Advanced Synthetic Route for High-Purity Benzimidazole Pharmaceutical Intermediates
The pharmaceutical industry continuously seeks robust synthetic pathways for complex heterocyclic compounds, particularly those serving as critical intermediates for antithrombotic agents. Patent CN1972919B discloses a groundbreaking method for producing optionally substituted 4-(benzimidazol-2-ylmethylamino)-benzamidines, a class of compounds known for their potent thrombin inhibitory activity. This technology represents a significant leap forward in process chemistry, addressing the longstanding inefficiencies associated with traditional benzimidazole synthesis. By shifting away from harsh nitrile hydrolysis towards a milder oxadiazolone-based condensation strategy, the patent offers a cleaner, more controllable route to high-value active pharmaceutical ingredients (APIs). 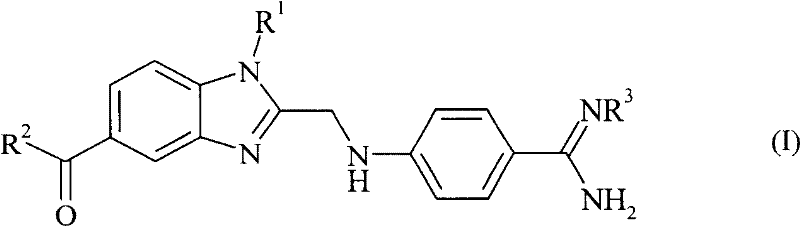 For procurement and supply chain leaders, understanding the nuances of this synthesis is vital, as it directly impacts the reliability and cost structure of the final drug substance. The method outlined in CN1972919B not only improves yield profiles but also drastically reduces the environmental footprint by minimizing acidic waste streams, aligning perfectly with modern green chemistry mandates in fine chemical manufacturing.
For procurement and supply chain leaders, understanding the nuances of this synthesis is vital, as it directly impacts the reliability and cost structure of the final drug substance. The method outlined in CN1972919B not only improves yield profiles but also drastically reduces the environmental footprint by minimizing acidic waste streams, aligning perfectly with modern green chemistry mandates in fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods, specifically those detailed in international patent application WO 98/37075, typically rely on the conversion of substituted (4-benzimidazol-2-ylmethylamino)-benzonitriles into the corresponding benzamidines via reaction with ammonia. While chemically feasible, this legacy approach presents severe operational drawbacks for large-scale manufacturing. The primary bottleneck lies in the subsequent processing steps, which require extensive and worryingly high burdens of acid treatment to isolate the final product. This reliance on aggressive acidic conditions not only complicates the workup procedure but also poses significant challenges for equipment corrosion and waste disposal compliance. Furthermore, the handling of nitrile intermediates often introduces safety hazards and purity issues related to incomplete hydrolysis, leading to difficult-to-remove impurities that can compromise the quality of the final API. These factors collectively drive up production costs and extend lead times, making the conventional route less attractive for commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
In stark contrast, the method disclosed in CN1972919B introduces a streamlined three-step sequence that circumvents these historical pain points. The core innovation involves the condensation of an optionally substituted diaminobenzene with 2-[4-(1,2,4-oxadiazol-5-one-3-yl)-phenylamino]-acetic acid. This strategic choice of starting materials allows for the formation of the benzimidazole ring under much milder conditions.  Following the condensation, the resulting intermediate undergoes a catalytic hydrogenation step, which cleanly converts the oxadiazolone moiety into the desired amidine functionality without the need for harsh hydrolytic agents. This shift from hydrolysis to hydrogenation is a game-changer for process safety and efficiency. Finally, the method allows for optional carbonylation of the amidino group using alkyl haloformates, providing flexibility in generating various prodrug forms or protected intermediates. This novel approach effectively decouples the synthesis from the limitations of acid-heavy workflows, offering a more direct and economically viable path to the target molecules.
Following the condensation, the resulting intermediate undergoes a catalytic hydrogenation step, which cleanly converts the oxadiazolone moiety into the desired amidine functionality without the need for harsh hydrolytic agents. This shift from hydrolysis to hydrogenation is a game-changer for process safety and efficiency. Finally, the method allows for optional carbonylation of the amidino group using alkyl haloformates, providing flexibility in generating various prodrug forms or protected intermediates. This novel approach effectively decouples the synthesis from the limitations of acid-heavy workflows, offering a more direct and economically viable path to the target molecules.
Mechanistic Insights into Oxadiazolone-Mediated Condensation
The success of this synthetic route hinges on the precise mechanistic execution of the condensation step (Step A). The reaction between the diaminobenzene and the oxadiazole-acetic acid derivative is facilitated by powerful coupling agents such as 1,1'-carbonyldiimidazole (CDI) or propylphosphonic anhydride (PPA). When CDI is employed, it activates the carboxylic acid group of the oxadiazole precursor to form a reactive acyl-imidazole intermediate, which is then attacked by the amine group of the diaminobenzene. This activation strategy ensures high conversion rates even at moderate temperatures, typically around 50°C in solvents like tetrahydrofuran (THF). Alternatively, the use of PPA in the presence of organic bases like DIPEA offers a water-scavenging effect that drives the equilibrium towards product formation. The choice of solvent is critical; aprotic polar solvents such as THF, N-methylpyrrolidone (NMP), or dimethylformamide (DMF) are preferred to maintain solubility of the polar intermediates while preventing side reactions.  The subsequent hydrogenation step (Step B) utilizes transition metal catalysts, with 10% palladium on carbon (Pd/C) being particularly effective. Operating at low hydrogen pressures (1-2 bar) and ambient temperatures, this step selectively reduces the oxadiazolone ring to the amidine while preserving other sensitive functional groups, such as esters or pyridyl moieties, which might be present on the R-groups. This chemoselectivity is paramount for maintaining the integrity of complex molecular architectures often found in modern drug candidates.
The subsequent hydrogenation step (Step B) utilizes transition metal catalysts, with 10% palladium on carbon (Pd/C) being particularly effective. Operating at low hydrogen pressures (1-2 bar) and ambient temperatures, this step selectively reduces the oxadiazolone ring to the amidine while preserving other sensitive functional groups, such as esters or pyridyl moieties, which might be present on the R-groups. This chemoselectivity is paramount for maintaining the integrity of complex molecular architectures often found in modern drug candidates.
From an impurity control perspective, this mechanism offers distinct advantages over nitrile hydrolysis. Traditional acid hydrolysis often generates ammonium salts and partially hydrolyzed amide byproducts that are structurally similar to the target, making chromatographic separation difficult and costly. In the hydrogenation pathway, the primary byproducts are typically reduced forms of the catalyst support or minor over-reduction products that are easily removed via crystallization or simple filtration. The use of Pd/C allows for the physical removal of the catalyst post-reaction, significantly reducing the risk of heavy metal contamination in the final product. Furthermore, the optional carbonylation step (Step C) using reagents like hexyl chloroformate proceeds cleanly in the presence of mild bases like potassium carbonate, ensuring that the final protected amidine is obtained with high purity (>95% HPLC area) without the need for extensive downstream purification. This robust impurity profile is essential for meeting the stringent specifications required by regulatory bodies for pharmaceutical intermediates.
How to Synthesize 4-(Benzimidazolylmethylamino)-benzamidine Efficiently
Implementing this synthesis requires careful attention to reaction parameters to maximize yield and safety. The process begins with the preparation of the key oxadiazole-acetic acid precursor, which can be synthesized from 4-aminobenzonitrile via amidoxime formation and cyclization with dialkyl carbonates. Once the precursor is secured, the condensation with the appropriate diaminobenzene is performed in THF using CDI or PPA as the coupling mediator. The reaction mixture is typically heated to facilitate ring closure, followed by solvent exchange or direct addition of the hydrogenation solvent. The hydrogenation is conducted in ethanol or ethanol/water mixtures using wet Pd/C catalyst at roughly 2 bar pressure. Post-hydrogenation, the product can be isolated as a salt, such as the tosylate, by adding p-toluenesulfonic acid to the filtrate. For the final derivatization, the free base is reacted with hexyl chloroformate in acetone/water to yield the carbonylated product. Detailed standardized operating procedures for each of these stages are critical for reproducibility.
- Condense substituted diaminobenzene with 2-[4-(1,2,4-oxadiazol-5-one-3-yl)-phenylamino]-acetic acid using coupling agents like CDI or PPA in THF.
- Hydrogenate the resulting intermediate using a palladium on carbon (Pd/C) catalyst in ethanol or ethanol/water mixtures at ambient temperature and low pressure.
- Optionally carbonylate the amidino group using alkyl haloformates, such as hexyl chloroformate, in the presence of a base to form the final protected derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the CN1972919B methodology translates into tangible operational improvements beyond mere chemical elegance. The elimination of harsh acid treatment steps fundamentally alters the cost equation for manufacturing these intermediates. By removing the need for specialized acid-resistant reactors and the associated neutralization and disposal costs for acidic waste streams, manufacturers can achieve substantial cost savings in production overhead. Additionally, the milder reaction conditions reduce energy consumption, as the process does not require the high temperatures often necessary for nitrile hydrolysis. This energy efficiency contributes to a lower carbon footprint, aligning with the sustainability goals of major pharmaceutical buyers who are increasingly scrutinizing the environmental impact of their supply chains.
- Cost Reduction in Manufacturing: The streamlined workflow significantly lowers the cost of goods sold (COGS) by reducing the number of unit operations and simplifying the purification process. The high yields reported in the patent examples, often exceeding 85-90% for key steps, mean less raw material is wasted per kilogram of output. Furthermore, the use of common, commodity solvents like ethanol and THF, rather than exotic or highly regulated reagents, ensures that raw material costs remain stable and predictable. The ability to isolate intermediates as stable salts also reduces storage and handling costs, minimizing degradation losses during inventory holding periods.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply continuity by reducing the risk of batch failures due to sensitive reaction conditions. The hydrogenation step is a well-established unit operation in the fine chemical industry, meaning that contract manufacturing organizations (CMOs) can easily replicate the process without requiring specialized, hard-to-source equipment. The availability of starting materials, such as diaminobenzenes and oxadiazole precursors, is high, mitigating the risk of raw material shortages that often plague more exotic synthetic pathways. This reliability allows for more accurate demand forecasting and tighter delivery schedules, crucial for just-in-time manufacturing models in the pharmaceutical sector.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial tonnage is straightforward due to the absence of exothermic hazards associated with strong acid additions. The hydrogenation can be safely managed in standard pressure vessels, and the filtration of Pd/C catalyst is a routine operation. From an environmental standpoint, the reduction in acidic effluent simplifies wastewater treatment compliance, lowering the regulatory burden on manufacturing sites. The process generates less hazardous waste, facilitating easier disposal and reducing the overall environmental liability for the supplier, which is a key consideration for long-term partnerships with environmentally conscious multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis. Understanding these details helps stakeholders evaluate the feasibility of integrating this technology into their existing supply networks. The answers are derived directly from the experimental data and claims within the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: How does this new method improve upon the prior art WO 98/37075?
A: The conventional method described in WO 98/37075 relies on reacting benzonitriles with ammonia, which necessitates harsh acid treatment and generates significant waste burdens. The novel approach utilizes an oxadiazolone precursor that undergoes mild hydrogenation, eliminating the need for aggressive acid hydrolysis and simplifying the purification workflow.
Q: What catalysts are recommended for the hydrogenation step?
A: The patent specifies the use of transition metal catalysts such as Raney nickel, platinum oxide, or preferably palladium on activated carbon (Pd/C). Using 10% Pd/C at low pressures (1-2 bar) offers a balance of high activity and ease of filtration, which is critical for maintaining low heavy metal residues in pharmaceutical intermediates.
Q: Can this process be scaled for commercial API production?
A: Yes, the process utilizes standard unit operations such as condensation in common solvents like THF and catalytic hydrogenation in ethanol. The avoidance of extreme temperatures and the use of robust coupling agents like PPA or CDI make the route highly amenable to scale-up from kilogram to multi-ton production capacities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-(Benzimidazolylmethylamino)-benzamidine Supplier
The technological advancements detailed in CN1972919B underscore the complexity and sophistication required to produce high-quality thrombin inhibitor intermediates. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate these patented methodologies into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to verify that every batch meets the highest standards of quality and safety required for pharmaceutical applications.
We invite you to collaborate with us to leverage this efficient synthetic route for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing capabilities can enhance your supply chain efficiency and reduce your overall procurement costs.
