Advanced Synthesis of Ticagrelor Five-Membered Ring Intermediate for Commercial Scale-up
Advanced Synthesis of Ticagrelor Five-Membered Ring Intermediate for Commercial Scale-up
The pharmaceutical industry continuously seeks robust and efficient synthetic pathways for critical cardiovascular medications, particularly for antiplatelet agents like Ticagrelor. A recent technological breakthrough documented in patent CN115785058A introduces a streamlined method for synthesizing the essential five-membered ring intermediate, known chemically as Compound 5. This intermediate serves as a pivotal building block in the assembly of Ticagrelor, a drug widely prescribed for acute coronary syndrome. The disclosed methodology represents a significant departure from traditional multi-step sequences, offering a route that is not only shorter but also demonstrates superior atom economy. For research and development teams focused on process optimization, this patent provides a compelling alternative that addresses long-standing challenges regarding waste generation and reaction safety. By leveraging direct ring-closing strategies with ethylene oxide, the process minimizes the environmental footprint while maintaining high stereochemical integrity, positioning it as a viable candidate for next-generation manufacturing protocols.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of this specific cyclopentane derivative has relied on a more convoluted synthetic trajectory that introduces unnecessary complexity and hazard into the production line. As illustrated in the prior art, the conventional route typically involves the alkylation of a hydroxyl group using ethyl bromoacetate under strongly alkaline conditions, followed by a reduction step utilizing sodium borohydride to generate the necessary alcohol functionality. This traditional approach suffers from several critical drawbacks that impact both operational safety and economic efficiency. The reduction phase, in particular, is problematic as it generates substantial volumes of hydrogen gas, creating a high-risk environment that requires specialized containment and ventilation systems. Furthermore, the reliance on strong alkalis and anhydrous solvents necessitates rigorous moisture control and complicates the downstream workup procedures. The accumulation of inorganic salts and organic byproducts results in a significant burden of wastewater and waste liquid, driving up disposal costs and complicating regulatory compliance for environmental discharge.
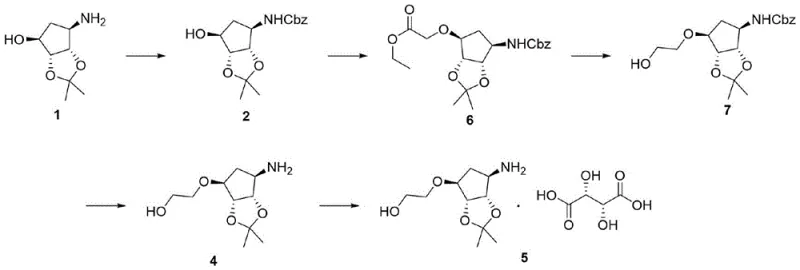
The Novel Approach
In stark contrast to the legacy methods, the innovative strategy outlined in the patent data simplifies the molecular construction by directly engaging ethylene oxide in a ring-closing reaction. This novel pathway bypasses the hazardous reduction step entirely, replacing it with a cleaner nucleophilic substitution that builds the side chain in a single operation. The overall sequence proceeds through four distinct stages: amino protection, ring closure, deprotection, and salt formation. By eliminating the intermediate ester formation and subsequent reduction, the new route drastically shortens the synthetic timeline and reduces the number of unit operations required. This consolidation of steps leads to a marked improvement in overall yield and throughput. The use of ethylene oxide, a readily available commodity chemical, allows for a more predictable supply chain and reduces dependency on specialized reagents. Consequently, this approach offers a more sustainable and economically attractive solution for the large-scale production of high-purity pharmaceutical intermediates.
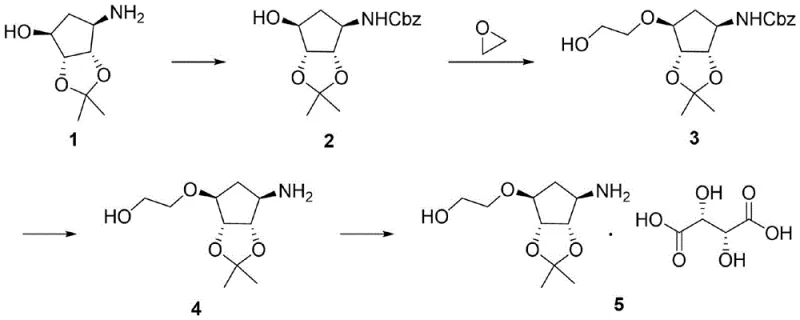
Mechanistic Insights into Ethylene Oxide Ring Closing
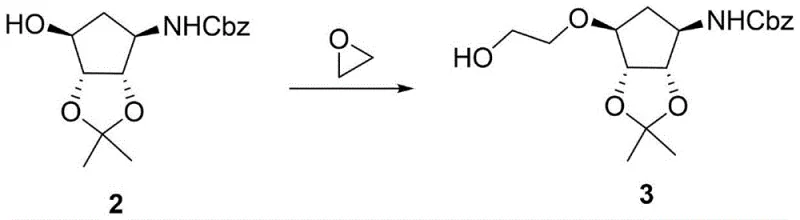
The core innovation of this synthesis lies in the second step, where the protected amine intermediate undergoes a ring-opening reaction with ethylene oxide to install the hydroxyethyl ether side chain. This transformation is conducted in a sealed autoclave using solvents such as tetrahydrofuran or toluene at elevated temperatures ranging from 50 to 150 degrees Celsius. The mechanism involves the nucleophilic attack of the cyclopentyl hydroxyl group on the strained epoxide ring of ethylene oxide, driven by thermal energy. This direct alkylation is highly efficient because it avoids the formation of leaving groups associated with halogenated reagents, thereby reducing halogenated waste streams. The reaction conditions are carefully optimized to ensure complete conversion while minimizing polymerization of the ethylene oxide. Following this, the benzyl carbamate protecting group is removed via catalytic hydrogenation using palladium on carbon. This deprotection step is mild and selective, preserving the acetonide protection on the diol system while cleanly exposing the primary amine required for the final salt formation. The stereochemistry of the cyclopentane ring is maintained throughout these transformations, ensuring that the final product possesses the correct spatial configuration necessary for biological activity.
Impurity control is another critical aspect where this mechanistic approach excels. In conventional routes, over-alkylation or incomplete reduction can lead to difficult-to-remove impurities that compromise the purity profile of the final API. The new method mitigates these risks by utilizing stoichiometric control of ethylene oxide, typically between 0.8 to 1.5 equivalents, to prevent poly-ether formation. The subsequent crystallization of the L-tartaric acid salt serves as a powerful purification tool, leveraging the specific solubility differences between the desired enantiomer and potential impurities. The use of an ethanol-ethyl acetate solvent system during salt formation further enhances the rejection of non-polar byproducts. This rigorous control over the chemical landscape ensures that the final intermediate meets the stringent purity specifications demanded by global regulatory bodies, reducing the need for extensive chromatographic purification which is often cost-prohibitive at scale.
How to Synthesize Ticagrelor Intermediate Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize yield and safety. The process begins with the protection of the starting amine using benzyl chloroformate in a biphasic methanol-water system, followed by the critical high-pressure reaction with ethylene oxide. Operators must adhere to strict temperature profiles during the exothermic protection step and maintain secure sealing during the high-temperature ring closure. The final deprotection and salification steps are performed under ambient pressure but require careful monitoring of hydrogen uptake and crystallization kinetics. For a comprehensive breakdown of the standardized operating procedures, including specific reagent grades and equipment specifications, please refer to the detailed guide below.
- Perform amino protection on compound 1 using benzyl chloroformate in a methanol-water solution with sodium carbonate to obtain compound 2.
- Execute a ring closing reaction by reacting compound 2 with ethylene oxide in tetrahydrofuran or toluene at elevated temperatures to form compound 3.
- Conduct a hydrogen deprotection reaction on compound 3 using a palladium-carbon catalyst in methanol to yield compound 4.
- Complete the synthesis by reacting compound 4 with L-tartaric acid in an ethanol-ethyl acetate mixture to form the final salt, compound 5.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this novel synthetic route offers transformative benefits that extend beyond simple chemical efficiency. The elimination of hazardous reduction steps and the simplification of the workflow directly translate into lower operational expenditures and reduced capital investment in safety infrastructure. By removing the need for sodium borohydride and the associated quenching processes, manufacturers can significantly reduce the volume of hazardous waste requiring treatment. This shift not only lowers disposal costs but also aligns with increasingly strict environmental regulations regarding chemical manufacturing. Furthermore, the reliance on ethylene oxide, a bulk chemical with a stable global supply, mitigates the risk of raw material shortages that often plague specialty reagent markets. This stability ensures consistent production schedules and reliable delivery timelines for downstream API manufacturers.
- Cost Reduction in Manufacturing: The streamlined nature of this process inherently drives down manufacturing costs by reducing the total number of processing steps and solvent exchanges. Eliminating the esterification and reduction sequence removes the need for expensive reducing agents and the energy-intensive conditions they often require. Additionally, the simplified workup procedures reduce labor hours and utility consumption per kilogram of product. The higher atom economy means that a greater proportion of raw materials end up in the final product rather than as waste, optimizing material utilization rates. These cumulative efficiencies result in a substantially lower cost of goods sold, providing a competitive edge in the pricing of the final pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply chain resilience by minimizing dependency on complex, multi-vendor reagent chains. The use of common solvents like methanol, toluene, and ethyl acetate ensures that production is not halted due to the unavailability of niche chemicals. The improved safety profile reduces the likelihood of unplanned shutdowns caused by safety incidents or regulatory inspections related to hazardous operations. This reliability allows supply chain managers to forecast inventory levels with greater confidence and maintain leaner stock buffers without risking production continuity. Ultimately, this leads to shorter lead times and a more responsive supply capability to meet fluctuating market demands for cardiovascular medications.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the use of standard reactor types and the absence of extreme cryogenic or high-vacuum requirements. The reduction in wastewater generation simplifies effluent treatment plant loading, making it easier for facilities to remain compliant with local environmental discharge limits. The avoidance of heavy metal catalysts or toxic reagents further simplifies the validation process for GMP manufacturing. This environmental compatibility not only reduces the ecological footprint but also future-proofs the manufacturing site against tightening green chemistry regulations, ensuring long-term operational viability and sustainability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on yield expectations, safety protocols, and scalability factors. Understanding these details is crucial for technical teams evaluating the feasibility of technology transfer and for procurement officers assessing the long-term value proposition of this manufacturing route.
Q: How does this new synthesis route improve safety compared to conventional methods?
A: The novel method eliminates the use of sodium borohydride reduction, which traditionally generates large amounts of hydrogen gas, thereby significantly reducing explosion risks and enhancing operational safety in manufacturing facilities.
Q: What are the purity specifications achievable with this ethylene oxide route?
A: Experimental data indicates that the final compound 5 can achieve purity levels exceeding 99% after recrystallization, meeting the stringent quality standards required for active pharmaceutical ingredient (API) production.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes common solvents like methanol and tetrahydrofuran and avoids complex post-reaction treatments, making it highly scalable and cost-effective for commercial manufacturing of ticagrelor intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ticagrelor Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and safe synthetic routes in the modern pharmaceutical landscape. Our team of expert chemists has extensively analyzed the potential of this ethylene oxide-based methodology and is well-equipped to facilitate its transition from patent concept to commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this process are fully realized in a practical manufacturing setting. Our state-of-the-art facilities are designed to handle high-pressure reactions and sensitive hydrogenation steps with the utmost safety and precision. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of intermediate meets the exacting standards required for API synthesis.
We invite pharmaceutical partners to collaborate with us to leverage this advanced technology for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to determine how this optimized synthesis can enhance your production efficiency and reduce overall costs. Let us help you secure a reliable supply of high-quality ticagrelor intermediates while advancing your sustainability goals.
