Scalable Synthesis of Novel Cephem Antibacterial Compounds for Commercial API Production
Scalable Synthesis of Novel Cephem Antibacterial Compounds for Commercial API Production
The global demand for effective antibacterial agents continues to drive innovation in pharmaceutical intermediate synthesis, particularly for compounds capable of overcoming bacterial resistance mechanisms. Patent CN1030657C discloses a significant advancement in this field by introducing novel cephem compounds characterized by a quaternary ammonium methyl group at the 3-position and a specific aminothiazole oxime moiety at the 7-position. These structural features are critical for enhancing antimicrobial activity, especially against resistant Gram-negative bacteria such as Pseudomonas aeruginosa. The technology outlined in this patent provides a robust framework for producing high-purity intermediates that serve as the foundation for next-generation cephalosporin antibiotics. By leveraging condensed imidazole rings at the 3-position, these compounds offer superior stability and pharmacokinetic profiles compared to traditional monocyclic derivatives. This report analyzes the technical merits and commercial viability of this synthesis route for industry stakeholders.
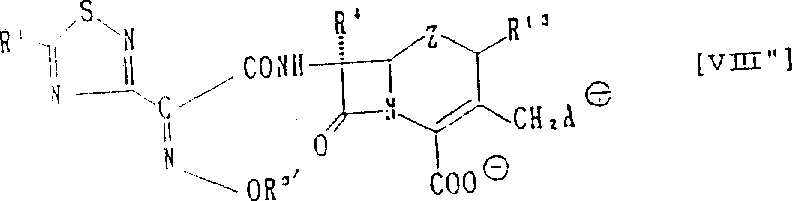
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for third-generation cephalosporins often rely on monocyclic pyridyl groups at the 3-position, which can limit the antimicrobial spectrum and stability of the final drug product. Many existing methods struggle to effectively combat beta-lactamase producing bacteria, leading to reduced efficacy in clinical settings where resistance is prevalent. Furthermore, conventional quaternization reactions may suffer from low yields or require harsh conditions that compromise the integrity of the sensitive beta-lactam ring. The purification of these intermediates can also be challenging, often necessitating complex chromatographic steps that increase production costs and lead times. Additionally, the solubility and bioavailability of compounds derived from older methodologies may not meet the rigorous standards required for modern parenteral formulations. These limitations highlight the urgent need for improved synthetic strategies that can deliver compounds with enhanced therapeutic indices.
The Novel Approach
The innovative process described in the patent overcomes these challenges by utilizing condensed imidazole rings, such as imidazo[1,2-a]pyridine or imidazo[1,2-b]pyridazine, to form the quaternary ammonium group. This structural modification significantly broadens the antimicrobial spectrum, providing potent activity against both Gram-positive and Gram-negative pathogens, including resistant strains. The synthesis employs mild reaction conditions that preserve the stereochemistry and stability of the cephem nucleus, ensuring high product quality and consistency. By optimizing the acylation and nucleophilic substitution steps, the process achieves superior yields and simplifies downstream purification requirements. The use of readily available starting materials and common solvents further enhances the economic feasibility of this approach for large-scale manufacturing. Ultimately, this novel methodology represents a substantial leap forward in the development of effective antibacterial therapies.
Mechanistic Insights into Acylation and Nucleophilic Substitution
The core of this synthesis involves a two-step strategy focusing on functionalization at the 7-amino and 3-leaving group positions of the cephem nucleus. Initially, the 7-amino group undergoes acylation with a protected aminothiazole oxime acetic acid derivative, typically activated as an acid chloride or mixed anhydride. This reaction must be carefully controlled to prevent epimerization at the C6 position, which could lead to inactive isomers. The use of protecting groups on the amino functionality of the side chain ensures selective reaction at the desired site while maintaining stability during subsequent steps. Solvent selection plays a crucial role here, with polar aprotic solvents often preferred to facilitate the reaction kinetics without degrading the beta-lactam ring. The resulting 7-acylated intermediate serves as the key precursor for the final quaternization step.
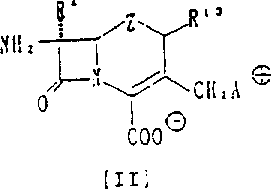
Following acylation, the critical transformation occurs at the 3-position through a nucleophilic substitution reaction. A leaving group at the 3-position, such as an acyloxy or halogen atom, is displaced by a nitrogen atom from the condensed imidazole ring. This quaternization process forms the positively charged ammonium center, which is balanced by a counterion to create a stable inner salt or external salt form. The reaction mechanism proceeds via an SN2 pathway, requiring precise temperature control and stoichiometry to maximize conversion while minimizing byproduct formation. The presence of catalysts like iodides can accelerate the reaction rate, improving overall efficiency. This step is pivotal as it installs the pharmacophore responsible for the enhanced biological activity and improved solubility characteristics of the final compound.
How to Synthesize Cephem Antibacterial Intermediates Efficiently
The synthesis of these high-value intermediates requires a systematic approach to ensure reproducibility and compliance with quality standards. The process begins with the preparation of the 7-amino cephem starting material, followed by sequential acylation and quaternization steps under controlled conditions. Each stage demands careful monitoring of reaction parameters such as temperature, pH, and reaction time to optimize yield and purity. Detailed operational procedures are essential for scaling this chemistry from laboratory benchtop to commercial production volumes. The following guide outlines the standardized workflow for implementing this technology effectively.
- Prepare the 7-amino cephem compound or its salt/ester precursor.
- Perform acylation at the 7-position using a suitable carboxylic acid derivative.
- Execute nucleophilic substitution at the 3-position with an imidazolium compound to form the quaternary ammonium group.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this advanced synthesis route offers significant strategic benefits for procurement and supply chain management within the pharmaceutical sector. The streamlined process reduces the number of unit operations required, thereby lowering overall manufacturing costs and minimizing waste generation. By utilizing robust reaction conditions, the technology ensures consistent product quality, reducing the risk of batch failures and supply disruptions. Furthermore, the use of common reagents and solvents simplifies sourcing logistics and mitigates the risk associated with specialized raw material availability. These factors collectively contribute to a more resilient and cost-effective supply chain for antibiotic production.
- Cost Reduction in Manufacturing: The elimination of complex protection-deprotection sequences and the use of efficient catalytic systems significantly lower the cost of goods sold. By improving reaction yields and reducing solvent consumption, the process minimizes raw material expenses and waste disposal costs. The simplified purification workflow further decreases operational expenditures associated with chromatography and crystallization steps. These efficiencies translate into substantial savings that can be passed on to customers or reinvested in R&D initiatives.
- Enhanced Supply Chain Reliability: The reliance on commercially available starting materials and standard chemical reagents ensures a stable supply base不受 geopolitical or market volatility. The robustness of the synthesis allows for flexible production scheduling, enabling manufacturers to respond quickly to fluctuations in market demand. Additionally, the high stability of the intermediates facilitates easier storage and transportation, reducing the risk of degradation during logistics. This reliability is crucial for maintaining continuous production lines and meeting delivery commitments to downstream API manufacturers.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, allowing for seamless transition from pilot scale to multi-ton commercial production without significant re-engineering. The reduction in hazardous waste and solvent usage aligns with increasingly stringent environmental regulations, supporting sustainable manufacturing practices. Efficient atom economy and energy utilization further enhance the environmental profile of the process. These attributes make the technology attractive for companies aiming to reduce their carbon footprint while maintaining high production volumes.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its potential for integration into their production portfolios. The following questions address common inquiries regarding the chemical properties, regulatory status, and practical application of these intermediates. Answers are derived directly from the technical specifications and experimental data provided in the patent documentation. This information aims to clarify key aspects of the technology for decision-makers.
Q: What distinguishes these cephem compounds from conventional third-generation cephalosporins?
A: These compounds feature a condensed imidazole ring at the 3-position quaternary ammonium group, providing enhanced activity against Pseudomonas aeruginosa and beta-lactamase producing bacteria compared to standard monocyclic pyridyl derivatives.
Q: Are the synthesis methods suitable for large-scale manufacturing?
A: Yes, the patent describes robust reaction conditions using common solvents and reagents, allowing for scalable production from kilogram to multi-ton quantities with established purification protocols.
Q: What is the antimicrobial spectrum of these intermediates?
A: They exhibit broad-spectrum activity against both Gram-positive and Gram-negative bacteria, including resistant strains like Pseudomonas, making them valuable for developing next-generation antibiotics.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cephem Intermediates Supplier
NINGBO INNO PHARMCHEM stands at the forefront of pharmaceutical intermediate manufacturing, offering extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle complex synthetic routes like the one described in CN1030657C, ensuring stringent purity specifications and rigorous QC labs validate every batch. We understand the critical nature of antibiotic supply chains and are committed to delivering high-quality cephem intermediates that meet global regulatory standards. Our team of experts works closely with clients to optimize processes for maximum efficiency and cost-effectiveness.
We invite you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific production needs. By partnering with us, you gain access to specific COA data and route feasibility assessments that demonstrate the viability of this technology for your projects. Let us help you secure a reliable supply of advanced antibacterial intermediates to support your drug development pipeline. Reach out today to discuss how we can collaborate to bring these life-saving medicines to market faster.
