Advanced Palladium-Catalyzed Synthesis of Eltrombopag Intermediates for Commercial Pharmaceutical Production
The pharmaceutical industry continuously seeks robust synthetic routes for complex active pharmaceutical ingredients, and the synthesis of Eltrombopag represents a significant area of focus due to its critical role in treating thrombocytopenia. Patent CN114072386A introduces a transformative methodology for preparing key intermediates, specifically targeting the efficiency and purity challenges associated with the production of 3'-amino-2'-hydroxybiphenyl-3-carboxylic acid (BPCA). This innovation leverages a novel palladium-catalyzed coupling strategy that utilizes alkali metal salts of 2-bromo-4-chlorophenol, fundamentally altering the reaction kinetics and thermodynamic stability of the intermediate species. By shifting from free phenol forms to stable potassium or sodium salts, the process mitigates common side reactions and enhances the overall reproducibility of the synthesis on a large scale. The technical breakthrough lies in the ability to isolate crystalline intermediates with exceptional purity, which serves as a cornerstone for ensuring the quality of the final drug substance. This approach not only addresses the chemical complexities but also aligns with modern manufacturing standards that demand high efficiency and minimal environmental impact. The implications for global supply chains are profound, as this method offers a reliable pathway to secure the availability of this life-saving medication. 
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of BPCA and related Eltrombopag intermediates has been plagued by inefficient multi-step sequences that suffer from low overall yields and significant impurity burdens. Prior art methods, such as those described in earlier patents, often involve lengthy protection and deprotection strategies that add unnecessary complexity and cost to the manufacturing process. For instance, conventional routes may require up to six or more chemical transformations, each introducing potential points of failure and material loss, resulting in an overall molar yield that can be as low as 7%. These traditional pathways frequently rely on harsh reaction conditions and expensive reagents that are difficult to source consistently in large quantities, creating bottlenecks for commercial production. Furthermore, the intermediates generated in these older processes are often oils or amorphous solids that are challenging to purify, leading to variable quality and extended processing times. The accumulation of impurities throughout these long sequences necessitates rigorous and costly purification steps, which further erodes the economic viability of the process. Consequently, manufacturers have struggled to meet the growing global demand for Eltrombopag with these outdated synthetic strategies. 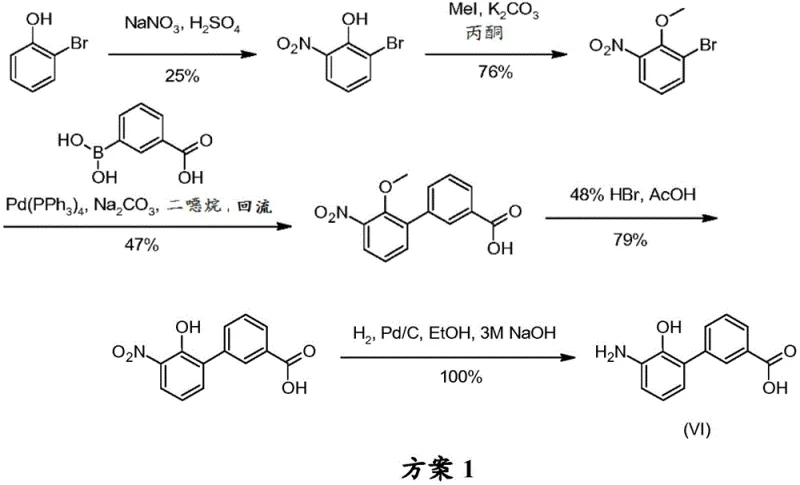
The Novel Approach
In stark contrast, the methodology disclosed in CN114072386A streamlines the synthesis into a more direct and chemically elegant sequence that capitalizes on the reactivity of alkali metal phenolate salts. This novel approach reduces the number of synthetic steps significantly while simultaneously improving the conversion rates of the key coupling reactions. By employing 2-bromo-4-chlorophenol alkali metal salts, the process achieves a molar yield for the initial coupling step ranging from 80% to 95%, which is a dramatic improvement over the suboptimal yields of previous methods. The use of these salts enhances the solubility and stability of the reactants in the reaction medium, facilitating a smoother catalytic cycle and reducing the formation of unwanted by-products. Additionally, the ability to isolate the intermediate biphenyl carboxylic acid salts as crystalline solids allows for straightforward purification through crystallization, ensuring high chemical purity without the need for complex chromatography. This strategic shift not only accelerates the production timeline but also reduces the consumption of solvents and reagents, aligning with green chemistry principles. The robustness of this new route makes it highly suitable for industrial application, providing a sustainable solution for the long-term supply of Eltrombopag intermediates.
Mechanistic Insights into Palladium-Catalyzed Coupling
The core of this technological advancement lies in the mechanistic nuances of the palladium-catalyzed coupling reaction between the phenol salt and the boronic acid derivative. The reaction proceeds through a catalytic cycle where the palladium species facilitates the formation of the carbon-carbon bond between the aryl halide and the boronate component with high selectivity. The presence of the alkali metal cation, particularly potassium, plays a crucial role in stabilizing the transition state and enhancing the nucleophilicity of the phenolate oxygen, which indirectly influences the electronic environment of the aromatic ring. This electronic modulation ensures that the coupling occurs preferentially at the desired position, minimizing the risk of isomeric impurities that are common in biphenyl syntheses. The choice of solvent, such as 1,2-dimethoxyethane mixed with water, creates a biphasic or homogeneous system that optimizes the interaction between the inorganic base and the organic substrates. Furthermore, the use of palladium on carbon as a catalyst offers a heterogeneous system that is easier to remove and recycle compared to homogeneous palladium complexes, reducing metal contamination in the final product. The reaction conditions are mild, typically operating at temperatures around 40°C to 50°C, which prevents thermal degradation of sensitive functional groups. 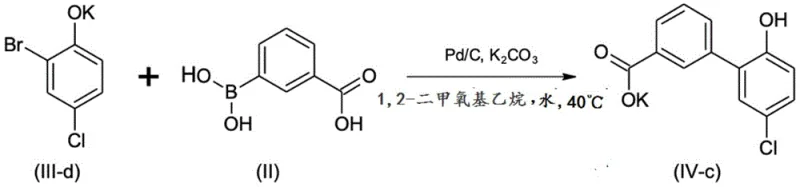
Impurity control is another critical aspect where this mechanism excels, as the crystallization behavior of the intermediate salts acts as a built-in purification step. The specific crystal lattice structure of the potassium salt of the biphenyl intermediate excludes most organic impurities during the solidification process, resulting in a product with HPLC purity exceeding 99%. This high level of purity is maintained through subsequent steps, such as nitration and hydrogenation, because the starting material for each step is of exceptional quality. The nitration step, for example, proceeds with high regioselectivity due to the directing effects of the existing substituents on the biphenyl ring, ensuring that the nitro group is installed precisely at the 3' position. The subsequent hydrogenation reduces the nitro group to an amine without affecting other sensitive functionalities like the carboxylic acid or the phenolic hydroxyl group. This chemoselectivity is vital for maintaining the integrity of the molecule throughout the synthesis. The overall process design demonstrates a deep understanding of physical organic chemistry, leveraging both kinetic and thermodynamic factors to drive the reaction towards the desired product with minimal waste.
How to Synthesize Eltrombopag Intermediate Efficiently
The practical implementation of this synthesis route involves a series of well-defined operational steps that are designed for scalability and safety in a manufacturing environment. The process begins with the preparation of the alkali metal salt of 2-bromo-4-chlorophenol, which can be achieved by reacting the phenol with a base like potassium hydroxide in a suitable solvent system. This salt is then subjected to the palladium-catalyzed coupling with 3-carboxyphenylboronic acid under controlled temperature and atmosphere to yield the biphenyl intermediate. Following isolation and purification, the intermediate undergoes nitration using nitric acid in acetic acid, followed by a careful workup to isolate the nitro compound. The final chemical transformation involves catalytic hydrogenation to reduce the nitro group to the amine, yielding the target BPCA intermediate. Each step is monitored using high-performance liquid chromatography to ensure that quality standards are met before proceeding to the next stage. The detailed standardized synthesis steps are provided in the guide below.
- Perform palladium-catalyzed coupling of 2-bromo-4-chlorophenol alkali metal salt with 3-carboxyphenylboronic acid to form the biphenyl intermediate.
- Execute nitration of the biphenyl intermediate using nitric acid to introduce the nitro group at the 3' position.
- Conduct catalytic hydrogenation of the nitro compound to reduce it to the final amino intermediate BPCA.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process offers substantial benefits that directly address the pain points of procurement and supply chain management in the pharmaceutical sector. The reduction in the number of synthetic steps translates to a shorter manufacturing cycle time, which significantly enhances the responsiveness of the supply chain to market demands. By eliminating the need for multiple isolation and purification stages associated with longer routes, the process reduces the consumption of raw materials and solvents, leading to a lower cost of goods sold. The high yield and purity of the intermediates minimize the risk of batch failures, ensuring a more consistent and reliable supply of material for downstream drug product manufacturing. Furthermore, the use of commercially available starting materials and common catalysts reduces the dependency on specialized or scarce reagents, mitigating supply chain risks. The ability to produce crystalline intermediates also simplifies logistics and storage, as solids are generally more stable and easier to handle than oils or solutions. These factors combined create a more resilient and cost-effective supply chain for Eltrombopag production.
- Cost Reduction in Manufacturing: The streamlined synthetic route eliminates several expensive and time-consuming steps found in conventional methods, resulting in significant operational cost savings. By achieving higher yields in the key coupling step, the amount of starting material required per kilogram of final product is drastically reduced, optimizing raw material utilization. The use of heterogeneous palladium catalysts allows for easier recovery and reuse, further lowering the cost associated with precious metal consumption. Additionally, the reduced need for extensive purification processes decreases the consumption of chromatography media and solvents, which are major cost drivers in pharmaceutical manufacturing. These efficiencies collectively contribute to a more competitive pricing structure for the intermediate without compromising on quality.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials such as 2-bromo-4-chlorophenol and 3-carboxyphenylboronic acid ensures that the supply chain is not vulnerable to shortages of exotic reagents. The robustness of the reaction conditions means that the process can be transferred between different manufacturing sites with minimal re-optimization, providing flexibility in sourcing. The high purity of the intermediates reduces the likelihood of quality-related delays or rejections, ensuring a smooth flow of materials through the production pipeline. This reliability is crucial for maintaining continuous production schedules and meeting regulatory commitments for drug availability. The process stability also allows for better inventory management and forecasting, reducing the need for excessive safety stock.
- Scalability and Environmental Compliance: The process is designed with scale-up in mind, utilizing solvents and conditions that are safe and manageable at large volumes. The crystallization of intermediates facilitates efficient solid-liquid separation, which is a key unit operation in large-scale manufacturing. The reduction in waste generation due to higher yields and fewer steps aligns with environmental regulations and sustainability goals. The use of water in conjunction with organic solvents reduces the overall organic solvent load, simplifying waste treatment and disposal. This environmental compatibility not only reduces compliance costs but also enhances the corporate social responsibility profile of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this improved synthesis process for Eltrombopag intermediates. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation to provide clarity on feasibility and performance. Understanding these details is essential for stakeholders evaluating the adoption of this technology for their supply chains. The information covers aspects of yield, purity, and operational parameters that are critical for decision-making.
Q: How does the new process improve yield compared to prior art?
A: The new process utilizes alkali metal salts of the starting phenol, which significantly enhances reaction conversion and stability, leading to overall yields exceeding 65% compared to the 7% typical of older multi-step routes.
Q: What are the purity specifications for the intermediate?
A: The crystalline intermediates produced via this method consistently achieve HPLC purity levels greater than 99%, minimizing the need for extensive downstream purification.
Q: Is the process scalable for commercial manufacturing?
A: Yes, the use of stable crystalline salts and common solvents like 1,2-dimethoxyethane facilitates safe and efficient scale-up from kilogram to multi-ton production scales.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Eltrombopag Intermediate Supplier
NINGBO INNO PHARMCHEM stands at the forefront of chemical manufacturing, possessing the technical expertise and infrastructure to leverage advanced synthetic routes like the one described in CN114072386A for commercial production. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Eltrombopag intermediate meets the highest global standards for pharmaceutical use. Our commitment to quality and consistency makes us a trusted partner for multinational pharmaceutical companies seeking to secure their supply chains for critical medications. We understand the complexities of regulatory compliance and are dedicated to supporting our clients through every stage of the product lifecycle.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific production needs and cost structures. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the potential economic advantages of switching to this more efficient manufacturing process. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your project requirements. Our goal is to collaborate with you to drive innovation and efficiency in the production of life-saving therapies. Let us help you optimize your supply chain with our proven expertise in fine chemical synthesis.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
