Scalable Synthesis of 5-Methyl-3,4-Diphenyl Isoxazole for Pharmaceutical Intermediates
Scalable Synthesis of 5-Methyl-3,4-Diphenyl Isoxazole for Pharmaceutical Intermediates
The pharmaceutical industry constantly seeks robust, cost-effective pathways for critical intermediates, particularly those serving as scaffolds for potent analgesics. Patent CN112409284A discloses a groundbreaking synthetic method for 5-methyl-3,4-diphenyl isoxazole, a pivotal intermediate in the manufacture of Parecoxib sodium, a widely used COX-2 inhibitor. This novel approach addresses longstanding inefficiencies in traditional synthesis by leveraging a continuous, three-step sequence starting from the inexpensive and commercially abundant dibenzoyl methane. By replacing precious metal catalysts with accessible Lewis acids, this technology offers a compelling value proposition for reliable API intermediate suppliers aiming to optimize their production portfolios. The structural integrity and biological relevance of this isoxazole derivative make it a cornerstone in modern pain management therapeutics.
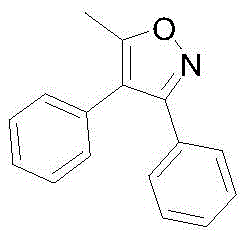
As a key building block, 5-methyl-3,4-diphenyl isoxazole possesses a unique heterocyclic framework that imparts significant metabolic stability and binding affinity in downstream drug molecules. The demand for high-purity variants of this compound is driven by the expanding market for non-opioid analgesics, which seek to mitigate the side effects associated with morphine derivatives. The technical breakthrough detailed in the patent lies not just in the chemical transformation itself, but in the holistic design of the process flow, which prioritizes operational simplicity and raw material accessibility. For procurement teams and R&D directors alike, understanding the nuances of this synthesis is essential for securing a stable supply chain for next-generation pain relief medications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3,4-diaryl isoxazoles has been plagued by reliance on scarce precursors and expensive catalytic systems. As illustrated in prior art, such as the route published in the Journal of Heterocyclic Chemistry, traditional methods often employ silver salts to facilitate the crucial rearrangement and cyclization steps. While chemically effective, the use of silver introduces severe economic and environmental drawbacks, including high catalyst costs and the complex downstream processing required to remove trace heavy metals to meet pharmaceutical standards. Furthermore, the starting materials in these legacy routes are often not commodity chemicals, requiring multi-step preparation that inflates the overall cost of goods sold (COGS) and extends lead times significantly.
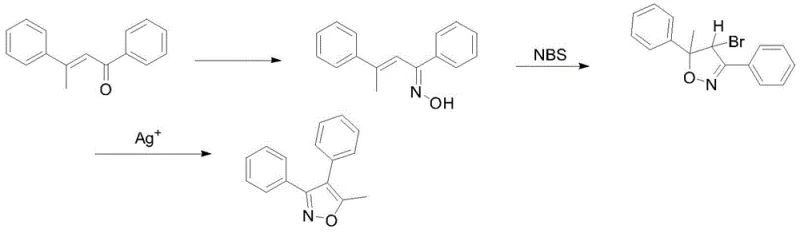
Beyond the financial implications, the operational complexity of silver-catalyzed reactions poses risks for commercial scale-up of complex pharmaceutical intermediates. The sensitivity of silver catalysts to reaction conditions and the potential for catalyst deactivation can lead to batch-to-batch variability, complicating quality control efforts. Additionally, the disposal of silver-containing waste streams requires specialized treatment protocols to comply with increasingly stringent environmental regulations. These factors collectively create a fragile supply chain that is vulnerable to price volatility in the precious metals market and regulatory shifts regarding heavy metal residues in active pharmaceutical ingredients.
The Novel Approach
In stark contrast, the methodology outlined in CN112409284A revolutionizes the production landscape by utilizing dibenzoyl methane as the primary feedstock. This strategic shift eliminates the dependency on exotic starting materials, grounding the synthesis in the realm of bulk fine chemicals. The new route proceeds through a logical sequence of oximation, Grignard addition, and Lewis acid-catalyzed cyclization, each step designed for maximum atom economy and ease of execution. By substituting the silver catalyst with boron-based Lewis acids like boron trifluoride etherate or boron trichloride, the process achieves cost reduction in pharmaceutical intermediates manufacturing without compromising on yield or purity.
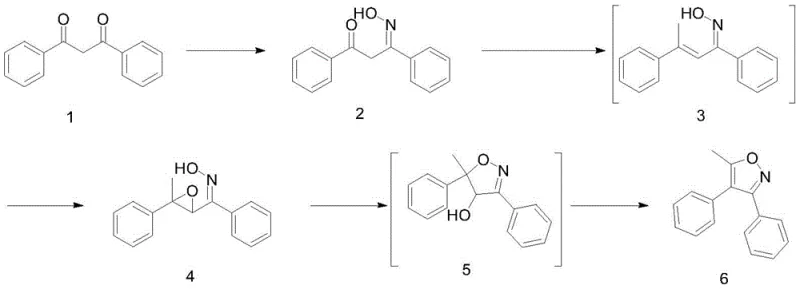
The continuity of this novel process is another major advantage, allowing for seamless integration of unit operations. The transition from the ketoxime intermediate to the final isoxazole product involves standard workup procedures such as extraction, concentration, and recrystallization, which are familiar to any mid-to-large scale chemical manufacturing facility. This familiarity reduces the barrier to entry for contract development and manufacturing organizations (CDMOs) looking to adopt the technology. The result is a streamlined workflow that enhances reaction continuity and industrial operability, providing a solid foundation for the large-scale application of downstream medicines like Parecoxib sodium.
Mechanistic Insights into Lewis Acid-Catalyzed Cyclization
The heart of this synthetic innovation lies in the final cyclization step, where a sophisticated rearrangement transforms the epoxide-oxime intermediate into the target isoxazole ring. The mechanism begins with the activation of the epoxide oxygen by the Lewis acid (e.g., BF3), which increases the electrophilicity of the adjacent carbon atoms. This activation facilitates a nucleophilic attack by the oxime nitrogen, triggering a ring-opening and subsequent cyclization cascade. The precise control of reaction temperature, maintained between 35-40°C, is critical to directing the pathway towards the desired 5-methyl substitution pattern while suppressing potential regioisomers or polymerization byproducts.
Impurity control is rigorously managed through the selection of specific acidic conditions during the dehydration phase. The use of trifluoroacetic acid or p-toluenesulfonic acid ensures efficient water elimination, driving the equilibrium towards the aromatic isoxazole system. The patent data indicates that this mechanistic precision results in a final product purity exceeding 99.0% after a single recrystallization from ethanol-water. Such high purity is paramount for high-purity API intermediates, as it minimizes the burden on downstream purification processes and ensures the safety profile of the final drug substance meets global pharmacopeial standards.
How to Synthesize 5-Methyl-3,4-Diphenyl Isoxazole Efficiently
The synthesis is executed in three distinct stages, beginning with the formation of the monooxime, followed by Grignard addition and epoxidation, and concluding with the Lewis acid cyclization. Each stage utilizes common solvents like methanol, tetrahydrofuran, and dichloromethane, ensuring compatibility with standard glass-lined or stainless steel reactors. The detailed standardized synthetic steps below outline the specific molar ratios, temperature profiles, and workup procedures required to replicate the high yields reported in the patent documentation.
- Perform oximation of dibenzoyl methane using hydroxylamine hydrochloride or sulfate in methanol/water or ethanol/water at 25-32°C to form the monooxime intermediate.
- React the monooxime with methyl Grignard reagent (MeMgCl or MeMgBr) at -15 to 0°C, followed by acid-catalyzed dehydration and epoxidation to obtain the oxirane ketoxime.
- Execute Lewis acid catalyzed cyclization using Boron trifluoride etherate or Boron trichloride in dichloromethane, followed by acid treatment and recrystallization to yield the final isoxazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route translates into tangible strategic benefits that extend far beyond simple chemistry. The shift from precious metal catalysts to commodity Lewis acids fundamentally alters the cost structure of the intermediate, removing exposure to the volatile pricing of silver. Moreover, the reliance on dibenzoyl methane, a widely produced chemical, ensures a robust and diversified supply base, mitigating the risk of raw material shortages that can halt production lines. This resilience is crucial for maintaining reducing lead time for high-purity pharmaceutical intermediates in a competitive global market.
- Cost Reduction in Manufacturing: The elimination of expensive silver catalysts and the use of low-cost starting materials like dibenzoyl methane lead to a substantial decrease in direct material costs. Furthermore, the simplified purification process, which avoids complex heavy metal scavenging steps, reduces the consumption of auxiliary chemicals and lowers waste disposal expenses. These cumulative savings allow for a more competitive pricing strategy for the final API, enhancing market share potential.
- Enhanced Supply Chain Reliability: By utilizing reagents such as methyl magnesium chloride and boron trifluoride, which are staples in the fine chemical industry, the process ensures consistent availability and short delivery times. The robustness of the reaction conditions also means that the process is less susceptible to minor fluctuations in utility supplies or environmental conditions, guaranteeing a steady flow of material to downstream customers.
- Scalability and Environmental Compliance: The process is designed with scale-up in mind, utilizing exothermic reactions that can be safely managed in large vessels with standard cooling capabilities. The absence of heavy metals simplifies effluent treatment, aligning with green chemistry principles and reducing the regulatory burden associated with environmental compliance. This makes the technology an attractive option for manufacturers seeking to expand capacity without significant capital investment in specialized waste treatment infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of 5-methyl-3,4-diphenyl isoxazole. These answers are derived directly from the experimental data and beneficial effects described in the patent literature, providing a clear picture of the technology's capabilities and limitations for potential partners.
Q: Why is the new synthesis route preferred over the conventional silver-catalyzed method?
A: The conventional method relies on expensive silver catalysts and difficult-to-obtain raw materials, creating significant cost and supply chain bottlenecks. The new route utilizes readily available dibenzoyl methane and common Lewis acids, drastically reducing raw material costs and eliminating the need for complex heavy metal removal processes.
Q: What represents the critical quality attributes for this intermediate?
A: The process described in patent CN112409284A achieves a final chemical purity of greater than 99.0% through a final recrystallization step. The overall yield across the three steps ranges from 62% to 65%, ensuring high efficiency and minimal waste generation compared to older methodologies.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process utilizes standard fine chemical unit operations such as simple filtration, concentration, and recrystallization. The reaction conditions are moderate (mostly ambient to 45°C), and the reagents are industrially common, making the scale-up from pilot to multi-ton production highly feasible and safe.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Methyl-3,4-Diphenyl Isoxazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient intermediate synthesis plays in the broader pharmaceutical ecosystem. Our team of expert chemists has thoroughly analyzed the route disclosed in CN112409284A and is fully prepared to implement this technology at scale. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 5-methyl-3,4-diphenyl isoxazole meets the highest industry standards.
We invite you to collaborate with us to leverage this advanced synthetic route for your Parecoxib sodium projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized manufacturing capabilities can drive value and efficiency in your supply chain.
