complex pharmaceutical intermediates, ensuring consistent quality and availability for downstream drug product manufacturers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of key intermediates for Eltrombopag has relied heavily on catalytic hydrogenation using molecular hydrogen gas to reduce nitro groups to amines. While chemically effective on a small laboratory scale, this approach presents significant barriers to industrial implementation. The requirement for high-pressure hydrogenation equipment introduces substantial capital expenditure and rigorous safety compliance burdens, including the need for specialized explosion-proof facilities and continuous gas monitoring systems. Furthermore, the handling of hydrogen gas at scale increases the risk profile of the manufacturing site, potentially leading to production stoppages during safety audits or maintenance. From a supply chain perspective, reliance on this hazardous step can create bottlenecks, as not all contract manufacturing organizations possess the necessary infrastructure to perform high-pressure hydrogenation safely and consistently, thereby limiting the pool of qualified suppliers for this critical API intermediate.
The Novel Approach
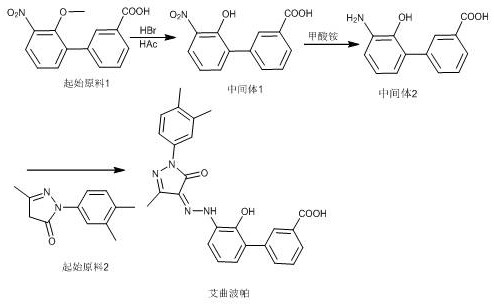
The methodology outlined in patent CN114507186A introduces a transformative shift by utilizing transfer hydrogenation with ammonium formate in the presence of a palladium catalyst. This chemical innovation effectively circumvents the need for direct hydrogen gas, replacing a high-risk unit operation with a safer, liquid-phase reduction step that can be conducted in standard glass-lined or stainless steel reactors. The process begins with the demethylation of the starting material using glacial acetic acid and hydrobromic acid, followed by the critical reduction step where ammonium formate serves as the hydrogen donor. This modification not only enhances operational safety but also simplifies the reaction workflow, allowing for faster completion times and easier workup procedures. By eliminating the dependency on high-pressure gas infrastructure, this route significantly lowers the barrier to entry for manufacturing partners, thereby expanding the potential supply base and enhancing the overall resilience of the Eltrombopag supply chain against disruptions.
Mechanistic Insights into Pd-C Catalyzed Transfer Hydrogenation
At the heart of this improved synthesis lies the efficient reduction of the nitro group on the biphenyl scaffold, a transformation that is crucial for the subsequent diazotization and coupling reactions. The use of ammonium formate as a hydrogen source in conjunction with a palladium on carbon (Pd/C) catalyst facilitates a transfer hydrogenation mechanism that is both chemoselective and mild. In this system, ammonium formate decomposes on the catalyst surface to generate reactive hydrogen species in situ, which then reduce the nitro group to the corresponding amine without affecting other sensitive functional groups such as the carboxylic acid or the ether linkage prior to demethylation. This selectivity is paramount for maintaining the integrity of the molecular structure and preventing the formation of over-reduced byproducts or side reactions that could complicate downstream purification. The reaction conditions, typically conducted in water at elevated temperatures around 85°C, provide an environmentally friendlier medium compared to organic solvents often required for traditional hydrogenation, aligning with modern green chemistry principles.
Impurity control is another critical aspect where this mechanistic approach offers distinct advantages over conventional methods. The patent data indicates that by carefully controlling the stoichiometry of the diazotization step, specifically maintaining a molar ratio of intermediate II to sodium nitrite to starting material II at approximately 1:1.05:1, the formation of side products is minimized. This precise control ensures that the single maximum impurity does not exceed 0.1% and the total impurity remains below 1.0%, meeting stringent regulatory requirements for pharmaceutical intermediates. The recrystallization process, utilizing solvent systems such as tetrahydrofuran and n-hexane, further purifies the crude product, removing residual catalysts and organic byproducts. This robust purification strategy ensures that the final Eltrombopag intermediate possesses the high purity necessary for subsequent coupling reactions, reducing the risk of batch failures and ensuring consistent quality for the final drug substance.
How to Synthesize Eltrombopag Efficiently
The synthesis of Eltrombopag via this patented route involves a streamlined three-step sequence that balances chemical efficiency with operational safety. The process initiates with the demethylation of the nitro-biphenyl starting material, followed by the key transfer hydrogenation step, and concludes with diazotization and coupling. Each stage is optimized to maximize yield while minimizing waste, making it an attractive option for manufacturers seeking to improve their production metrics. The detailed standardized synthesis steps, including specific temperature profiles, reaction times, and workup procedures, are critical for ensuring reproducibility and quality control across different production batches. For technical teams looking to implement this route, adherence to the specified molar ratios and solvent choices is essential to replicate the high purity and yield reported in the patent examples.
- Demethylation of Starting Material I using glacial acetic acid and hydrobromic acid at 120°C to yield Intermediate I.
- Reduction of Intermediate I using ammonium formate and a palladium catalyst in water at 85°C to generate Intermediate II.
- Diazotization of Intermediate II followed by coupling with Starting Material II and recrystallization to obtain final Eltrombopag.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route translates into tangible strategic benefits that extend beyond simple chemical yield. The primary advantage lies in the drastic simplification of the manufacturing infrastructure required. By eliminating the need for high-pressure hydrogenation, companies can utilize existing general-purpose reactor trains, thereby avoiding significant capital investment in specialized safety equipment. This flexibility allows for a broader range of manufacturing partners to qualify for production, increasing competition and potentially driving down processing costs. Furthermore, the reduced safety risk profile minimizes the likelihood of regulatory shutdowns or insurance premium hikes associated with hazardous gas handling, contributing to a more stable and predictable supply chain. The ability to source this intermediate from a wider pool of qualified suppliers enhances supply security, mitigating the risk of shortages that can impact downstream drug product availability.
- Cost Reduction in Manufacturing: The transition to transfer hydrogenation using ammonium formate eliminates the operational costs associated with high-pressure hydrogen gas, including storage, handling, and safety monitoring systems. This shift allows for the use of standard reaction vessels, significantly reducing capital expenditure and maintenance costs for manufacturing facilities. Additionally, the use of water as a solvent in the reduction step lowers solvent procurement and disposal costs, contributing to overall process economics. The simplified workup procedure reduces labor hours and energy consumption, further enhancing the cost-efficiency of the production process without compromising on product quality.
- Enhanced Supply Chain Reliability: By removing the dependency on specialized hydrogenation infrastructure, the supply base for this intermediate is significantly expanded, reducing the risk of single-source bottlenecks. The safer reaction profile minimizes the potential for production delays caused by safety incidents or rigorous regulatory inspections, ensuring more consistent lead times for buyers. The robustness of the process across different scales, from laboratory to commercial production, ensures that supply can be ramped up quickly to meet market demand fluctuations. This reliability is crucial for pharmaceutical companies managing tight inventory levels and seeking to maintain continuous production of their finished dosage forms.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability, with patent examples showing successful translation from gram to kilogram scales without loss of efficiency or purity. The use of aqueous conditions and the avoidance of hazardous gases align with increasingly strict environmental regulations, reducing the burden of waste treatment and emissions compliance. The high purity achieved through this route minimizes the need for extensive downstream purification, reducing solvent waste and energy usage. This environmental compatibility not only lowers compliance costs but also enhances the sustainability profile of the supply chain, a key metric for modern corporate procurement strategies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Eltrombopag synthesis route. These answers are derived directly from the technical specifications and beneficial effects described in patent CN114507186A, providing a reliable basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of adopting this method for their specific supply chain needs. The focus is on safety, purity, and operational efficiency, which are the primary drivers for process adoption in the fine chemical industry.
Q: How does this patent improve safety compared to traditional hydrogenation?
A: The process replaces high-pressure hydrogen gas reduction with ammonium formate transfer hydrogenation, significantly reducing explosion risks and infrastructure costs.
Q: What purity levels can be achieved with this synthesis route?
A: The method controls single maximum impurities to not exceed 0.1% and total impurities to not exceed 1.0%, with examples showing up to 99.5% purity.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the use of readily available raw materials and the avoidance of hazardous hydrogen gas makes the route highly operable and safe for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Eltrombopag Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes like the one described in CN114507186A to ensure the consistent supply of high-quality pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex chemistries are translated efficiently from the lab to the plant. Our facilities are equipped to handle the specific requirements of this transfer hydrogenation process, maintaining stringent purity specifications and operating within rigorous QC labs to guarantee that every batch meets the highest industry standards. We are committed to providing a secure and reliable supply of Eltrombopag intermediates, leveraging our technical expertise to optimize yield and minimize impurities for our global partners.
We invite procurement leaders and technical directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Our technical procurement team is ready to provide specific COA data and route feasibility assessments to demonstrate how our implementation of this patented process can enhance your supply chain resilience. By partnering with us, you gain access to a manufacturing capability that prioritizes safety, quality, and efficiency, ensuring that your production of Eltrombopag remains uninterrupted and cost-effective. Contact us today to discuss how we can support your long-term strategic goals in the thrombopoietin agonist market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
