Advanced Iodine-Catalyzed Triazole Synthesis: Scalable Production of High-Purity Pharmaceutical Intermediates
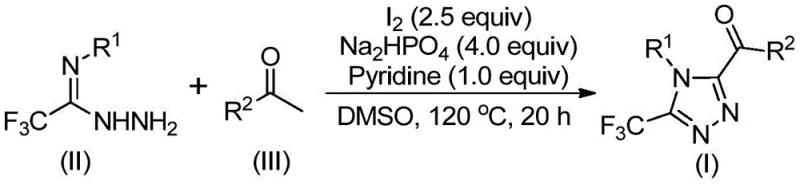
Patent CN113105402B introduces a groundbreaking iodine-catalyzed methodology for synthesizing 3,4,5-trisubstituted 1,2,4-triazole compounds that addresses critical limitations in traditional synthetic approaches while meeting stringent pharmaceutical manufacturing requirements. This innovative process eliminates the requirement for heavy metal catalysts while maintaining high reaction efficiency and product purity profiles essential for active pharmaceutical ingredients. The methodology represents a significant advancement in heterocyclic chemistry with direct applications in drug development where compounds like Maraviroc and Sitagliptin demonstrate the therapeutic importance of triazole structures. Unlike conventional methods that necessitate anhydrous and oxygen-free conditions requiring specialized equipment investments, this novel approach operates under standard laboratory settings without infrastructure modifications. The patent demonstrates exceptional substrate versatility across various aryl groups at R¹ and R² positions while consistently delivering high-purity products suitable for pharmaceutical intermediates with documented yields ranging from 37% to 86% across diverse substitution patterns.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for preparing multi-substituted triazole compounds typically rely on transition metal-catalyzed cyclization reactions that introduce significant challenges for pharmaceutical manufacturing operations worldwide. These methods require strictly controlled anhydrous and oxygen-free environments that necessitate substantial capital investment in specialized equipment while increasing operational complexity and maintenance costs substantially across production facilities. The use of palladium or copper-based catalysts creates critical impurity concerns requiring additional purification stages to meet regulatory thresholds for metal residues in final drug products. Conventional approaches often exhibit limited substrate scope with poor functional group tolerance that restricts structural diversity of accessible compounds while increasing development timelines for new molecular entities. Most critically for supply chain managers, the presence of residual metal catalysts necessitates complex validation protocols when scaling up production processes from laboratory to commercial scale.
The Novel Approach
The iodine-catalyzed methodology described in patent CN113105402B overcomes these limitations through an elegant single-pot reaction sequence that operates under standard laboratory conditions without requiring specialized infrastructure investments or complex operational procedures. This innovative process utilizes molecular iodine as an efficient catalyst that avoids toxic heavy metals while maintaining high reaction yields across diverse substrate combinations including various substituted phenyl groups at both R¹ and R² positions. The reaction proceeds through a well-defined mechanism involving iodination/Kornblum oxidation followed by condensation and cyclization steps conducted in dimethyl sulfoxide as both solvent and reagent system component. Crucially for procurement teams, this method eliminates the need for anhydrous and oxygen-free environments while demonstrating exceptional functional group tolerance across halogenated and alkyl-substituted aryl groups without requiring process modifications.
Mechanistic Insights into Iodine-Catalyzed Triazole Formation
The reaction mechanism begins with the iodination/Kornblum oxidation of aryl ethyl ketone under thermal conditions (90–110°C) in dimethyl sulfoxide solvent over a period of 4–6 hours to form the corresponding aryl diketone intermediate through a well-documented oxidative transformation pathway. This key activation step enables subsequent cyclization chemistry by generating the necessary electrophilic center required for hydrazone formation with trifluoroethylimide hydrazide substrates under controlled thermal conditions (110–130°C). The aryl diketone intermediate then undergoes condensation with trifluoroethylimide hydrazide to form a hydrazone intermediate through dehydration chemistry that occurs spontaneously under the reaction conditions without requiring additional catalysts or reagents.
Impurity control is achieved through multiple mechanistic advantages inherent in this iodine-catalyzed system compared to conventional approaches using transition metals. The elimination of palladium or copper catalysts completely removes potential sources of metal impurities that typically require extensive purification processes involving chelating agents or specialized chromatography techniques to meet regulatory standards below parts-per-million levels. The reaction's tolerance for various functional groups including halogens (Cl), alkyl groups (Me), alkoxy groups (OMe), and trifluoromethyl groups across different substitution patterns minimizes side reactions that could generate complex impurity profiles requiring additional analytical monitoring during quality control procedures. The well-defined sequential reaction pathway prevents formation of multiple byproducts commonly observed in less controlled synthetic methods using heterogeneous catalytic systems.
How to Synthesize High-Purity Triazole Compounds Efficiently
This patent describes a streamlined synthetic route that significantly improves upon conventional methods for producing high-purity triazole compounds essential for pharmaceutical applications by eliminating complex setup requirements while maintaining consistent product quality standards required by global regulatory authorities. The methodology offers exceptional operational simplicity through its single-pot design while providing superior flexibility across different manufacturing environments without requiring specialized infrastructure investments or extensive operator training programs.
- Combine aryl ethyl ketone with iodine in DMSO solvent at 90-110°C for 4-6 hours to form aryl diketone intermediate
- Add trifluoroethylimide hydrazide with sodium dihydrogen phosphate (4 equiv), pyridine (1 equiv), and iodine (2.5 equiv) maintaining precise molar ratios
- Heat reaction mixture to 110-130°C for 12-20 hours followed by standard column chromatography purification to obtain final product
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology addresses critical pain points in pharmaceutical supply chains by offering a more sustainable route to essential triazole intermediates while reducing total cost of ownership through simplified process design and enhanced operational flexibility across global manufacturing networks.
- Cost Reduction in Manufacturing: Elimination of expensive transition metal catalysts creates substantial cost savings throughout the manufacturing cycle by removing both raw material expenses associated with precious metals and additional processing costs required for metal removal procedures that typically involve multiple purification stages.
- Enhanced Supply Chain Reliability: Operation without anhydrous or oxygen-free conditions significantly reduces dependency on specialized infrastructure while improving production flexibility across different manufacturing environments with varying humidity levels or atmospheric conditions without requiring facility modifications.
- Scalability and Environmental Compliance: Demonstrated scalability from laboratory to commercial production levels addresses growing environmental regulations by eliminating heavy metal waste streams while reducing overall environmental impact through simplified processing requirements.
Frequently Asked Questions (FAQ)
The following questions address common concerns from procurement professionals regarding implementation of this innovative triazole synthesis methodology based on detailed technical analysis of patent CN113105402B experimental validation data.
Q: How does this method eliminate heavy metal contamination concerns in pharmaceutical production?
A: The iodine-catalyzed process completely avoids transition metal catalysts while maintaining high product purity through a well-defined reaction mechanism that eliminates metal residue risks requiring additional purification steps.
Q: What scalability advantages does this synthesis offer compared to conventional methods?
A: This methodology operates without anhydrous/oxygen-free requirements and demonstrates consistent performance from gram-scale laboratory batches to commercial production volumes with simplified process validation.
Q: How does the substrate versatility impact supply chain flexibility for pharmaceutical manufacturers?
A: The broad functional group tolerance across R¹ and R² positions enables multiple raw material sourcing options while maintaining consistent product quality without requiring process revalidation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triazole Compound Supplier
Our company possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through rigorous QC labs dedicated to pharmaceutical intermediates manufacturing using validated analytical methodologies including NMR spectroscopy and HRMS verification protocols.
Request a Customized Cost-Saving Analysis from our technical procurement team to understand how this methodology can be implemented for your specific triazole compound requirements; we will provide detailed COA data and route feasibility assessments to support your decision-making process.
