Advanced Load Change Strategy for Diphenylmethane Diamine Manufacturing and Commercial Scale-Up
Advanced Load Change Strategy for Diphenylmethane Diamine Manufacturing and Commercial Scale-Up
The global demand for high-performance polyurethanes requires a supply chain capable of adapting to fluctuating market needs without compromising product integrity. Patent CN110291063B introduces a groundbreaking methodology for producing di-and polyamines of the diphenylmethane series (MDA) that addresses one of the most persistent challenges in continuous chemical manufacturing: maintaining product consistency during production load changes. Traditionally, altering the throughput of a continuous MDA plant inevitably changes the residence time of the reaction mixture, often leading to incomplete rearrangement or the formation of colored impurities. This invention solves that problem by dynamically adapting the molar ratios of aniline to formaldehyde and acidic catalyst to aniline during the transition state. By strictly controlling these parameters, the process ensures that the rearrangement reaction proceeds to completion regardless of whether the plant is ramping up to full capacity or scaling down, thereby securing a reliable MDA supplier status for downstream users.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
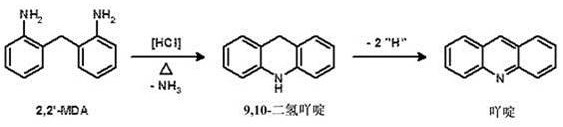
In conventional continuous processes for MDA synthesis, operating parameters are typically optimized for a steady-state condition, often at a specific rated load. When market demands shift and production capacity needs to be altered, operators face a significant technical hurdle. A change in load directly impacts the residence time of the reaction mixture within the fixed volume of the reactor cascade. If the load is increased without compensatory measures, the residence time decreases, potentially leaving the acid-catalyzed rearrangement incomplete. Conversely, decreasing the load increases residence time, which can promote secondary reactions. A critical failure mode in these scenarios is the formation of undesirable byproducts, particularly those with acridine and 9,10-dihydroacridine structures. These impurities are not merely cosmetic defects; they can severely impact the quality of the subsequent phosgenation step to produce MDI, leading to off-spec isocyanates with poor color values and stability.
The Novel Approach
The novel approach disclosed in the patent fundamentally decouples product quality from residence time fluctuations by introducing a dynamic control strategy for the transition state. Instead of keeping molar ratios constant, the method mandates specific adjustments based on the direction of the load change. For instance, when increasing the load (and thus decreasing residence time), the process decreases the molar ratio of total aniline to total formaldehyde while simultaneously increasing the molar ratio of the acidic catalyst to total aniline. This dual adjustment accelerates the reaction kinetics to match the shorter residence time, ensuring complete conversion. This sophisticated control logic allows for seamless transitions between operating states, such as moving from half-load to rated load, without generating the off-spec material that plagues traditional methods. It represents a paradigm shift from static operation to adaptive process control in fine chemical manufacturing.
Mechanistic Insights into Acid-Catalyzed Rearrangement Dynamics
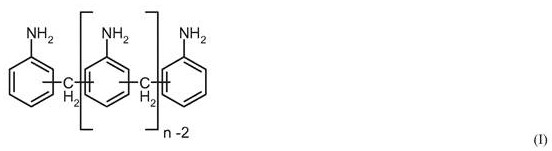
The core of this technology lies in the precise manipulation of the acid-catalyzed rearrangement of aminals to diamines. The reaction cascade typically involves a series of reactors where temperature is progressively increased, for example from 50.0°C in the first reactor to over 150.0°C in the final reactor. The degree of protonation, defined as the molar ratio of acid catalyst to aniline, is a critical lever. In the transition state, if the load is increased, the patent specifies increasing the protonation degree (e.g., from 10% to 13%) to drive the equilibrium forward more rapidly. This is chemically intuitive yet operationally complex, as excessive acid can promote the degradation of sensitive isomers like 2,2'-MDA into polycyclic aromatic impurities. The patent data demonstrates that by carefully balancing the acid concentration against the aniline excess, the formation of acridine derivatives is minimized even under non-steady-state conditions. This mechanistic understanding is vital for R&D teams aiming to replicate this stability in their own pilot or commercial units.
Furthermore, the control of isomer distribution is paramount for downstream polyurethane applications. The process targets a specific composition of 4,4'-MDA, 2,4'-MDA, and higher homologues (PMDA). The data indicates that the inventive method maintains a consistent dinuclear content (around 51%) and low levels of N-methyl and N-formyl impurities during load transitions. This consistency is achieved because the adjusted molar ratios effectively normalize the reaction environment experienced by the molecules, regardless of flow rate. For a research director, this implies that the kinetic profile of the rearrangement is being actively managed to counteract the hydrodynamic changes in the reactor. The suppression of N-substituted byproducts is particularly noteworthy, as these can act as chain terminators or discoloration precursors in the final polymer, making this level of impurity control a significant technical advantage for high-purity MDA production.
How to Synthesize Diphenylmethane Diamines Efficiently
The synthesis of high-quality MDA requires a rigorous adherence to the adaptive protocol described in the patent, particularly during the critical phases of production scaling. The process begins with the condensation of aniline and formaldehyde in an aminal reactor, followed by phase separation to isolate the organic aminal phase. This stream is then fed into a reactor cascade where the acid-catalyzed rearrangement occurs. The key to success lies in the transition management: operators must monitor the formaldehyde feed rate as the primary indicator of load and adjust the aniline and acid feeds in real-time according to the specified ratio boundaries. Detailed standard operating procedures for managing these molar ratio shifts, including the specific temperature gradients required across the reactor cascade to ensure viscosity control and reaction completion, are essential for safe and effective implementation. The following guide outlines the standardized steps for executing this synthesis efficiently.
- Condense aniline and formaldehyde in an aminal reactor at elevated temperatures to form the aminal intermediate, followed by phase separation to remove aqueous byproducts.
- Feed the organic aminal phase into a reactor cascade with an acidic catalyst, maintaining a temperature gradient from approximately 50°C to 150°C across the series.
- During production load changes, dynamically adjust the total aniline-to-formaldehyde molar ratio and the acid catalyst-to-aniline ratio to compensate for residence time variations.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the ability to maintain product quality during production fluctuations translates directly into cost avoidance and supply security. Traditional batch or rigid continuous processes often require lengthy stabilization periods after a load change, during which tons of off-spec material may be produced and subsequently discarded or reworked. This inventive method drastically reduces the generation of such waste by ensuring that the product remains within specification throughout the transition. This capability allows manufacturers to respond agilely to sudden spikes in demand or raw material shortages without the fear of compromising the quality of the entire batch. Consequently, this leads to substantial cost savings by maximizing yield and minimizing the disposal costs associated with hazardous chemical waste, offering a clear economic advantage in cost reduction in polyurethane precursor manufacturing.
- Cost Reduction in Manufacturing: The elimination of off-spec production during load changes removes a significant hidden cost center in chemical manufacturing. By avoiding the formation of byproducts like acridines and incomplete rearrangement products, the process ensures that nearly all raw materials are converted into saleable product. This efficiency means that expensive raw materials like aniline and formaldehyde are not wasted on material that must be incinerated. Furthermore, the reduction in byproduct formation simplifies the downstream purification steps, potentially reducing energy consumption in distillation columns and extending the lifecycle of catalysts and equipment. These operational efficiencies compound over time to deliver significant financial benefits without requiring capital-intensive plant modifications.
- Enhanced Supply Chain Reliability: In a volatile global market, the ability to scale production up or down rapidly is a strategic asset. This technology enables a production facility to switch from half-load to full-load operation within a short timeframe, typically around two hours, while maintaining product consistency. For supply chain planners, this means shorter lead times and the ability to fulfill urgent orders without the traditional lag time associated with process stabilization. It reduces the risk of supply interruptions caused by technical issues related to load changes, ensuring a steady flow of high-purity MDA to downstream customers. This reliability strengthens the partnership between the supplier and the manufacturer, fostering long-term contractual stability.
- Scalability and Environmental Compliance: The process is designed for continuous operation, which is inherently more scalable than batch processing. The controlled management of reaction parameters ensures that environmental emissions remain stable even during transients. By minimizing the formation of heavy byproducts and ensuring complete reaction, the load on wastewater treatment facilities is reduced. The method aligns with modern green chemistry principles by maximizing atom economy and reducing the E-factor (mass of waste per mass of product). For companies facing increasing regulatory scrutiny regarding industrial emissions and waste disposal, adopting this optimized load change strategy provides a pathway to improved environmental compliance and a smaller carbon footprint per ton of product produced.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this load change strategy in industrial settings. Understanding these nuances is critical for engineering teams evaluating the feasibility of adopting this method in existing infrastructure. The answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making.
Q: How does the patented method prevent off-spec products during load changes?
A: The method prevents off-spec products by dynamically adjusting the molar ratios of reactants and catalysts. Specifically, when increasing load, the aniline-to-formaldehyde ratio is decreased while the acid catalyst-to-aniline ratio is increased, ensuring complete rearrangement despite reduced residence time.
Q: What are the critical byproducts suppressed by this process?
A: The process significantly suppresses the formation of undesirable byproducts such as acridine and 9,10-dihydroacridine, which typically form from 2,2'-MDA under improper acid catalysis conditions and can cause color issues in downstream MDI production.
Q: Is this process suitable for large-scale continuous manufacturing?
A: Yes, the patent explicitly describes a continuous operation mode suitable for industrial plants, allowing for rapid transitions between half-load and rated-load production without technical interruptions or quality deviations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable MDA Supplier
At NINGBO INNO PHARMCHEM, we recognize that the theoretical advantages of a patent must be translated into practical, commercial reality to create value for our partners. Our team of process engineers possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex synthetic routes like the adaptive load change method for MDA are implemented with precision. We understand that maintaining stringent purity specifications is non-negotiable for polyurethane applications, which is why our rigorous QC labs employ advanced analytical techniques to monitor isomer ratios and trace impurities like acridines. We are committed to delivering high-purity MDA that meets the exacting standards required for next-generation polymer applications, leveraging our deep technical expertise to optimize every step of the value chain.
We invite you to collaborate with us to explore how this advanced manufacturing technology can optimize your supply chain. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage potential partners to contact us to request specific COA data and route feasibility assessments, allowing you to verify the superior consistency and purity of our offerings. By partnering with us, you gain access to a supply chain that is not only robust and reliable but also technologically advanced enough to adapt to your evolving business needs without compromising on quality.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
