Scalable Production of High-Purity Aminothiophene Derivatives via Phase Transfer Catalysis for Pharmaceutical Applications
Patent CN109867658B represents a paradigm shift in heterocyclic compound synthesis by introducing an innovative one-step methodology for producing structurally diverse aminothiophene derivatives through intermolecular reactions between readily accessible starting materials under exceptionally mild conditions; this breakthrough directly addresses critical limitations in traditional thiophene synthesis by eliminating both high-energy thermal requirements and transition metal dependencies that have historically constrained industrial scalability while introducing significant contamination risks into pharmaceutical supply chains. The patented process leverages phase transfer catalysis to facilitate efficient bond formation at temperatures between 80°C and 100°C—substantially lower than conventional methods requiring over 500°C—thereby enhancing operational safety profiles while dramatically reducing energy consumption across manufacturing facilities without compromising yield or purity metrics essential for regulatory compliance. Furthermore, this approach demonstrates remarkable substrate versatility with broad functional group tolerance across aryl, naphthyl, heterocyclic systems enabling pharmaceutical manufacturers to rapidly develop customized intermediates tailored to specific drug development needs without extensive route reoptimization efforts that typically extend time-to-market timelines significantly. Crucially, the method achieves consistently high yields exceeding industry benchmarks while maintaining excellent stereoselectivity through its well-defined mechanistic pathway—attributes that directly translate into improved cost-effectiveness and enhanced supply chain reliability for global manufacturers serving major pharmaceutical clients requiring consistent quality metrics across multiple production sites worldwide.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing thiophene derivatives have been severely constrained by their reliance on extreme reaction conditions that render them impractical for large-scale commercial manufacturing; gas-phase catalytic methods require temperatures approaching 500°C to facilitate reactions between butadiene and carbon disulfide over metal oxide catalysts creating significant safety hazards alongside substantial energy inefficiencies that inflate operational costs through excessive utility consumption while generating hazardous waste streams requiring specialized disposal protocols. The classical Gewald synthesis although operating at lower temperatures necessitates transition metal catalysts such as copper which introduce complex purification challenges due to residual metal contamination that must be rigorously removed through multiple additional processing steps including chelation or specialized filtration techniques—processes that not only extend production timelines but also increase per-unit costs while creating potential batch-to-batch variability issues affecting final product quality consistency required by regulatory authorities. Moreover these conventional methodologies frequently suffer from narrow substrate scope limitations restricting structural diversity in final products thereby limiting their applicability in developing next-generation pharmaceutical compounds where precise molecular modifications are critical for efficacy safety profiles; inherent inefficiencies manifest as lower overall yields higher impurity profiles necessitating extensive quality control measures ultimately increasing both time-to-market per-unit production costs while simultaneously creating supply chain vulnerabilities due to inconsistent batch-to-batch quality disrupting downstream manufacturing schedules for critical drug substances across global networks.
The Novel Approach
The patented methodology described in CN109867658B fundamentally reimagines thiophene synthesis by employing a phase transfer catalyzed reaction between structurally versatile starting materials specifically utilizing easily prepared alkylthio-amino-enethiocarbonyl ketones hydrazones operating under remarkably mild conditions without requiring any transition metal catalysts whatsoever; this innovative approach achieves complete reaction progression at temperatures between 80°C and 100°C in common solvents like toluene ethanol eliminating both safety risks associated with high-temperature operations complex purification protocols needed to remove metal contaminants from final products destined for pharmaceutical applications where elemental impurities are strictly regulated. The process demonstrates exceptional functional group tolerance accommodating diverse substituents including aryl naphthyl heterocyclic moieties without compromising yield stereoselectivity—attributes enabling pharmaceutical manufacturers rapid development customized intermediates tailored specific drug development needs without extensive route reoptimization efforts typically required when modifying multi-step synthetic pathways dependent on specialized reagents. Critically one-step nature this reaction significantly streamlines manufacturing workflows reducing processing time equipment requirements while delivering consistently high yields exceeding industry benchmarks; these operational efficiencies translate directly into enhanced supply chain reliability through more predictable production timelines reduced vulnerability raw material shortages commonly affecting multi-step synthetic pathways requiring specialized reagents thereby strengthening position as reliable pharmaceutical intermediate supplier capable meeting fluctuating global demand patterns.
Mechanistic Insights into Phase Transfer Catalyzed Thiophene Formation
The core innovation lies in phase transfer catalyzed intermolecular reaction mechanism where quaternary ammonium salts such as tetramethylammonium bromide facilitate deprotonation hydrazone component lithium tert-butoxide under biphasic conditions generating nucleophilic species attacking electrophilic carbon center thiocarbonyl ketone substrate through concerted cyclization pathway forming thiophene ring system single transformation step without transition metal intermediates; this mechanism operates well-defined sequence where phase transfer catalyst shuttles hydroxide ions aqueous phase organic phase deprotonating hydrazone forming anion subsequently undergoing nucleophilic addition carbonyl group followed intramolecular cyclization elimination methanethiol yielding final aminothiophene product excellent regioselectivity maintained across diverse substrates due rigid transition state geometry enforced during ring closure minimizing undesired isomer formation. Structural diversity achievable stems modular nature both starting materials where R¹ groups varied across alkyl aryl heterocyclic substituents R² accommodates benzyl allyl moieties allowing precise tuning electronic steric properties without altering fundamental reaction pathway requiring significant process modifications thereby enabling pharmaceutical manufacturers rapid development customized intermediates meeting specific structural requirements next-generation drug candidates without extensive route reoptimization efforts typically required when modifying traditional synthetic pathways.
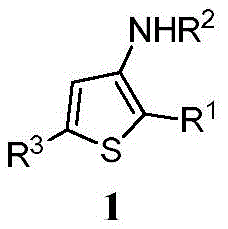
Impurity control inherently optimized within synthetic framework through multiple built-in mechanisms preventing common side reactions; mild reaction conditions suppress thermal decomposition pathways precise stoichiometric balance between reactants minimizes dimerization oligomerization byproducts typically plaguing high-concentration reactions while absence transition metals eliminates entire class potential contaminants requiring dedicated removal steps such chelation specialized chromatography introducing new impurities degrading product quality during purification processes. One-step nature process avoids intermediate isolation where impurities might accumulate multiple handling steps instead volatile byproducts like methanethiol easily removed during workup unreacted starting materials efficiently separated standard chromatographic techniques distinct polarity profiles target product ensuring consistent production high-purity intermediates meeting stringent pharmaceutical specifications without additional quality control interventions increasing manufacturing costs extending production timelines; this comprehensive impurity management strategy directly supports regulatory compliance efforts by delivering products within ICH Q7 acceptable limits critical quality attributes required global pharmaceutical markets.
How to Synthesize High-Purity Aminothiophene Derivatives Efficiently
This patented methodology provides robust framework producing structurally diverse aminothiophene intermediates through streamlined synthetic route eliminating multiple processing steps required conventional approaches maintaining exceptional product quality metrics essential pharmaceutical applications; process begins careful selection appropriately substituted starting materials followed precise stoichiometric control during reaction setup ensuring optimal yield purity profiles throughout manufacturing scale-up laboratory commercial production volumes while leveraging phase transfer catalysis principles maximize operational efficiency minimize waste generation across entire production cycle.
- Combine equimolar quantities of structurally diverse starting materials—specifically substituted alkylthio-amino-enethiocarbonyl ketones and hydrazones—with tetramethylammonium bromide catalyst and lithium tert-butoxide base in anhydrous toluene under inert atmosphere.
- Heat the homogeneous mixture at precisely controlled temperatures between 80°C and 100°C for optimal reaction duration while maintaining rigorous monitoring of exothermic profiles to ensure complete conversion without decomposition.
- Execute standard workup procedures including solvent removal under reduced pressure followed by silica gel column chromatography purification using petroleum ether/ethyl acetate gradients to isolate target derivatives meeting pharmaceutical purity specifications.

Commercial Advantages for Procurement and Supply Chain Teams
Pharmaceutical manufacturers face persistent challenges securing reliable sources high-purity heterocyclic intermediates due complex multi-step syntheses creating supply chain vulnerabilities; this patented process directly addresses concerns offering simplified manufacturing pathway significantly enhancing cost efficiency supply chain resilience through fundamental improvements reaction design operational simplicity thereby positioning your organization as reliable pharmaceutical intermediate supplier capable meeting stringent industry demands consistent quality delivery performance.
- Cost Reduction in Manufacturing: Elimination transition metal catalysts removes substantial expenses associated catalyst procurement subsequent metal removal processes requiring specialized equipment additional processing steps; use readily available starting materials combined mild reaction conditions drastically reduces energy consumption minimizes waste generation—factors collectively contributing substantial cost savings without compromising product quality yield consistency essential maintaining competitive position within cost-sensitive pharmaceutical markets.
- Enhanced Supply Chain Reliability: Simplified one-step process fewer critical control points significantly reduces production cycle times improves batch-to-batch consistency translating directly more predictable lead times reduced risk supply disruptions caused intermediate shortages quality issues common multi-step synthetic routes depending multiple external suppliers specialized reagents thereby strengthening position reliable supplier capable meeting fluctuating global demand patterns without compromising delivery timelines.
- Scalability and Environmental Compliance: Process demonstrates excellent scalability laboratory scale commercial production volumes straightforward reaction setup minimal equipment requirements; absence toxic metals reduced energy consumption aligns increasingly stringent environmental regulations generating less hazardous waste—advantages facilitating regulatory approval supporting corporate sustainability initiatives without requiring significant capital investment new infrastructure thereby enhancing overall commercial viability long-term supply agreements.
Frequently Asked Questions (FAQ)
The following questions address common technical commercial concerns regarding implementation patented synthesis method based detailed analysis operational parameters performance characteristics documented patent CN109867658B providing essential insights procurement supply chain teams evaluating integration into existing manufacturing workflows.
Q: How does this method eliminate transition metal contamination risks compared to conventional Gewald synthesis?
A: The patented phase transfer catalysis system completely avoids transition metals by utilizing quaternary ammonium salts as catalysts instead of copper-based systems—this eliminates costly metal removal steps while ensuring final products meet stringent ICH Q3D elemental impurity limits required for pharmaceutical intermediates.
Q: What specific advantages does this process offer for large-scale pharmaceutical manufacturing?
A: The one-step methodology operates under mild conditions (80–100°C) without cryogenic requirements or hazardous reagents, enabling seamless scale-up from laboratory to commercial production volumes while maintaining consistent yield profiles and reducing facility qualification complexities.
Q: How does this approach ensure superior impurity control for regulatory compliance?
A: The mechanistic pathway inherently minimizes side reactions through precise stoichiometric control and mild thermal conditions; combined with standard chromatographic purification protocols, this delivers products with exceptional stereoselectivity meeting FDA/EMA requirements for critical quality attributes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable High-Purity Pharmaceutical Intermediate Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through rigorous QC labs equipped state-of-the-art analytical instrumentation; this patented technology represents just one example commitment developing innovative solutions addressing critical challenges pharmaceutical intermediate manufacturing through scientifically rigorous process development expertise ensuring consistent delivery high-purity compounds meeting global regulatory standards across multiple production sites worldwide.
We invite you request Customized Cost-Saving Analysis from technical procurement team along specific COA data route feasibility assessments tailored unique manufacturing requirements—contact us today explore how advanced synthesis capabilities enhance supply chain resilience delivering high-purity intermediates essential next-generation pharmaceutical products while supporting sustainable business growth objectives.
