Advanced Manufacturing of Rosuvastatin Salts: Technical Breakthroughs and Commercial Scalability for Global Supply Chains
The global demand for high-performance lipid-lowering agents continues to drive innovation in the synthesis of statin intermediates, particularly for rosuvastatin, a leading compound in cardiovascular therapy. Patent CN102317267A introduces a transformative approach to preparing rosuvastatin salts, specifically targeting the critical challenges of purity and stability that have long plagued conventional manufacturing routes. This technical disclosure outlines a novel method for converting rosuvastatin tert-butylammonium salt into pharmacologically acceptable divalent cation salts, such as calcium or zinc salts, using a sophisticated biphasic solvent system. By shifting away from purely aqueous environments, this process addresses the inherent instability of the statin structure, offering a robust pathway for producing active pharmaceutical ingredients with superior quality attributes. For industry stakeholders, this represents a significant leap forward in process chemistry, enabling the reliable production of high-purity pharmaceutical intermediates that meet rigorous international regulatory standards.
Traditional methods for synthesizing rosuvastatin calcium salt have predominantly relied on aqueous media, where the free acid or sodium salt is reacted with calcium sources in water. While conceptually straightforward, these conventional approaches suffer from severe limitations regarding product quality and process efficiency. The high water content in the reaction medium often leads to substantial hydration of the final crystal lattice, making subsequent drying processes energy-intensive and time-consuming. More critically, the presence of water during isolation and drying promotes the degradation of the sensitive statin side chain, resulting in the formation of undesirable lactone impurities and 5-oxo derivatives. These degradation products not only compromise the therapeutic efficacy of the drug but also necessitate complex purification steps that reduce overall yield and increase manufacturing costs significantly.
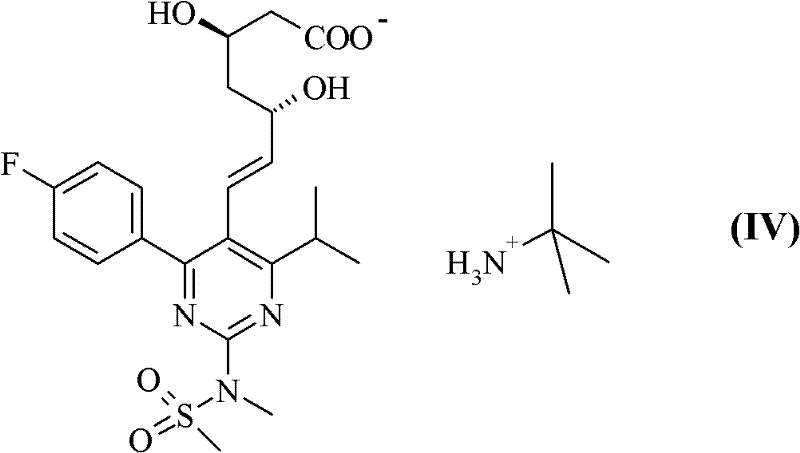
In stark contrast to these legacy techniques, the novel approach detailed in the patent utilizes a biphasic mixture of water and a water-immiscible organic solvent, preferably ethyl acetate, to facilitate the salt formation reaction. This strategic modification fundamentally alters the reaction environment, shielding the sensitive rosuvastatin molecule from excessive hydrolytic stress during the critical precipitation phase. The process begins with the dissolution of rosuvastatin tert-butylammonium salt in the organic-aqueous mixture, followed by the controlled addition of a calcium or zinc ion source. This method allows for precise control over supersaturation and crystal growth, resulting in a product with markedly lower water content and significantly reduced levels of amine residues. The ability to isolate the salt directly from the organic phase minimizes exposure to bulk water, thereby preserving the structural integrity of the molecule and ensuring a much cleaner impurity profile from the outset.
Mechanistic Insights into Two-Phase Salt Formation and Impurity Control
The core mechanistic advantage of this invention lies in the manipulation of solubility and partition coefficients within the biphasic system. When the rosuvastatin tert-butylammonium salt interacts with the calcium ion source in the presence of ethyl acetate, the resulting calcium salt exhibits low solubility in the organic phase but is protected from the bulk aqueous environment by the solvent interface. This unique microenvironment prevents the extensive hydration typically observed in single-phase aqueous crystallizations. Furthermore, the organic solvent facilitates the efficient removal of tert-butylamine, a byproduct of the salt exchange reaction, by keeping it dissolved in the organic layer or allowing it to be washed away effectively during the workup phase. This mechanism ensures that the final crystalline product is virtually free from amine contamination, a common issue in prior art methods that often require multiple recrystallizations to achieve acceptable limits.
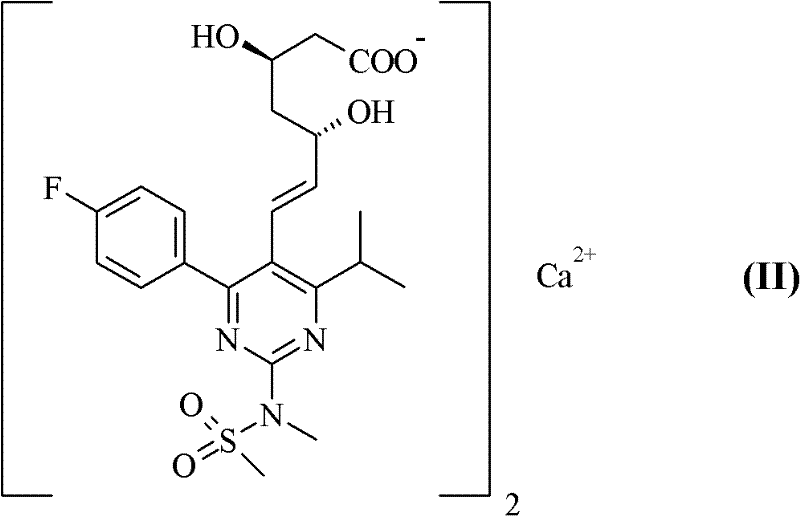
Impurity control is another critical aspect where this new methodology excels, particularly concerning the formation of rosuvastatin lactone and 5-oxo compounds. In conventional aqueous processes, the drying of wet cake at elevated temperatures often triggers intramolecular cyclization, converting the open acid form into the inactive lactone. The patent data demonstrates that products obtained via the biphasic method possess inherently lower moisture content, often below 2%, compared to over 50% in some aqueous precipitates before drying. This reduced water load means that less thermal energy is required to dry the product, significantly lowering the thermal stress on the molecule. Consequently, the generation of lactone impurities during the drying process is drastically suppressed, maintaining the potency and stability of the high-purity rosuvastatin calcium salt throughout its shelf life and downstream processing.
How to Synthesize Rosuvastatin Calcium Salt Efficiently
The synthesis protocol described in the patent provides a clear roadmap for implementing this advanced chemistry in a production setting. It emphasizes the importance of solvent ratios, temperature control, and the mode of reagent addition to optimize yield and purity. The process is designed to be operationally simple yet chemically precise, allowing manufacturers to transition from lab-scale experiments to commercial production with minimal risk. Detailed standard operating procedures regarding the specific molar equivalents of calcium sources and the precise timing of phase separation are crucial for replicating the high success rates reported in the examples. For a comprehensive guide on the standardized synthesis steps, please refer to the technical section below.
- Dissolve rosuvastatin tert-butylammonium salt in a mixture of ethyl acetate and water under vigorous stirring at controlled temperatures between 0°C and 50°C.
- Add a calcium ion source, such as saturated calcium chloride solution or solid calcium acetate, dropwise to the biphasic mixture while maintaining agitation.
- Separate the organic phase, wash with aqueous calcium salt solutions, dehydrate via distillation, and precipitate the final product using non-polar solvents like cyclohexane.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this novel synthesis route offers profound benefits for procurement managers and supply chain directors seeking to optimize their sourcing strategies for statin intermediates. The primary advantage lies in the substantial cost reduction in API manufacturing driven by the elimination of inefficient processing steps. Traditional methods often require extensive drying cycles and multiple recrystallizations to remove water and amine impurities, which consume significant energy and time. By producing a drier, purer crude product directly from the reaction, this new process streamlines the manufacturing workflow, reducing utility consumption and increasing equipment throughput. This efficiency translates directly into lower production costs, allowing suppliers to offer more competitive pricing without compromising on quality standards.
- Cost Reduction in Manufacturing: The biphasic process eliminates the need for energy-intensive drying protocols associated with highly hydrated aqueous precipitates. By reducing the initial water content of the filter cake, manufacturers can significantly cut down on vacuum drying time and heating requirements. Furthermore, the high purity of the crude product reduces the necessity for costly recrystallization steps, saving both solvent costs and labor hours. These operational efficiencies accumulate to provide substantial cost savings over the lifecycle of the product, making it a financially attractive option for large-scale production runs.
- Enhanced Supply Chain Reliability: Supply chain stability is often compromised by batch failures due to out-of-specification impurity profiles. The robust nature of this two-phase method ensures consistent product quality with minimal batch-to-batch variation. The reduced sensitivity to drying conditions means that production schedules are less likely to be disrupted by extended processing times needed to meet moisture specifications. This reliability allows for more accurate forecasting and inventory management, ensuring a continuous supply of high-purity pharmaceutical intermediates to downstream formulation partners without unexpected delays.
- Scalability and Environmental Compliance: The use of common solvents like ethyl acetate and cyclohexane makes this process highly scalable using standard chemical engineering equipment. Unlike methods requiring exotic reagents or extreme conditions, this route can be easily adapted to existing manufacturing facilities. Additionally, the improved yield and reduced waste generation contribute to a smaller environmental footprint. The ability to recycle solvents and the reduction in wastewater treatment loads due to lower aqueous volumes align with modern green chemistry principles, facilitating easier regulatory compliance and sustainability reporting for manufacturing sites.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. They are derived from the specific advantages and data points presented in the patent documentation, providing clarity on how this method compares to existing industry standards. Understanding these details is essential for technical teams evaluating the feasibility of adopting this new route for their specific production needs.
Q: How does the new two-phase method improve purity compared to traditional aqueous methods?
A: The novel two-phase system significantly reduces water content in the final product, minimizing the formation of lactone impurities during drying and storage, thereby achieving HPLC purity levels exceeding 99.8%.
Q: What are the residual amine levels in the rosuvastatin calcium salt produced by this method?
A: Unlike prior art methods which often leave high levels of tert-butylamine, this process reduces amine content to below 100ppm, meeting stringent pharmaceutical quality requirements without extensive recrystallization.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the process utilizes common solvents like ethyl acetate and operates at mild temperatures, making it highly scalable and safe for industrial manufacturing while maintaining consistent yield and quality.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rosuvastatin Calcium Salt Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes to maintain competitiveness in the global pharmaceutical market. Our team of expert chemists has thoroughly analyzed the technical potential of the biphasic salt formation method described in CN102317267A and is fully prepared to implement this technology at scale. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are realized in actual manufacturing output. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the detection of trace lactone and amine impurities at ppm levels, guaranteeing that every batch meets the highest international standards.
We invite procurement leaders and technical directors to collaborate with us to leverage this innovative process for your supply chain. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this higher-efficiency route for your projects. We encourage you to contact our technical procurement team to request specific COA data from our pilot runs and detailed route feasibility assessments tailored to your volume requirements. Let us help you secure a stable, cost-effective, and high-quality supply of rosuvastatin intermediates that drives your business forward.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
