Advanced Catalytic Synthesis of 3-Hydroxytetrahydrofuran for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways for producing critical intermediates like 3-hydroxytetrahydrofuran, a key building block in the synthesis of antiretroviral and anticancer agents. Patent CN113264908B introduces a transformative preparation method that leverages heterogeneous catalytic reaction steps to convert 3,4-epoxytetrahydrofuran compounds into high-value 3-hydroxytetrahydrofuran derivatives. This technical breakthrough addresses long-standing challenges in traditional synthesis, such as complex purification and high catalyst costs, by implementing a green, operationally simple process that is highly conducive to large-scale industrial production. The methodology utilizes a sequential approach involving acid-catalyzed ring-opening followed by catalytic hydrogenolysis, ensuring high selectivity and yield while maintaining rigorous safety standards. For R&D directors and procurement specialists, this patent represents a significant opportunity to optimize supply chains and reduce the overall cost of goods sold for complex pharmaceutical intermediates without compromising on purity or regulatory compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-hydroxytetrahydrofuran has relied on starting materials such as malic acid or tartaric acid, which require multi-step sequences involving esterification, reduction, and dehydration cyclization. These traditional routes often suffer from low atom economy and generate substantial chemical waste, leading to increased environmental compliance costs and complex downstream processing. Alternative methods utilizing butenol or 2-oxiranyl ethanol have been reported, but they face significant hurdles including the high cost and instability of raw materials, as well as the formation of difficult-to-separate by-products like 2-hydroxymethyloxetane. Furthermore, processes employing homogeneous chiral boron catalysts, while effective for asymmetric synthesis, involve expensive reagents and intricate removal steps that are not ideal for cost-sensitive commercial manufacturing. The reliance on harsh reaction conditions and precious metal catalysts in prior art has consistently limited the scalability and economic viability of producing this critical intermediate for the global pharmaceutical market.
The Novel Approach
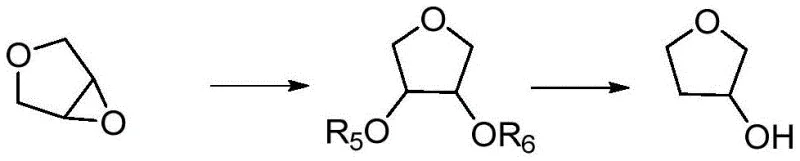
The novel approach disclosed in the patent data fundamentally shifts the paradigm by utilizing 3,4-epoxytetrahydrofuran as a more economical and accessible starting原料,derived from the epoxidation of dihydrofuran. This method employs a heterogeneous catalytic system that simplifies the reaction workflow into two distinct, controllable steps: ring-opening addition and subsequent hydrogenolysis. By using solid acid catalysts and supported metal catalysts, the process eliminates the need for complex liquid-liquid extractions associated with homogeneous catalysis, thereby streamlining the separation process. The reaction conditions are optimized to operate within a broad temperature range of -20°C to 200°C and hydrogen pressures up to 12MPa, allowing for flexibility in reactor design and operation. This strategic shift not only reduces the consumption of expensive reagents but also enhances the overall safety profile of the manufacturing process, making it a superior choice for companies aiming to secure a reliable and cost-effective supply of high-purity pharmaceutical intermediates.
Mechanistic Insights into Heterogeneous Catalytic Hydrogenolysis
The core of this innovative synthesis lies in the precise control of the catalytic cycle, beginning with the acid-catalyzed ring-opening of the epoxy group. In the first step, the 3,4-epoxytetrahydrofuran reacts in the presence of a solid acid catalyst, such as an acidic ion exchange resin or sulfonated metal oxide, to form an intermediate alcoholyzate. This step is critical for setting the stereochemistry and ensuring the correct positioning of the hydroxyl groups for the subsequent reduction. The use of a fixed-bed reactor for this stage ensures continuous contact between the reactants and the catalyst surface, maximizing conversion efficiency while minimizing catalyst degradation. The intermediate formed is then subjected to hydrogenolysis using a specialized supported catalyst, where the choice of metal active components like Ruthenium, Cobalt, or Nickel plays a pivotal role in cleaving the carbon-oxygen bond selectively. This dual-catalyst system ensures that the reaction proceeds with high specificity, avoiding over-reduction or ring-opening side reactions that could compromise the integrity of the tetrahydrofuran ring structure.
Impurity control is inherently built into the mechanistic design of this process through the selection of specific catalyst supports and active components. For instance, the use of oxide carriers like ZnO, MgO, or CeO2 doped with promoters such as Sn or Mo enhances the dispersion of the active metal sites, which in turn improves the selectivity towards the desired 3-hydroxy product. The patent data highlights that by adjusting the valence states of metal oxides within the catalyst, the electronic environment of the active sites can be tuned to favor the formation of the target molecule over potential by-products. This level of control is essential for meeting the stringent purity specifications required by pharmaceutical regulators, as it minimizes the presence of genotoxic impurities or structurally similar analogs that are difficult to remove in later stages. The ability to achieve high yields, such as the reported 82.0% in optimized examples, demonstrates the robustness of this mechanistic approach in maintaining product quality under continuous production conditions.
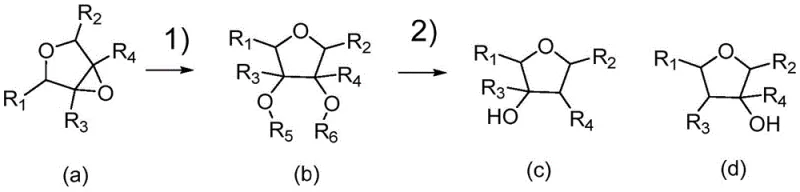
How to Synthesize 3-Hydroxytetrahydrofuran Efficiently
Implementing this synthesis route requires a clear understanding of the operational parameters defined in the patent to ensure optimal performance and safety. The process begins with the preparation of the reaction mixture, where 3,4-epoxytetrahydrofuran is combined with a solvent such as methanol or ethanol and passed through a fixed-bed reactor containing the solid acid catalyst. The effluent from this first stage, containing the ring-opened intermediate, is then directly fed into a hydrogenation reactor without the need for intermediate isolation, which significantly reduces processing time and solvent consumption. In the second stage, the mixture reacts under hydrogen pressure in the presence of the supported hydrogenolysis catalyst, where temperature and pressure are carefully controlled to drive the reaction to completion. This continuous flow setup not only enhances throughput but also allows for real-time monitoring of reaction progress, enabling immediate adjustments to maintain consistent product quality.
- Perform ring-opening addition of 3,4-epoxytetrahydrofuran using a solid acid catalyst in a fixed-bed reactor at controlled temperatures.
- Transfer the intermediate alcoholyzate directly to a hydrogenation reactor containing a supported hydrogenolysis catalyst.
- React under hydrogen pressure at elevated temperatures to yield high-purity 3-hydroxytetrahydrofuran with simplified separation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented technology offers substantial strategic benefits that extend beyond simple technical feasibility. The shift to a heterogeneous catalytic process significantly reduces the dependency on expensive homogeneous catalysts and complex purification solvents, leading to a drastic simplification of the manufacturing cost structure. By eliminating the need for costly metal scavenging steps and reducing solvent waste, the overall production cost is lowered, allowing for more competitive pricing in the global market for pharmaceutical intermediates. Furthermore, the use of robust solid catalysts enhances the reliability of the supply chain by reducing the risk of batch-to-batch variability and catalyst supply disruptions, ensuring a steady flow of materials for downstream drug manufacturing. This process stability is crucial for maintaining long-term contracts with pharmaceutical clients who require guaranteed availability and consistent quality for their critical drug substances.
- Cost Reduction in Manufacturing: The implementation of low-cost solid acid and supported metal catalysts removes the financial burden associated with precious metal homogeneous catalysts and their subsequent removal processes. This transition leads to substantial cost savings by simplifying the downstream processing workflow, as the heterogeneous nature of the catalysts allows for easy filtration and reuse, thereby minimizing raw material consumption. Additionally, the ability to operate in a continuous fixed-bed mode reduces energy consumption and labor costs compared to traditional batch processes, further enhancing the economic efficiency of the production line. These cumulative efficiencies translate into a more favorable cost of goods sold, providing a competitive edge in the pricing of high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The reliance on readily available raw materials like 3,4-epoxytetrahydrofuran, which can be sourced from stable dihydrofuran feedstocks, mitigates the risk of supply chain bottlenecks associated with specialized starting materials like butenol. The robustness of the solid catalysts ensures longer operational lifecycles and reduced frequency of catalyst replacement, which stabilizes production schedules and minimizes unplanned downtime. This reliability is essential for meeting the just-in-time delivery requirements of large pharmaceutical companies, ensuring that the supply of critical intermediates remains uninterrupted even during periods of high market demand. Consequently, partners can rely on a more predictable and resilient supply chain that supports their own production planning and inventory management strategies.
- Scalability and Environmental Compliance: The design of this process for continuous flow operation in fixed-bed reactors inherently supports seamless scale-up from pilot plant to commercial production volumes without the need for significant process re-engineering. The green chemistry principles embedded in the method, such as reduced solvent usage and the elimination of toxic homogeneous catalysts, align with increasingly stringent environmental regulations and corporate sustainability goals. This compliance reduces the regulatory burden and potential fines associated with waste disposal, while also enhancing the corporate image of manufacturers as responsible stewards of the environment. The combination of scalability and environmental safety makes this technology a future-proof solution for the long-term production of essential chemical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, based on the detailed specifications provided in the patent documentation. These answers are designed to clarify the operational benefits and feasibility of adopting this method for large-scale manufacturing of pharmaceutical intermediates. Understanding these details is crucial for technical teams evaluating the integration of this process into existing production facilities or for procurement teams assessing the long-term viability of the supply source.
Q: What are the primary advantages of this heterogeneous catalytic route over traditional methods?
A: This method eliminates the need for expensive chiral boron catalysts or complex multi-step reductions from malic acid, utilizing low-cost solid acid and supported metal catalysts that facilitate easier separation and recycling.
Q: How does this process address impurity control in pharmaceutical intermediates?
A: By employing specific supported catalysts like Ru-SnOx-CeO2, the reaction selectively promotes the formation of the target 3-hydroxy structure while minimizing by-products like 2-hydroxymethyloxetane often seen in alternative cyclization routes.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the patent explicitly describes the use of fixed-bed reactors and continuous flow processes, which are inherently designed for scalability, safety, and consistent quality in commercial manufacturing environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Hydroxytetrahydrofuran Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for complex pharmaceutical intermediates like 3-hydroxytetrahydrofuran. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the innovative catalytic technologies described in patent CN113264908B can be effectively translated into industrial reality. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch meets the high standards required by global regulatory bodies. We are committed to leveraging our technical expertise to optimize these processes further, ensuring maximum yield and minimal environmental impact for our partners.
We invite pharmaceutical and chemical companies to collaborate with us to explore the full potential of this advanced synthesis method for their supply chain needs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage you to reach out for specific COA data and route feasibility assessments to determine how this technology can enhance your production efficiency and reduce overall manufacturing costs. Let us help you secure a reliable and cost-effective supply of high-quality intermediates for your next generation of therapeutic products.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
