Advanced Rhodium-Catalyzed Synthesis of Indanone Derivatives for Scalable Pharmaceutical Manufacturing
Advanced Rhodium-Catalyzed Synthesis of Indanone Derivatives for Scalable Pharmaceutical Manufacturing
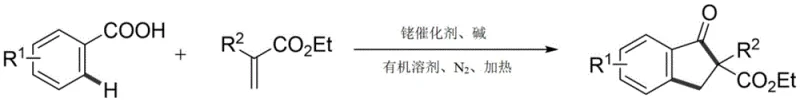
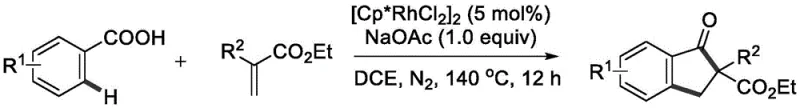
The landscape of organic synthesis for complex heterocyclic scaffolds is continually evolving, driven by the demand for more efficient and sustainable manufacturing processes. A significant breakthrough in this domain is detailed in patent CN112939780A, which discloses a novel synthetic method for indanone derivatives. This technology leverages transition metal-catalyzed C-H bond activation to achieve a one-step synthesis of 2-substituted indanone compounds directly from benzoic acid and acrylate precursors. For R&D directors and procurement specialists in the pharmaceutical sector, this represents a pivotal shift away from traditional, multi-step methodologies towards a more streamlined, atom-economical approach. The ability to construct the indanone core—a ubiquitous motif in bioactive natural products and drug molecules—via a direct coupling reaction offers substantial implications for reducing process mass intensity (PMI) and simplifying supply chain logistics for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the indanone framework has relied heavily on classical organic transformations such as the Friedel-Crafts acylation and the Nazarov cyclization. While these methods are well-established in academic literature, they present significant hurdles when translated to industrial-scale manufacturing. Friedel-Crafts reactions typically necessitate the use of stoichiometric amounts of strong Lewis acids or protic acids, which generate substantial quantities of corrosive waste and require rigorous quenching and neutralization protocols. Furthermore, these traditional routes often demand pre-functionalized substrates, meaning the starting materials must undergo additional synthetic steps to install leaving groups or activating motifs before the cyclization can occur. This not only increases the overall step count and production time but also cumulatively reduces the overall yield, making the process less economically viable for large-scale API intermediate production.
The Novel Approach
In stark contrast, the methodology outlined in CN112939780A utilizes a rhodium-catalyzed C-H activation strategy that fundamentally alters the synthetic logic. By employing a benzoic acid derivative as both a substrate and a directing group, the reaction achieves ortho-selective C-H activation followed by insertion of an acrylate coupling partner. This tandem process effectively merges the arylation and cyclization events into a single operational step. The use of a pentamethylcyclopentadienylrhodium dichloride catalyst system allows for mild reaction conditions compared to the harsh environments of classical chemistry. This novel approach not only simplifies the operational workflow by eliminating the need for substrate pre-functionalization but also significantly enhances the functional group compatibility, allowing for the incorporation of diverse substituents such as halogens and cyano groups which are often sensitive to traditional acidic conditions.
Mechanistic Insights into Rhodium-Catalyzed C-H Activation and Cyclization
The core of this technological advancement lies in the mechanistic efficiency of the Rh(III) catalytic cycle. The reaction initiates with the coordination of the carboxylic acid moiety of the benzoic acid substrate to the cationic rhodium species, forming a stable five-membered metallacycle intermediate. This coordination directs the metal center to the ortho-position of the aromatic ring, facilitating the cleavage of the C-H bond through a concerted metalation-deprotonation (CMD) pathway assisted by the acetate base. Following C-H activation, the coordinatively unsaturated rhodium species undergoes migratory insertion with the alkene double bond of the acrylate ester. This step is critical as it establishes the new carbon-carbon bond that forms the backbone of the five-membered ring. Subsequent intramolecular nucleophilic attack by the carbonyl oxygen or a related cyclization event, followed by protonolysis or reductive elimination, releases the final 2-substituted indanone product and regenerates the active catalyst species for the next turnover.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based or acid-catalyzed pathways. The directed nature of the C-H activation ensures high regioselectivity, minimizing the formation of meta- or para-substituted byproducts that are common in electrophilic aromatic substitutions. Furthermore, the mild basic conditions provided by sodium acetate prevent the degradation of acid-sensitive functional groups, such as the cyano and ester moieties found in the acrylate coupling partners. The patent data indicates that even with sterically demanding or electronically diverse substrates, the reaction maintains robust performance, yielding products like 5-bromo-2-cyano-7-methyl-1-oxo-2,3-dihydro-1H-indene-2-carboxylic acid ethyl ester in respectable yields. This level of precision is crucial for pharmaceutical applications where impurity profiles must be tightly controlled to meet regulatory standards.
How to Synthesize 2-Substituted Indanone Efficiently
The practical execution of this synthesis is designed for reproducibility and scalability, utilizing standard laboratory equipment that translates well to pilot and production scales. The process involves charging a reactor with the benzoic acid derivative, the acrylate coupling partner, the rhodium catalyst, and sodium acetate in 1,2-Dichloroethane (DCE). The mixture is then heated under an inert nitrogen atmosphere to ensure the exclusion of oxygen, which could potentially oxidize the catalyst or substrates. Detailed standardized operating procedures for temperature ramping, agitation speeds, and work-up protocols are essential for maximizing yield and consistency. For a comprehensive guide on the specific molar ratios, temperature profiles, and purification techniques validated in the patent examples, please refer to the structured synthesis guide below.
- Combine benzoic acid compound, acrylate compound, [Cp*RhCl2]2 catalyst, and sodium acetate base in 1,2-Dichloroethane (DCE) solvent under nitrogen atmosphere.
- Heat the reaction mixture to 140 °C and maintain stirring for 12 hours to facilitate the C-H activation and cyclization process.
- Upon completion, cool the mixture, perform suction filtration, mix with silica gel, and purify via column chromatography to isolate the target indanone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this rhodium-catalyzed technology translates into tangible strategic benefits beyond mere chemical elegance. The primary value proposition lies in the drastic simplification of the supply chain for raw materials. By enabling the use of simple, commercially available benzoic acids and acrylates, the method bypasses the need for expensive, custom-synthesized pre-functionalized intermediates. This reduction in upstream complexity directly correlates to enhanced supply security and reduced lead times, as the reliance on niche specialty chemical suppliers is minimized. Furthermore, the high atom economy of the reaction means that a greater proportion of the input mass is converted into the desired product, inherently lowering the cost of goods sold (COGS) by reducing waste disposal fees and raw material consumption.
- Cost Reduction in Manufacturing: The elimination of stoichiometric Lewis acids and the reduction in synthetic steps significantly lower the operational expenditure associated with reagent procurement and waste treatment. Traditional methods often require expensive chlorinating agents or acyl chlorides, whereas this method utilizes stable carboxylic acids. Additionally, the high reaction yields reported (ranging from 47% to 78% across diverse substrates) ensure that material throughput is maximized, reducing the cost per kilogram of the final API intermediate. The ability to tolerate diverse functional groups also means that downstream protection and de-protection steps can often be avoided, further streamlining the cost structure.
- Enhanced Supply Chain Reliability: The robustness of the catalytic system against various substituents ensures that supply disruptions for specific substituted precursors do not halt production. Since the reaction tolerates halogens like fluorine, chlorine, and bromine, as well as alkyl groups, manufacturers have flexibility in sourcing starting materials. This flexibility is critical for maintaining business continuity in the face of global raw material volatility. The use of a defined catalytic cycle also allows for better predictability in batch-to-batch consistency, a key metric for supply chain reliability in regulated industries.
- Scalability and Environmental Compliance: The reaction conditions, involving heating to 140 °C in DCE, are amenable to scale-up in standard stainless steel reactors without requiring exotic high-pressure equipment. The use of nitrogen as the only atmospheric requirement simplifies the engineering controls needed for the plant. Moreover, the improved atom economy aligns with green chemistry principles, reducing the environmental footprint of the manufacturing process. This is increasingly important for meeting corporate sustainability goals and complying with tightening environmental regulations regarding chemical waste discharge.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic route. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for evaluating the technology's fit within your existing manufacturing portfolio. Understanding these nuances is vital for making informed decisions about process adoption and vendor qualification.
Q: What are the key advantages of this Rhodium-catalyzed method over traditional Friedel-Crafts acylation?
A: Unlike traditional Friedel-Crafts reactions which require harsh Lewis acids and pre-functionalized substrates, this method utilizes direct C-H bond activation. This results in superior atom economy, milder reaction conditions, and eliminates the need for cumbersome substrate pre-functionalization steps.
Q: What is the substrate scope regarding functional group compatibility?
A: The process demonstrates excellent functional group tolerance, successfully accommodating alkyl groups (methyl), halogens (fluorine, chlorine, bromine), and various electron-withdrawing groups like cyano and ethoxycarbonyl moieties without significant yield degradation.
Q: Can this method produce unsubstituted indanone compounds?
A: Yes, the patent describes a subsequent decarboxylation step. By heating the 2-substituted indanone intermediate with an inorganic base in an alcohol solvent like HFIP at 150 °C, the unsubstituted indanone skeleton can be efficiently obtained.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indanone Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced C-H activation technologies in modern drug discovery and development. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN112939780A can be seamlessly transitioned to industrial reality. Our state-of-the-art facilities are equipped to handle transition metal catalysis with stringent purity specifications, supported by rigorous QC labs that utilize advanced analytical techniques to guarantee the quality of every batch. We are committed to delivering high-purity indanone derivatives that meet the exacting standards of the global pharmaceutical industry.
We invite you to collaborate with us to leverage this cutting-edge synthesis for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. Please contact our technical procurement team today to request specific COA data for our indanone catalog and to discuss route feasibility assessments for your custom synthesis needs. Let us help you optimize your supply chain with reliable, cost-effective, and high-quality chemical solutions.
