Advanced Rh-Catalyzed Process Delivers High-Purity Indanone Derivatives with Unmatched Scalability for Pharmaceutical Manufacturing
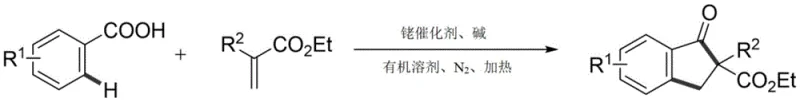
Patent CN112939780B introduces a groundbreaking synthetic methodology for indanone derivatives addressing critical limitations in traditional organic synthesis routes This innovative approach enables the direct conversion of readily available benzoic acid compounds and acrylate esters into valuable 2-substituted indanone structures through a single catalytic transformation The method eliminates the need for pre-functionalization steps and harsh reaction conditions that have long plagued conventional Friedel-Crafts acylation and Nazarov cyclization processes By leveraging rhodium-catalyzed C-H bond activation under mild nitrogen atmosphere this technology achieves superior functional group tolerance and operational simplicity while delivering high reaction yields across diverse substrate combinations The patent represents a significant advancement in sustainable pharmaceutical intermediate manufacturing offering enhanced atom economy and reduced environmental impact compared to legacy methodologies This breakthrough is particularly relevant for multinational pharmaceutical companies seeking efficient routes to complex molecular scaffolds found in numerous drug candidates and natural products
The Limitations of Conventional Methods vs The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to indanone synthesis primarily Friedel-Crafts acylation and Nazarov cyclization suffer from severe operational constraints that hinder their industrial applicability These methods require strong Lewis or Brønsted acids under rigorous conditions often leading to substrate decomposition and poor selectivity when sensitive functional groups are present The necessity for pre-functionalization of starting materials creates additional synthetic steps significantly reducing overall atom economy and increasing production costs through multiple purification stages Furthermore these conventional routes exhibit limited substrate scope particularly with electron-deficient aromatic systems and frequently generate hazardous waste streams requiring complex disposal protocols The harsh reaction environments also necessitate specialized corrosion-resistant equipment adding substantial capital expenditure for manufacturing facilities These cumulative drawbacks result in extended production timelines inconsistent product quality and elevated manufacturing costs that are increasingly incompatible with modern pharmaceutical industry demands for sustainable and cost-effective processes
The Novel Approach
The patented methodology overcomes these challenges through an elegant rhodium-catalyzed C-H activation strategy that directly couples benzoic acid derivatives with acrylate compounds in a single transformation step This innovative process operates under mild nitrogen atmosphere at moderate temperatures (140°C) using environmentally benign organic solvents like 1,2-dichloroethane eliminating the need for corrosive acids or extensive pre-treatment of substrates The rhodium catalyst system demonstrates exceptional functional group compatibility tolerating halogens (F Cl Br) alkyl groups and cyano substituents without protection-deprotection sequences Crucially the reaction achieves high yields while maintaining excellent regioselectivity for the 2-substituted indanone products This streamlined approach reduces the synthetic sequence from multiple steps to a single operation significantly improving process efficiency and reducing waste generation through superior atom economy The methodology's robustness across diverse substrates makes it particularly valuable for producing complex pharmaceutical intermediates with stringent purity requirements
![Optimized reaction conditions showing rhodium-catalyzed synthesis parameters including [Cp*RhCl₂]₂ catalyst loading sodium acetate base DCE solvent at 140°C under nitrogen atmosphere](/insights/img/indanone-rh-catalysis-pharma-supplier-20260301112016-013.webp)
Mechanistic Insights into Rh-Catalyzed C-H Activation for Indanone Synthesis
The catalytic cycle begins with coordination of the rhodium complex to the carboxylate group of the benzoic acid substrate facilitating ortho-C-H bond activation through a concerted metalation-deprotonation mechanism This key step generates a rhodacycle intermediate that undergoes migratory insertion with the acrylate compound forming a new carbon-carbon bond with precise regiocontrol Subsequent β-hydride elimination and reductive elimination steps complete the cyclization process yielding the indanone core structure while regenerating the active rhodium catalyst species The base (sodium acetate) plays a critical role in facilitating proton transfer during the C-H activation step without participating in side reactions contributing to the method's high functional group tolerance This mechanism avoids the formation of unstable carbocation intermediates common in traditional acid-catalyzed routes thereby minimizing unwanted rearrangements and byproduct formation that typically complicate purification processes in pharmaceutical manufacturing
Impurity profile control is inherently achieved through the reaction's high regioselectivity and mild conditions which prevent common side reactions such as over-acylation or polymerization observed in conventional methods The absence of strong acids eliminates acid-mediated decomposition pathways while the well-defined catalytic cycle minimizes catalyst-derived impurities through efficient turnover Post-reaction workup involves straightforward filtration and chromatographic purification with no requirement for heavy metal removal steps since the rhodium catalyst loading is minimal (5 mol%) and easily separated from the product stream This inherent process robustness ensures consistent production of high-purity indanone derivatives meeting pharmaceutical industry standards without additional costly purification stages directly addressing R&D directors' concerns about impurity management in complex molecule synthesis
How to Synthesize Indanone Derivatives Efficiently
This patented synthetic route represents a significant advancement in indanone production methodology offering pharmaceutical manufacturers a streamlined pathway to these valuable molecular scaffolds The process eliminates multiple synthetic steps required by conventional approaches while maintaining excellent yield and purity profiles across diverse substrate combinations By utilizing commercially available starting materials and standard laboratory equipment this method provides immediate operational advantages for R&D teams seeking to accelerate process development timelines The following standardized procedure details the precise implementation of this innovative technology for reliable scale-up from laboratory to commercial production environments
- Combine benzoic acid compound (0.3 mmol), acrylate compound (0.6 mmol), [Cp*RhCl₂]₂ catalyst (5 mol%), and sodium acetate (0.3 mmol) in 0.4 mL DCE solvent under nitrogen atmosphere.
- Heat the reaction mixture at 140°C for 12 hours with continuous stirring under nitrogen protection.
- After cooling, perform suction filtration followed by silica gel sample mixing and column chromatography purification using petroleum ether/ethyl acetate solvent system.
Commercial Advantages for Procurement and Supply Chain Teams
Pharmaceutical procurement and supply chain professionals will recognize substantial value in this patented methodology through its direct impact on cost structure supply reliability and manufacturing flexibility The elimination of multi-step sequences and hazardous reagents creates immediate opportunities for cost optimization while enhancing operational safety profiles across the production chain This section details how the technology addresses specific pain points faced by procurement teams managing complex chemical supply networks for pharmaceutical intermediates
- Cost Reduction in Manufacturing: The one-step synthesis significantly reduces manufacturing costs by eliminating multiple reaction vessels purification stages and associated labor requirements compared to conventional multi-step routes The avoidance of expensive transition metal catalysts in subsequent purification steps further contributes to cost savings while the use of standard organic solvents minimizes waste disposal expenses This streamlined approach delivers substantial cost reductions through improved process efficiency without compromising product quality or yield consistency
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials with established global supply chains ensures consistent raw material availability regardless of geopolitical disruptions The simplified process design reduces dependency on specialized equipment or rare reagents making production more resilient to supply chain fluctuations This inherent robustness translates to more predictable lead times and reduced risk of production delays that could impact critical drug development timelines
- Scalability and Environmental Compliance: The methodology demonstrates excellent scalability from laboratory to commercial production scales without requiring significant process re-engineering as evidenced by consistent yields across different batch sizes The elimination of strong acids and hazardous byproducts substantially reduces environmental impact while simplifying waste treatment protocols This green chemistry approach aligns with increasingly stringent regulatory requirements while supporting corporate sustainability initiatives through reduced energy consumption and waste generation
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial concerns regarding implementation of this patented indanone synthesis methodology These responses are derived directly from the patent documentation and experimental data provided in CN112939780B
Q: How does this method improve functional group compatibility compared to traditional approaches?
A: The rhodium-catalyzed C-H activation mechanism operates under mild conditions without strong acids allowing tolerance of halogens alkyl groups and cyano substituents without protection-deprotection sequences that complicate conventional routes
Q: What are the scalability advantages of this one-step synthesis?
A: The process demonstrates consistent yields from laboratory scale to commercial production without requiring significant re-engineering as evidenced by successful scale-up experiments documented in patent examples
Q: How does this methodology address environmental compliance concerns?
A: By eliminating strong acids and multi-step sequences the process reduces hazardous waste generation while using standard organic solvents that simplify waste treatment protocols compared to traditional methods
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indanone Derivatives Supplier
Our patented technology represents a paradigm shift in indanone derivative manufacturing offering pharmaceutical companies unprecedented efficiency in producing these critical molecular building blocks NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through our state-of-the-art QC labs Our commitment to quality assurance ensures consistent delivery of high-purity indanone intermediates that meet or exceed pharmaceutical industry standards for complex molecule synthesis
We invite procurement teams to request a Customized Cost-Saving Analysis tailored to your specific production requirements Contact our technical procurement team today to obtain specific COA data and route feasibility assessments for your next-generation pharmaceutical development projects
