Advanced Synthesis of Ultra-Short Acting Benzylisoquinoline Neuromuscular Blockers for Clinical Anesthesia
Advanced Synthesis of Ultra-Short Acting Benzylisoquinoline Neuromuscular Blockers for Clinical Anesthesia
The landscape of modern surgical anesthesia is undergoing a significant transformation driven by the demand for safer, more predictable neuromuscular blocking agents. Patent CN111662230A discloses a groundbreaking class of benzyl isoquinoline compounds designed to address the critical limitations of current muscle relaxants. These novel molecules function as non-depolarizing neuromuscular junction blockers with an ultra-short duration of action, offering a pharmacological profile that rivals or exceeds the onset speed of succinylcholine while eliminating its dangerous side effect profile. The core innovation lies in the strategic placement of metabolically labile linkers within the molecular scaffold, facilitating rapid clearance from the body without the need for pharmacological reversal agents. This technological leap represents a major opportunity for pharmaceutical manufacturers seeking to develop next-generation anesthetics with improved patient safety margins.
In the context of clinical anesthesia, the management of skeletal muscle relaxation is paramount for procedures requiring tracheal intubation and abdominal access. Historically, succinylcholine has been the gold standard for rapid sequence induction due to its unparalleled onset speed. However, its mechanism of action as a depolarizing agent triggers widespread agonism of acetylcholine receptors, leading to potentially life-threatening complications such as hyperkalemia, malignant hyperthermia, and cardiac arrhythmias. Conversely, existing non-depolarizing agents like aminosteroids and traditional benzylisoquinolines avoid these specific receptor-mediated toxicities but suffer from prolonged durations of action. This extended activity often results in postoperative residual curarization, a serious risk factor for respiratory compromise in the recovery unit. The compounds described in this patent bridge this therapeutic gap by combining the safety of non-depolarizing blockade with the rapid offset kinetics previously exclusive to succinylcholine.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to muscle relaxation rely heavily on two distinct chemical classes, each with inherent drawbacks that complicate perioperative management. Depolarizing agents, while fast-acting, induce a sustained depolarization of the motor endplate that can cause fasciculations and release intracellular potassium, posing severe risks to patients with burns, trauma, or neuromuscular diseases. On the other hand, conventional non-depolarizing agents, such as rocuronium or cisatracurium, provide excellent safety regarding receptor specificity but lack the ultra-short half-life required for brief procedures or rapid awakening. The persistence of these drugs in the plasma often necessitates the administration of cholinesterase inhibitors like neostigmine to accelerate recovery. However, this reversal strategy is imperfect; it requires the patient to have already recovered some degree of spontaneous ventilation to be effective and can itself induce cholinergic side effects like bradycardia and bronchospasm. Furthermore, the reliance on exogenous reversal agents adds complexity and cost to the anesthetic protocol, creating a bottleneck in high-turnover surgical environments.
The Novel Approach
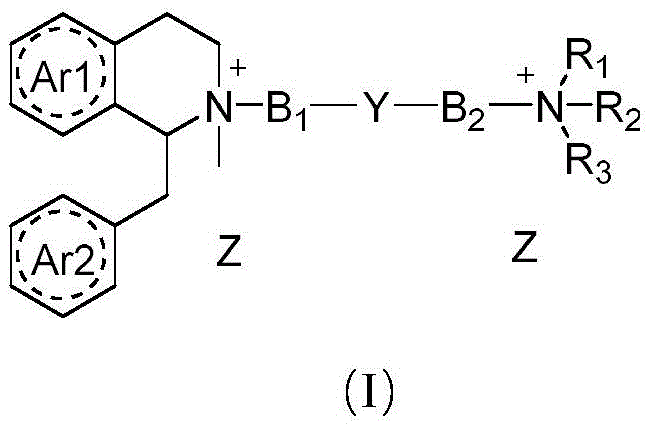
The synthetic strategy outlined in the patent introduces a modular design where the pharmacophore is interrupted by a chemically defined linker group, denoted as Y in Formula I. This linker is specifically engineered to be susceptible to hydrolysis by ubiquitous plasma esterases and hepatic enzymes. By incorporating carbonate or ester functionalities into the backbone connecting the two quaternary ammonium heads, the molecule becomes a "soft drug" that self-destructs rapidly upon entering the systemic circulation. This design ensures that once the infusion is stopped or the single dose is metabolized, the active drug concentration plummets, allowing for spontaneous and rapid return of muscle function. This approach effectively decouples the onset of action from the duration of action, enabling clinicians to achieve profound muscle relaxation quickly while guaranteeing a swift recovery profile that minimizes the risk of residual paralysis and eliminates the dependency on rescue medications.
Mechanistic Insights into Enzymatic Hydrolysis and Quaternary Ammonium Stability
The chemical architecture of these benzylisoquinoline derivatives is predicated on the stability of the tetrahydroisoquinoline ring system coupled with the lability of the central linker. The synthesis typically begins with the construction of the isoquinoline core via a Bischler-Napieralski type cyclization or similar condensation reactions. As illustrated in the reaction schemes, the formation of the quaternary ammonium centers is achieved through exhaustive alkylation of the tertiary amine precursors. The critical mechanistic feature is the introduction of the linker Y, which acts as a metabolic switch. In physiological conditions, nucleophilic attack by water or serine residues in esterase active sites targets the carbonyl carbon of the linker. This hydrolytic cleavage severs the connection between the two pharmacophoric heads, rendering the resulting fragments inactive at the nicotinic acetylcholine receptor. This mechanism mimics the metabolism of natural neurotransmitters but is accelerated by the specific electronic properties of the synthetic linker.
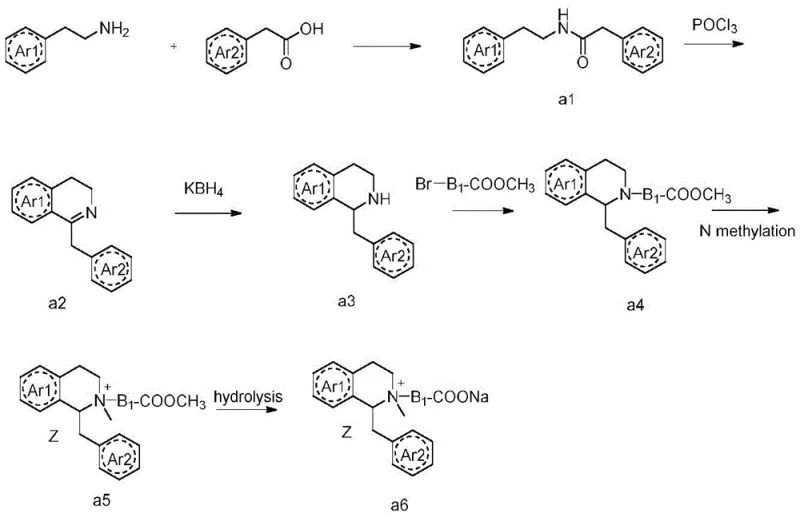
Furthermore, the stereochemistry of the benzylisoquinoline core plays a pivotal role in receptor binding affinity and metabolic rate. The patent details methods for resolving stereoisomers using chiral separating reagents such as tartaric acid derivatives, ensuring that the final pharmaceutical product consists of the most potent enantiomer. This chiral purity is essential for minimizing the dose required to achieve neuromuscular blockade, which in turn reduces the metabolic load on the patient. The synthetic pathway involves precise control over reaction conditions, such as the use of phosphorus oxychloride for dehydration and potassium borohydride for selective reduction, to maintain the integrity of the sensitive functional groups while establishing the necessary quaternary nitrogen centers. The robustness of this chemistry allows for the introduction of diverse substituents on the aromatic rings, enabling fine-tuning of the drug's lipophilicity and receptor interaction profile.
How to Synthesize Benzyl Isoquinoline Muscle Relaxant Intermediates Efficiently
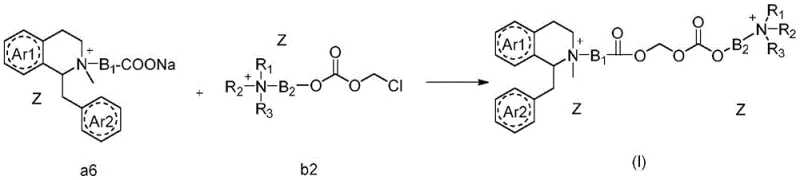
The production of these high-value pharmaceutical intermediates follows a convergent synthesis strategy that maximizes yield and purity while minimizing waste. The process is divided into the preparation of two key fragments: the isoquinoline-containing carboxylate (Fragment A) and the linker-bearing quaternary ammonium salt (Fragment B). These fragments are synthesized independently using robust, scalable organic transformations before being coupled in the final step. This modular approach allows for rigorous quality control of each building block prior to assembly, ensuring that the final active pharmaceutical ingredient meets stringent regulatory standards. The detailed standardized synthesis steps for this complex pathway are provided in the guide below.
- Condense aromatic ethylamine with aromatic acetic acid to form an amide, followed by cyclization using phosphorus oxychloride to generate the dihydroisoquinoline core.
- Reduce the imine intermediate using a reducing agent like potassium borohydride to obtain the tetrahydroisoquinoline secondary amine.
- Perform N-alkylation with bromo-esters to introduce the linker chain, followed by quaternization with methyl iodide or benzyl bromides to form the final bis-quaternary ammonium salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers compelling economic and logistical benefits derived from its chemical efficiency. The methodology relies on commodity chemicals and established reaction types, avoiding the need for scarce or prohibitively expensive transition metal catalysts often found in cross-coupling reactions. This fundamental simplicity translates directly into a more resilient supply chain, as the raw materials are sourced from stable, high-volume chemical markets rather than specialized niche suppliers. Additionally, the avoidance of heavy metal catalysts eliminates the costly and time-consuming downstream processing steps required to reduce metal residues to ppm levels, a critical requirement for parenteral drugs. This streamlined purification process not only reduces manufacturing costs but also shortens the overall production cycle time.
- Cost Reduction in Manufacturing: The synthetic pathway utilizes straightforward alkylation and esterification reactions that can be performed in standard stainless steel reactors without the need for specialized high-pressure or cryogenic equipment. By eliminating the dependence on precious metal catalysts such as palladium or platinum, the direct material costs are significantly lowered. Furthermore, the high selectivity of the reactions minimizes the formation of difficult-to-remove impurities, thereby increasing the overall yield and reducing the solvent consumption associated with extensive chromatographic purification. These factors combine to create a highly cost-effective manufacturing process suitable for large-scale commercial production.
- Enhanced Supply Chain Reliability: The starting materials, including substituted phenethylamines and benzyl bromides, are widely produced fine chemicals with multiple global suppliers. This diversity in the supply base mitigates the risk of shortages that can plague processes dependent on single-source proprietary reagents. The robustness of the chemistry also means that the process is less sensitive to minor variations in raw material quality, ensuring consistent batch-to-batch performance. For supply chain planners, this reliability translates into predictable lead times and the ability to maintain strategic stockpiles of key intermediates without fear of rapid degradation or obsolescence.
- Scalability and Environmental Compliance: The reaction conditions described are inherently scalable, moving seamlessly from laboratory gram-scale to multi-ton industrial production. The solvents employed, such as acetonitrile and dichloromethane, are standard industrial solvents with well-established recovery and recycling protocols, aligning with modern green chemistry initiatives. The absence of toxic heavy metals simplifies waste stream management, reducing the environmental footprint and the associated costs of hazardous waste disposal. This environmental compatibility facilitates smoother regulatory approvals and supports the sustainability goals of modern pharmaceutical enterprises.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these novel neuromuscular blocking agents. The answers are derived directly from the experimental data and structural analysis provided in the patent documentation, offering clarity on the compound's stability, synthesis, and therapeutic potential. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this technology into their product pipelines.
Q: What is the primary advantage of these benzylisoquinoline compounds over succinylcholine?
A: Unlike succinylcholine, which is a depolarizing agent associated with severe side effects like hyperkalemia and malignant hyperthermia, these novel benzylisoquinoline compounds are non-depolarizing. Furthermore, they possess an ultra-short duration of action due to rapid enzymatic hydrolysis, eliminating the need for reversal agents and reducing the risk of postoperative residual paralysis.
Q: How is the ultra-short acting effect achieved chemically?
A: The molecular structure incorporates specific linker groups (Y), such as carbonate or ester bonds, positioned between the two quaternary ammonium heads. These linkers are highly sensitive to hydrolysis by plasma esterases and liver enzymes, leading to rapid metabolic cleavage and immediate loss of neuromuscular blocking activity.
Q: Are the starting materials for this synthesis commercially available?
A: Yes, the synthesis relies on readily available fine chemical building blocks such as substituted phenethylamines, phenylacetic acids, and various benzyl bromides. The process avoids exotic catalysts, utilizing standard reagents like phosphorus oxychloride and common alkylating agents, which supports scalable manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzyl Isoquinoline Supplier
As the demand for safer anesthetic agents grows, the ability to manufacture complex benzyl isoquinoline intermediates at scale becomes a critical competitive advantage. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the precise temperature controls and moisture-sensitive reactions required for synthesizing these quaternary ammonium salts. We adhere to stringent purity specifications and operate rigorous QC labs to ensure that every batch of intermediate meets the exacting standards required for clinical trial material and eventual commercial API production.
We invite pharmaceutical partners to collaborate with us on optimizing the supply chain for these next-generation muscle relaxants. Our technical team is prepared to conduct a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a secure, scalable, and cost-efficient source of high-purity pharmaceutical intermediates that can accelerate your development timelines and enhance your market competitiveness.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
