Advanced Aziridine Ring-Opening Technology for Scalable Pharmaceutical Intermediate Production
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to synthesize complex nitrogen-containing heterocycles, which serve as critical building blocks for active pharmaceutical ingredients (APIs). A significant breakthrough in this domain is detailed in patent CN102875421A, which discloses a novel method for the ring-opening of aziridine compounds utilizing p-nitrobenzoic acid. This technology addresses long-standing challenges in organic synthesis by replacing expensive and sensitive catalysts with robust, cost-effective alkali metal hydroxides. For R&D directors and procurement managers alike, this innovation represents a pivotal shift towards more sustainable and economically viable manufacturing processes for beta-amino compounds. The method leverages the unique nucleophilic properties of p-nitrobenzoic acid in a dimethyl sulfoxide (DMSO) solvent system, achieving high yields and exceptional regioselectivity under mild conditions. By understanding the technical nuances of this patent, stakeholders can better appreciate its potential to streamline supply chains and reduce the overall cost of goods sold for complex intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the nucleophilic ring-opening of aziridines has been fraught with significant technical and economic hurdles that hinder large-scale adoption. Traditional methodologies often rely on strong Lewis acids, such as boron trifluoride or scandium triflate, to activate the aziridine ring for nucleophilic attack. While effective in laboratory settings, these reagents are prohibitively expensive, highly moisture-sensitive, and difficult to handle safely on an industrial scale. For instance, the use of Indium Triflate, while offering good yields, introduces a heavy metal burden that requires rigorous and costly removal steps to meet pharmaceutical purity standards. Furthermore, many conventional processes suffer from poor regioselectivity, leading to complex mixtures of isomers that are difficult and wasteful to separate. The reliance on harsh reaction conditions, including extreme temperatures or anhydrous environments that are difficult to maintain, further exacerbates operational costs and safety risks. These limitations create a bottleneck in the supply chain, resulting in longer lead times and higher prices for downstream API manufacturers who depend on these critical intermediates.
The Novel Approach
In stark contrast to these legacy methods, the technology described in patent CN102875421A introduces a streamlined and highly efficient protocol that fundamentally alters the economic landscape of aziridine functionalization. By utilizing p-nitrobenzoic acid as the nucleophilic reagent and simple alkali metal hydroxides like potassium hydroxide (KOH) or cesium hydroxide (CsOH) as catalysts, the process eliminates the need for precious metal catalysts entirely. The reaction proceeds smoothly in dimethyl sulfoxide (DMSO), a solvent known for its ability to stabilize charged intermediates and its remarkable tolerance to trace amounts of water, thereby relaxing the stringent drying requirements typical of organometallic chemistry. This approach not only simplifies the operational workflow but also drastically reduces the raw material costs associated with the synthesis. The mild reaction temperatures, typically ranging from 40°C to 60°C, further enhance the safety profile and energy efficiency of the process. This novel pathway ensures that high-purity beta-amino esters can be produced with superior regiocontrol, making it an ideal solution for the commercial scale-up of complex pharmaceutical intermediates.
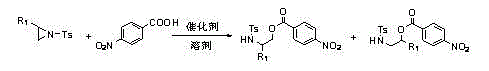
The core of this technological advancement lies in the specific interaction between the activated aziridine substrate and the carboxylic acid nucleophile. The tosyl group on the nitrogen atom acts as a powerful electron-withdrawing substituent, which significantly reduces the electron density on the aziridine ring. This electronic modulation renders the ring carbons highly susceptible to nucleophilic attack by the carboxylate anion generated in situ. The use of DMSO as the solvent is critical, as its high polarity facilitates the dissolution of both the organic substrate and the inorganic base, creating a homogeneous reaction environment that promotes rapid kinetics. Mechanistically, the alkali metal hydroxide deprotonates the p-nitrobenzoic acid to form a reactive carboxylate species, which then attacks the less sterically hindered carbon of the aziridine ring. This SN2-like displacement results in the cleavage of the C-N bond and the formation of the beta-amino ester linkage. The elegance of this mechanism is its simplicity; it avoids the formation of stable coordination complexes that often plague transition metal catalysis, thereby minimizing the risk of metal contamination in the final product. For quality control teams, this means a cleaner impurity profile and a more straightforward purification process, ultimately leading to higher overall process efficiency.
Furthermore, the regioselectivity of this reaction is a standout feature that provides substantial value to synthetic chemists designing complex molecular architectures. The patent data indicates that when the substituent on the aziridine ring is an aliphatic chain, the reaction exhibits extraordinary regioselectivity, favoring the attack at the less hindered position. This selectivity is driven by a combination of steric factors and the electronic influence of the tosyl group, which directs the nucleophile to the specific carbon center. In cases where aromatic substituents are present, the reaction still maintains high fidelity, producing the desired isomers with minimal formation of byproducts. This level of control is paramount in the synthesis of chiral drugs, where the wrong isomer can be inactive or even toxic. By providing a predictable and reliable method for installing the amino-ester functionality, this technology allows R&D teams to plan synthetic routes with greater confidence, reducing the need for extensive optimization studies and accelerating the timeline from benchtop discovery to pilot plant production.
How to Synthesize Beta-Amino Esters Efficiently
To implement this cutting-edge synthesis in a laboratory or pilot plant setting, operators must adhere to specific procedural guidelines that maximize yield and purity while ensuring safety. The process begins with the careful selection of reagents, specifically ensuring that the tosyl-activated aziridine is of high quality to prevent side reactions. The reaction is typically conducted by mixing the aziridine substrate with a slight excess of p-nitrobenzoic acid in DMSO, followed by the addition of the alkali hydroxide catalyst. The mixture is then heated to a moderate temperature, usually between 40°C and 60°C, and stirred for a period ranging from 2 to 10 hours depending on the specific substrate structure. Upon completion, the reaction mixture is quenched with a carbonate solution to neutralize any remaining acid, followed by standard extraction and purification techniques such as silica gel chromatography. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction system by dissolving the tosyl-activated aziridine compound and p-nitrobenzoic acid in anhydrous dimethyl sulfoxide (DMSO) solvent.
- Add the alkali metal hydroxide catalyst (such as KOH or CsOH) to the mixture, ensuring a molar ratio of 5-40% relative to the substrate.
- Heat the reaction mixture to a temperature between 40-60°C and stir for 2-10 hours until completion, followed by standard workup and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this aziridine ring-opening technology translates into tangible strategic advantages that directly impact the bottom line. The most immediate benefit is the drastic reduction in raw material costs achieved by substituting expensive Lewis acid catalysts with commodity chemicals like potassium hydroxide. This shift not only lowers the direct cost of goods but also mitigates the supply risk associated with sourcing specialized reagents that may have volatile markets or long lead times. Additionally, the robustness of the reaction conditions means that the process is less sensitive to environmental variables, reducing the rate of batch failures and ensuring a more consistent supply of intermediates. The ability to operate under mild temperatures and in a solvent system that tolerates micro-moisture further simplifies the infrastructure requirements, allowing for production in standard stainless steel reactors without the need for exotic lining or cryogenic cooling systems. These factors collectively contribute to a more resilient and cost-effective supply chain capable of meeting the demanding schedules of the global pharmaceutical market.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts such as indium or scandium removes a significant cost driver from the manufacturing equation. Unlike traditional methods that require stoichiometric or near-stoichiometric amounts of expensive metals, this process utilizes catalytic amounts of inexpensive alkali hydroxides. Furthermore, the simplified workup procedure reduces the consumption of auxiliary materials and solvents required for metal scavenging and purification. The overall process efficiency is enhanced by the high yields reported across a broad range of substrates, minimizing waste and maximizing the output per batch. This economic efficiency allows manufacturers to offer more competitive pricing for high-purity pharmaceutical intermediates while maintaining healthy profit margins.
- Enhanced Supply Chain Reliability: The reliance on widely available and stable reagents ensures that production schedules are not disrupted by shortages of niche chemicals. Potassium hydroxide and p-nitrobenzoic acid are commodity chemicals with established global supply chains, providing a buffer against market volatility. The tolerance of the reaction to trace water in the DMSO solvent also reduces the logistical burden of maintaining strictly anhydrous conditions, which can be a source of delay and complexity in large-scale operations. This operational flexibility enables suppliers to respond more rapidly to fluctuating demand, reducing lead times for high-purity pharmaceutical intermediates and ensuring continuity of supply for downstream API production.
- Scalability and Environmental Compliance: The benign nature of the reagents and the absence of heavy metals make this process inherently more environmentally friendly and easier to scale. Waste streams are simpler to treat, as they do not contain toxic metal residues that require specialized disposal protocols. The use of DMSO, while requiring proper recovery systems, is a well-understood solvent in the industry with established recycling methods. The mild reaction conditions reduce energy consumption compared to high-temperature or high-pressure alternatives, aligning with modern green chemistry principles. This alignment not only reduces the environmental footprint but also simplifies regulatory compliance, facilitating faster approval for commercial scale-up of complex pharmaceutical intermediates in regulated markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this aziridine ring-opening technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent literature, providing a reliable basis for decision-making. Understanding these details is crucial for evaluating the feasibility of integrating this method into existing production workflows. The responses cover aspects ranging from catalyst selection to product isolation, ensuring a comprehensive overview of the process capabilities.
Q: What are the advantages of using p-nitrobenzoic acid over traditional Lewis acids?
A: Unlike expensive and moisture-sensitive Lewis acids like Indium Triflate, p-nitrobenzoic acid combined with alkali metal hydroxides offers a significantly cheaper, more robust, and environmentally friendly catalytic system with excellent water tolerance.
Q: How does this method improve regioselectivity in aziridine ring-opening?
A: The method demonstrates extraordinary regioselectivity, particularly when the aziridine substituent is an aliphatic chain, allowing for the precise formation of specific beta-amino ester isomers crucial for drug synthesis.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the use of common solvents like DMSO, inexpensive catalysts like KOH, and mild reaction temperatures (40-60°C) makes this process highly scalable and safe for industrial production without requiring specialized equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aziridine Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic methodologies like the p-nitrobenzoic acid mediated ring-opening of aziridines. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate such innovative patent technologies into robust commercial processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab to plant is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which are equipped with state-of-the-art analytical instrumentation to verify identity and potency. Whether you require custom synthesis of complex beta-amino esters or large-scale supply of key intermediates, our facility is designed to support your growth with reliability and precision.
We invite you to collaborate with us to leverage this cost-effective technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact us to request specific COA data and route feasibility assessments that demonstrate how we can optimize your supply chain. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable aziridine compound supplier dedicated to driving innovation and efficiency in the pharmaceutical industry.
