Scalable Fmoc-Based Solid Phase Synthesis of Somatostatin for Commercial Production
Scalable Fmoc-Based Solid Phase Synthesis of Somatostatin for Commercial Production
The pharmaceutical industry continuously seeks robust methodologies for producing complex peptide hormones like somatostatin, a critical regulatory molecule. Patent CN100338090C discloses a refined solid-phase synthesis strategy that addresses historical bottlenecks in yield, purity, and operational safety. This technology leverages 2-chlorotrityl resin as a solid support and employs an Fmoc-protection strategy to construct the 14-peptide linear chain. Unlike earlier liquid-phase methods that suffered from tedious purification steps and low overall yields, this approach streamlines the workflow by integrating side-chain deprotection with resin cleavage. The process culminates in a selective oxidative cyclization to form the essential disulfide bridge, ensuring the bioactive conformation is achieved with high fidelity. For procurement and R&D teams, this represents a shift towards more manageable and scalable manufacturing protocols.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of somatostatin relied heavily on liquid-phase peptide synthesis or early solid-phase methods that utilized hazardous reagents. Liquid-phase routes, such as those described in older US patents, often required multiple intermediate purification steps, making solvent removal difficult and driving up operational costs significantly. Furthermore, traditional solid-phase methods frequently depended on hydrogen fluoride (HF) for the removal of side-chain protecting groups and final cleavage from the resin. The use of HF presents severe safety challenges, requiring specialized corrosion-resistant equipment and rigorous safety protocols that are difficult to maintain in standard manufacturing facilities. Additionally, these older methods were prone to racemization during the coupling of longer peptide chains, compromising the optical purity and biological activity of the final product.
The Novel Approach
The methodology outlined in the patent introduces a superior alternative by utilizing 2-chlorotrityl resin combined with Fmoc-protected amino acids. This combination allows for mild acidic cleavage conditions, effectively bypassing the need for dangerous hydrogen fluoride. The process employs a programmed reaction sequence where amino acids are coupled using DCC and HOBt activation, ensuring high coupling efficiency and minimizing deletion sequences. A key innovation is the use of a specific K-reagent mixture containing trifluoroacetic acid and scavengers like thioanisole and ethanedithiol to simultaneously cleave the peptide and remove side-chain protections. This streamlined approach not only enhances operator safety but also simplifies the downstream processing, making it highly suitable for the commercial scale-up of complex peptides required for therapeutic applications.
Mechanistic Insights into Fmoc Solid Phase Peptide Synthesis
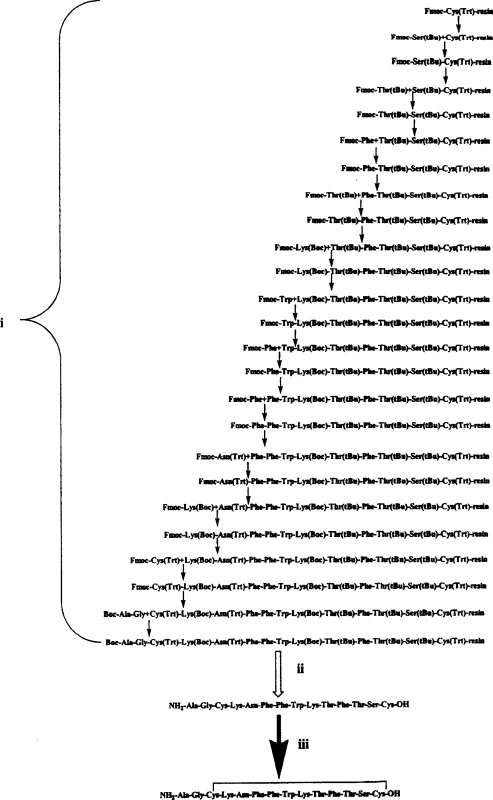
The core of this synthesis lies in the iterative cycle of deprotection and coupling on the solid support. The Fmoc group is removed using a piperidine solution, exposing the free amine for the next coupling reaction without affecting the acid-labile link to the resin. Activation of the incoming Fmoc-amino acid is achieved using dicyclohexylcarbodiimide (DCC) and hydroxybenzotriazole (HOBt), which forms an active ester intermediate that reacts rapidly with the resin-bound amine. This mechanism significantly suppresses racemization, a common issue in peptide chemistry, thereby maintaining the stereochemical integrity of the chiral centers throughout the chain elongation. The use of 2-chlorotrityl resin is particularly strategic, as its linkage is stable to the basic conditions of Fmoc removal but labile to mild acids, allowing for the preservation of acid-sensitive side chains until the final cleavage step.
Following the assembly of the linear peptide, the formation of the cyclic structure is critical for biological activity. The patent describes an oxidative cyclization mechanism where the two cysteine residues at positions 3 and 14 form an intramolecular disulfide bond. This is achieved by dissolving the linear peptide in a dilute aqueous solution containing either hydrogen peroxide or dimethyl sulfoxide. The oxidant facilitates the conversion of the free thiol groups into a disulfide bridge. This solution-phase oxidation is advantageous because it allows for precise control over the reaction environment, favoring the formation of the monomeric cyclic product over intermolecular polymers. The subsequent purification via medium-pressure C18 chromatography ensures that any linear precursors or over-oxidized byproducts are removed, delivering a product with stringent purity specifications.
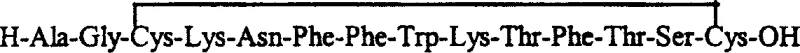
How to Synthesize Somatostatin Efficiently
The synthesis protocol detailed in the patent provides a clear roadmap for producing high-quality somatostatin. It begins with the loading of the first protected cysteine onto the resin, followed by sequential addition of the remaining amino acids in the correct order. Each coupling step is monitored using the Kaiser test to ensure completeness before proceeding. Once the full linear sequence is assembled, the peptide is cleaved and deprotected in a single step using the K-reagent cocktail. The resulting crude linear peptide is then subjected to oxidative cyclization in a controlled aqueous environment. Finally, the cyclic product is purified using reverse-phase chromatography and converted to the acetate salt form. For detailed standard operating procedures and specific reaction parameters, please refer to the guide below.
- Load Fmoc-Cys(Trt) onto 2-chlorotrityl resin and perform iterative deprotection using piperidine and coupling using DCC/HOBt.
- Cleave the linear peptide from the resin using K-reagent (TFA/scavengers) to remove side-chain protecting groups simultaneously.
- Perform oxidative cyclization in dilute aqueous hydrogen peroxide or DMSO to form the intramolecular disulfide bond, followed by C18 purification.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this synthesis route offers substantial strategic benefits for supply chain stability and cost management. By eliminating the requirement for hydrogen fluoride, manufacturers can avoid the high capital expenditure associated with specialized HF-resistant reactors and scrubbing systems. This reduction in infrastructure complexity directly translates to lower overhead costs and reduced regulatory burdens related to hazardous material handling. Furthermore, the use of widely available Fmoc-amino acids and standard solvents like dichloromethane and DMF ensures a robust supply chain for raw materials, mitigating the risk of production delays caused by specialty reagent shortages. The simplified purification workflow also reduces solvent consumption and waste generation, aligning with modern environmental compliance standards.
- Cost Reduction in Manufacturing: The transition from liquid-phase or HF-based solid-phase methods to this Fmoc/2-chlorotrityl protocol removes the need for expensive and dangerous reagents. The elimination of HF not only lowers safety compliance costs but also reduces equipment maintenance expenses due to corrosion. Additionally, the high coupling efficiency minimizes the loss of valuable amino acid building blocks, improving the overall material throughput. The ability to perform cleavage and deprotection in a single step further reduces labor hours and processing time, contributing to significant operational savings without compromising product quality.
- Enhanced Supply Chain Reliability: The reliance on standard Fmoc-protected amino acids ensures that raw material sourcing is not dependent on niche suppliers with long lead times. These building blocks are commodity chemicals in the peptide industry, available from multiple qualified vendors globally. This diversification of the supply base reduces the risk of single-source bottlenecks. Moreover, the robustness of the 2-chlorotrityl resin linkage allows for flexible batch sizes, enabling manufacturers to respond quickly to fluctuating market demand for somatostatin and its analogues without extensive process re-validation.
- Scalability and Environmental Compliance: This method is inherently designed for scale-up, moving away from the limitations of liquid-phase synthesis where solubility and purification become problematic at larger volumes. The solid-phase approach confines the growing peptide to the resin, simplifying washing and isolation steps. The avoidance of heavy metals and highly toxic gases like HF simplifies waste treatment protocols. The use of aqueous oxidation for cyclization is also greener compared to heavy metal-mediated methods, facilitating easier disposal of process effluents and ensuring compliance with increasingly strict environmental regulations in pharmaceutical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of somatostatin using this patented methodology. These insights are derived directly from the experimental data and process descriptions found in the patent documentation. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing capabilities. The answers highlight the specific advantages of the Fmoc strategy and the oxidative cyclization step.
Q: Why is the 2-chlorotrityl resin preferred for somatostatin synthesis?
A: 2-chlorotrityl resin allows for mild acid cleavage conditions, preventing the racemization often seen with stronger acid treatments and facilitating the preservation of sensitive amino acid sequences during the synthesis of complex peptides.
Q: How does this method improve safety compared to traditional liquid phase synthesis?
A: This solid-phase method eliminates the need for hazardous hydrogen fluoride (HF) for deprotection, replacing it with a trifluoroacetic acid-based K-reagent system, which significantly reduces operational risks and equipment corrosion issues.
Q: What is the advantage of the oxidative cyclization step described?
A: The method utilizes dilute hydrogen peroxide or DMSO in an aqueous environment for selective intramolecular disulfide bond formation, offering a controlled and efficient pathway to the cyclic structure without requiring complex enzymatic steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Somatostatin Supplier
The technological potential of this Fmoc-based solid-phase synthesis route is immense, offering a pathway to high-purity somatostatin that meets rigorous pharmaceutical standards. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex peptide syntheses are translated efficiently from the lab to the plant. Our facility is equipped with stringent purity specifications and rigorous QC labs capable of verifying the structural integrity and biological activity of every batch. We understand the critical nature of peptide intermediates in the broader drug development landscape and are committed to delivering consistency and reliability.
We invite you to engage with our technical procurement team to discuss how we can optimize your supply chain for somatostatin and related peptides. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our manufacturing efficiencies can benefit your bottom line. We encourage potential partners to contact us for specific COA data and route feasibility assessments tailored to your project requirements. Let us collaborate to secure a stable and cost-effective supply of this vital pharmaceutical ingredient.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
