Advanced Montmorillonite-Catalyzed Dehydration for High-Purity Trifluoromethyl Styrene Manufacturing
Advanced Montmorillonite-Catalyzed Dehydration for High-Purity Trifluoromethyl Styrene Manufacturing
The global demand for fluorinated polymers continues to surge, driven by applications in high-performance coatings, electronics, and advanced materials. At the heart of this supply chain lies the critical need for reliable monomer synthesis that balances yield, purity, and environmental compliance. A pivotal advancement in this domain is detailed in Chinese Patent CN101781162B, which discloses a novel method for preparing trifluoromethyl styrene compounds. This technology represents a significant departure from traditional harsh acidic dehydration processes, utilizing montmorillonite clay as a heterogeneous catalyst to achieve superior conversion rates. For R&D directors and procurement strategists, understanding this shift is essential for securing a stable supply of high-purity polymer intermediates. The patent outlines a robust pathway that not only enhances product quality but also drastically simplifies the downstream purification workflow, addressing long-standing pain points in the industrial production of fluorinated styrenics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl styrene has been plagued by significant technical and operational challenges that hinder scalable manufacturing. Traditional literature methods, such as those reported by G.B. Bachman and C.S. Marvel, rely heavily on aggressive dehydrating agents like potassium bisulfate (KHSO4) or phosphorus pentoxide (P2O5) in toxic solvents like benzene. These legacy processes operate under extreme thermal conditions, often exceeding 170°C, which inadvertently triggers the self-polymerization of the sensitive styrene product. This side reaction not only depresses yields to unacceptable levels, often hovering around 50% for P2O5 methods, but also generates complex impurity profiles that are difficult to separate. Furthermore, the generation of strong acidic by-products, such as phosphoric acid or acidic sulfate slurries, imposes severe corrosion risks on standard stainless steel reactors, necessitating expensive Hastelloy linings and rigorous waste neutralization protocols that inflate operational expenditures.
The Novel Approach
In stark contrast, the methodology described in CN101781162B introduces a paradigm shift by employing montmorillonite K10 or KSF as a solid acid catalyst. This approach effectively mitigates the corrosive nature of liquid mineral acids while maintaining the necessary acidity to drive the dehydration equilibrium forward. The reaction can be conducted under solvent-free conditions or in benign solvents like toluene or n-octane at moderate temperatures ranging from 100°C to 180°C. By eliminating the use of benzene and avoiding the formation of liquid acidic waste streams, this novel route significantly improves the safety profile of the manufacturing environment. The structural transformation from the alcohol precursor (Compound I) to the styrene derivative (Compound II) is achieved with remarkable efficiency, preserving the integrity of the trifluoromethyl group and preventing the oligomerization that plagues conventional acidic routes.
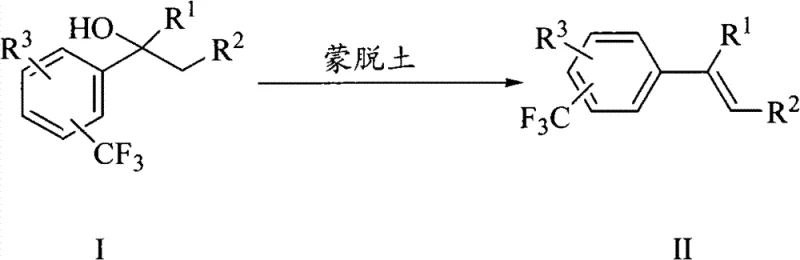
Mechanistic Insights into Montmorillonite-Catalyzed Dehydration
The efficacy of montmorillonite in this transformation stems from its unique layered silicate structure, which provides both Brønsted and Lewis acid sites on its surface. Unlike homogeneous acids that freely diffuse and aggressively protonate the alkene product leading to cationic polymerization, the constrained acid sites on the clay surface facilitate a controlled elimination of water from the benzylic alcohol. This heterogeneous mechanism ensures that once the trifluoromethyl styrene is formed, it desorbs from the catalyst surface rapidly, minimizing residence time in the acidic environment and thus suppressing self-polymerization. For process chemists, this means a cleaner reaction profile with fewer high-molecular-weight tarry by-products, simplifying the final distillation step. The ability to tune the catalyst loading between 5% and 30% by weight allows for precise optimization of the reaction kinetics, ensuring complete conversion of the starting material without over-exposure to thermal stress.
Furthermore, the impurity control mechanism is intrinsically linked to the absence of strong mineral acid residues. In traditional P2O5 methods, the resulting phosphoric acid can catalyze further degradation or rearrangement of the product during workup. With montmorillonite, the catalyst remains a solid that can be filtered off or left in the pot prior to distillation, leaving the crude product largely free of inorganic contaminants. This results in a final product purity consistently exceeding 99.0% as verified by gas chromatography, a critical specification for polymerization-grade monomers where trace impurities can act as chain transfer agents or terminators. The robustness of this catalytic system across various substrates, including chloro- and fluoro-substituted derivatives, demonstrates its versatility for generating a diverse library of functionalized styrenic monomers for specialized material applications.
How to Synthesize Trifluoromethyl Styrene Efficiently
Implementing this synthesis route requires careful attention to catalyst activation and temperature profiling to maximize the benefits of the solid-state dehydration. The process is designed to be operationally simple, removing the need for complex anhydrous solvent handling or inert atmosphere gloveboxes typically associated with sensitive organometallic chemistry. Operators can charge the reactor with the trifluoromethyl phenethyl alcohol and the montmorillonite catalyst directly, initiating the reaction upon heating. The flexibility to run the process neat (solvent-free) offers a distinct advantage in volumetric productivity, allowing manufacturers to produce larger batches in existing reactor volumes without being limited by solvent fill levels. Detailed standardized synthetic steps for implementing this protocol in a GMP-compliant environment are outlined below.
- Charge the reactor with trifluoromethyl phenethyl alcohol (Compound I) and montmorillonite K10 or KSF catalyst (5-30% w/w).
- Heat the mixture to 100-180°C under stirring for 4-10 hours to facilitate dehydration without strong liquid acids.
- Upon completion, perform vacuum distillation to isolate the pure trifluoromethyl styrene monomer (Compound II) with >99% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this montmorillonite-based technology translates into tangible reductions in total cost of ownership and risk mitigation. The elimination of toxic benzene solvents removes a major regulatory burden and reduces the costs associated with solvent procurement, storage, and VOC emission controls. Moreover, the significant reduction in equipment corrosion extends the lifecycle of reactor vessels and piping, deferring capital expenditure on specialized corrosion-resistant alloys. The ability to recycle the montmorillonite catalyst after simple drying further contributes to raw material cost optimization, creating a more sustainable and economically resilient supply chain for fluorinated intermediates.
- Cost Reduction in Manufacturing: The transition from stoichiometric dehydrating agents like phosphorus pentoxide to a catalytic amount of montmorillonite fundamentally alters the cost structure of the synthesis. By avoiding the purchase of expensive and hazardous dehydrating reagents and eliminating the downstream costs of neutralizing large volumes of acidic waste, the overall variable cost per kilogram is substantially lowered. Additionally, the solvent-free option removes the energy-intensive step of solvent recovery distillation, leading to significant utility savings in steam and cooling water consumption. These cumulative efficiencies allow for a more competitive pricing model for high-purity trifluoromethyl styrene derivatives in the global market.
- Enhanced Supply Chain Reliability: Reliance on hazardous chemicals like benzene and phosphorus pentoxide often introduces supply volatility due to strict transportation regulations and environmental restrictions. By shifting to a process that utilizes commercially available clays and optional green solvents like toluene or n-octane, the supply chain becomes more robust and less susceptible to regulatory disruptions. The simplicity of the workup procedure, requiring only filtration and distillation, also reduces the turnaround time between batches, enabling manufacturers to respond more agilely to fluctuations in downstream polymer demand without compromising on delivery schedules.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies waste management challenges, but this technology inherently minimizes the environmental footprint. The absence of acidic aqueous waste streams simplifies wastewater treatment requirements, ensuring easier compliance with increasingly stringent environmental protection laws. The solid nature of the catalyst facilitates safe handling and disposal or regeneration, reducing the liability associated with hazardous chemical storage. This green chemistry profile not only aligns with corporate sustainability goals but also future-proofs the manufacturing site against potential bans on volatile organic compounds and corrosive reagents, ensuring long-term operational continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this dehydration technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for evaluating the feasibility of this route for your specific application needs. Understanding these nuances is critical for making informed decisions about process adoption and vendor qualification.
Q: Why is montmorillonite preferred over phosphorus pentoxide for this dehydration?
A: Montmorillonite acts as a solid acid catalyst that minimizes equipment corrosion and avoids the generation of hazardous phosphoric acid waste, unlike phosphorus pentoxide which creates severe corrosion and environmental disposal issues.
Q: Can this process be operated without organic solvents?
A: Yes, the patented method explicitly supports solvent-free conditions, which eliminates the need for toxic solvents like benzene and removes the energy-intensive solvent recovery distillation step.
Q: What purity levels are achievable with this catalytic route?
A: Experimental data indicates that gas chromatography (GC) purity can consistently exceed 99.0%, with yields ranging from 91% to 96%, significantly outperforming traditional acidic dehydration methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl Styrene Supplier
As the industry moves towards greener and more efficient manufacturing paradigms, having a partner with deep technical expertise in fluorinated chemistry is indispensable. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to full-scale manufacturing is seamless. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of trifluoromethyl styrene meets the exacting standards required for high-performance polymer synthesis. We understand the critical nature of monomer purity in determining the final properties of fluoropolymers and are committed to delivering consistency.
We invite you to engage with our technical procurement team to discuss how this advanced catalytic route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this montmorillonite-catalyzed process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your volume requirements, ensuring a secure and cost-effective supply of these vital specialty chemical intermediates.
