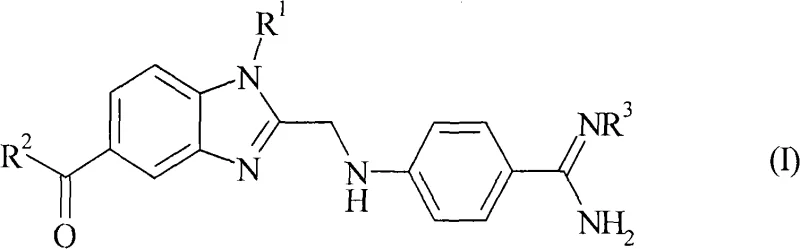
Advanced Manufacturing of 4-(Benzimidazolylmethylamino)-Benzamidines for Thrombin Inhibitors
The pharmaceutical landscape for anticoagulants continues to evolve, driven by the critical need for safer thrombin inhibitors to prevent deep vein thrombosis and stroke, particularly in patients with atrial fibrillation (SPAF). Patent CN101346360A introduces a groundbreaking improvement in the manufacturing of 4-(benzimidazolylmethylamino)-benzamidines, a core scaffold for these life-saving active pharmaceutical ingredients. This intellectual property details a robust synthetic pathway that circumvents the historically cumbersome ammonolysis of nitriles, replacing it with a more efficient condensation and hydrogenation cascade. By leveraging 2-[4-(1,2,4-oxadiazol-5-one-3-yl)-phenylamino]-acetic acid as a key building block, the process achieves high yields while utilizing inexpensive auxiliaries, marking a significant leap forward in process chemistry for cardiovascular therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of substituted (4-benzimidazol-2-ylmethylamino)-benzamidines relied heavily on the methods disclosed in international patent application WO 98/37075. This conventional approach necessitated the reaction of correspondingly substituted (4-benzimidazol-2-ylmethylamino)-benzonitriles with ammonia to generate the amidine functionality. From a production-technical perspective, this nitrile ammonolysis strategy is extremely complex and fraught with operational challenges. The reaction conditions often require high pressures and temperatures to drive the conversion of the stable nitrile group, leading to significant safety concerns in large-scale reactors. Furthermore, this method generates a high load of acidic waste that requires extensive and costly disposal procedures, creating a substantial environmental burden. The purification of the final product from the reaction mixture is also difficult, often resulting in lower overall purity and requiring additional chromatographic or recrystallization steps that erode profit margins.
The Novel Approach
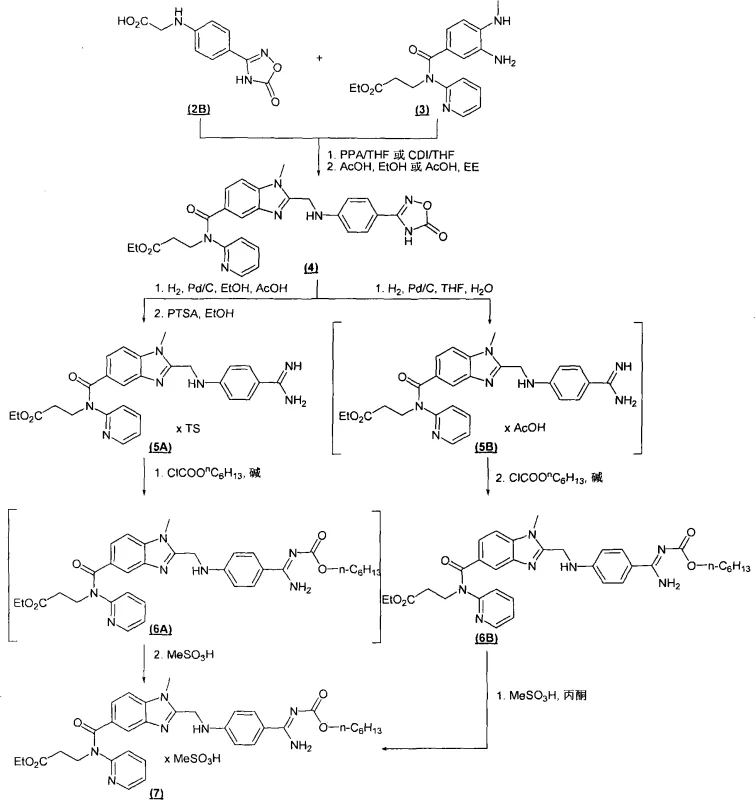
In stark contrast, the methodology presented in CN101346360A offers a streamlined alternative that fundamentally redesigns the synthetic logic. The novel approach initiates with the condensation of an optionally substituted diaminobenzene with 2-[4-(1,2,4-oxadiazol-5-one-3-yl)-phenylamino]-acetic acid. This condensation step is highly efficient and can be mediated by cost-effective coupling agents. Following this, the resulting product undergoes a catalytic hydrogenation which simultaneously reduces the nitro or other reducible groups and transforms the oxadiazolone ring into the desired amidine structure. Remarkably, this process allows for the optional carbonylation of the amidino group without the need to isolate the hydrogenated intermediate. This telescoping capability drastically reduces the number of unit operations, minimizes solvent consumption, and eliminates the hold times associated with intermediate isolation, thereby enhancing the overall throughput of the manufacturing facility.
Mechanistic Insights into Condensation and Hydrogenation Cascade
The core of this improved process lies in the strategic use of the 1,2,4-oxadiazol-5-one moiety as a masked amidine equivalent. In the initial condensation step, the carboxylic acid group of the oxadiazolone-acetic acid derivative is activated, typically using agents such as 1,1'-carbonyldiimidazole (CDI) or propane phosphonic anhydride (PPA). This activation facilitates a nucleophilic attack by the amine group of the diaminobenzene precursor, forming a stable amide bond. The choice of solvent is critical here; aprotic polar solvents like tetrahydrofuran (THF) or N-methylpyrrolidone (NMP) are preferred to ensure solubility and reaction kinetics. The presence of water-binding agents or organic bases like DIPEA further drives the equilibrium towards product formation by scavenging protons or byproducts, ensuring high conversion rates even at moderate temperatures ranging from -10°C to 50°C.
Subsequent hydrogenation is the transformative step where the oxadiazolone ring is cleaved to reveal the amidine. This is typically achieved using palladium on carbon (Pd/C) catalysts under hydrogen pressures of 1 to 10 bar. The mechanism involves the adsorption of hydrogen onto the metal surface, followed by the reduction of the N-O bonds within the heterocyclic ring. This ring-opening reaction is chemoselective, leaving other sensitive functional groups intact if properly managed. The patent highlights that this hydrogenation can be performed in protic solvents like ethanol or mixtures of THF and water, which aids in the solubility of the polar intermediates. Following hydrogenation, the free amidine can be immediately reacted with alkyl haloformates, such as n-hexyl chloroformate, in the presence of a base like potassium carbonate. This one-pot sequence prevents the degradation of the unstable free amidine and ensures that the final carbonylated product is obtained with exceptional purity, often exceeding 98% HPLC area without extensive purification.
![Synthesis pathway for the key intermediate 2-[4-(1,2,4-oxadiazol-5-one-3-yl)-phenylamino]-acetic acid](/insights/img/benzimidazolylmethylamino-benzamidine-synthesis-pharma-supplier-20260308214947-07.webp)
How to Synthesize 4-(Benzimidazolylmethylamino)-Benzamidines Efficiently
Executing this synthesis requires precise control over reaction parameters to maximize yield and minimize impurity formation. The process begins with the preparation of the oxadiazolone-acetic acid precursor, which itself is derived from 4-aminobenzonitrile via amidoxime formation and cyclization with dialkyl carbonates. Once the key acid building block is secured, it is coupled with the specific diaminobenzene derivative, such as 3-amino-4-methylaminobenzoic acid amides, under inert atmosphere. The subsequent hydrogenation must be carefully monitored to ensure complete reduction without over-reduction of the aromatic systems. For the final carbonylation, temperature control between 10°C and 20°C is vital to prevent side reactions. Detailed standardized operating procedures for each stage, including work-up and crystallization protocols, are essential for reproducible commercial manufacturing.
- Condense substituted diaminobenzene with 2-[4-(1,2,4-oxadiazol-5-one-3-yl)-phenylamino]-acetic acid using coupling agents like CDI or PPA.
- Hydrogenate the resulting intermediate using Pd/C catalyst in solvents like ethanol or THF/water mixtures to open the oxadiazolone ring.
- Optionally carbonylate the amidino group with alkyl haloformates and precipitate the final salt without isolating intermediates.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented process translates into tangible strategic benefits beyond mere technical elegance. The elimination of the high-pressure ammonia reaction significantly lowers the capital expenditure required for specialized reactor infrastructure, as standard hydrogenation vessels are sufficient. Furthermore, the ability to telescope the hydrogenation and carbonylation steps without intermediate isolation reduces the total cycle time per batch, effectively increasing the annual production capacity of existing facilities without the need for new construction. The use of commercially available and inexpensive reagents, such as alkyl chloroformates and common coupling agents, ensures that raw material costs remain stable and predictable, shielding the supply chain from volatility associated with exotic catalysts or reagents.
- Cost Reduction in Manufacturing: The new process drastically simplifies the waste management profile by avoiding the generation of high-load acidic waste streams inherent in the older nitrile ammonolysis methods. This reduction in hazardous waste directly lowers disposal costs and regulatory compliance burdens. Additionally, the high yields reported in the patent examples, often exceeding 80-90% for individual steps, mean that less raw material is required to produce the same amount of final API intermediate. The avoidance of intermediate isolation also saves on solvent usage and energy consumption for drying and evaporation, contributing to a leaner and more cost-effective manufacturing operation that enhances overall margin potential.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, such as substituted diaminobenzenes and 4-aminobenzonitrile derivatives, are commodity chemicals with robust global supply chains. This reduces the risk of supply disruption compared to processes relying on custom-synthesized, complex nitrile precursors. The robustness of the hydrogenation step, which tolerates a range of solvent systems including ethanol and water mixtures, provides flexibility in sourcing solvents, further de-risking the supply chain. Moreover, the process has been demonstrated to be scalable, with patent examples showing successful execution at the kilogram scale, indicating that technology transfer from lab to plant is straightforward and low-risk for contract manufacturing organizations.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is superior due to the milder reaction conditions and the absence of toxic ammonia gas handling at high pressures. The solvents employed, such as ethanol, ethyl acetate, and acetone, are generally regarded as green solvents with favorable recovery profiles. The ability to crystallize the final product directly from the reaction mixture or after simple solvent swaps minimizes the generation of mother liquors and reduces the volume of organic waste. This alignment with green chemistry principles not only facilitates easier permitting for new manufacturing lines but also supports the sustainability goals of modern pharmaceutical companies, making the supply chain more resilient to tightening environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this improved synthesis route. These insights are derived directly from the experimental data and claims within patent CN101346360A, providing a reliable foundation for decision-making. Understanding these nuances is critical for R&D teams evaluating technology transfer and for procurement teams assessing the long-term viability of this supply source. The answers reflect the specific advantages of the oxadiazolone pathway over traditional methods.
Q: What is the primary advantage of this new synthesis route over conventional nitrile ammonolysis?
A: The new route avoids the technically complex and waste-intensive reaction of benzimidazolylmethylamino-benzonitriles with ammonia, significantly reducing acid load disposal requirements.
Q: Can the hydrogenation and carbonylation steps be telescoped?
A: Yes, the patent describes a process where the amidino group can be carbonylated without prior isolation of the hydrogenated intermediate product, streamlining the workflow.
Q: What catalysts are preferred for the hydrogenation step?
A: Palladium on activated carbon (Pd/C), specifically 5-10% Pd/C moistened with water, is preferred for efficient reduction under mild pressure conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-(Benzimidazolylmethylamino)-Benzamidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable and high-quality supply of complex pharmaceutical intermediates like 4-(benzimidazolylmethylamino)-benzamidines. Our technical team has thoroughly analyzed the pathways described in CN101346360A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are equipped with state-of-the-art hydrogenation reactors and stringent purity specifications enforced by our rigorous QC labs to ensure that every batch meets the exacting standards required for thrombin inhibitor synthesis. Our commitment to process excellence means we can deliver this key intermediate with consistent quality, supporting your drug development timelines and commercial launch goals.
We invite you to collaborate with us to leverage these advanced manufacturing capabilities for your specific project needs. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this optimized route can improve your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us be your partner in navigating the complexities of cardiovascular API manufacturing with confidence and efficiency.
