Advanced Biocatalytic Henry Reaction for Scalable Pharmaceutical Intermediate Production
The landscape of chiral synthesis is undergoing a transformative shift driven by the urgent demand for sustainable and highly selective manufacturing processes. Patent CN106795542B introduces a groundbreaking methodology utilizing (R)-selective cupin-nitroaldolase enzymes to catalyze the Henry reaction, a pivotal carbon-carbon bond-forming transformation. This technology addresses the critical need for efficient production of chiral beta-nitroalcohol compounds, which serve as essential precursors for bioactive molecules such as ephedrine, phenstatin, and sphingosine derivatives. By leveraging the unique structural properties of the cupin superfamily, this invention enables the enantioselective conversion of aldehydes or ketones with nitroalkanes under mild physiological conditions. For R&D directors and process chemists, this represents a significant departure from traditional chemical catalysis, offering a pathway to high-purity intermediates with reduced environmental impact and simplified downstream processing requirements.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral beta-nitroalcohols has relied heavily on non-enzymatic organocatalysts or chiral metal complexes, which often necessitate harsh reaction conditions and generate substantial hazardous waste. Conventional chemical Henry reactions frequently require strong bases or expensive transition metal catalysts that pose significant challenges regarding toxicity, removal, and disposal in a regulated pharmaceutical environment. Furthermore, earlier biocatalytic attempts using hydroxynitrile lyases (HNLs) such as AtHNL from Arabidopsis thaliana demonstrated limited utility due to stability issues; specifically, the enantiomeric excess of the reaction product was observed to decrease significantly over extended reaction times without a corresponding increase in yield. This instability creates a bottleneck for process optimization, forcing manufacturers to sacrifice either conversion efficiency or optical purity, thereby complicating the supply chain for high-value active pharmaceutical ingredients.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes engineered cupin-nitroaldolases that exhibit exceptional stability and stereoselectivity over prolonged incubation periods. The invention highlights specific variants, such as GtHNL-A40H/V42T/Q110H, which achieve conversion rates nearing 70% with enantiomeric excess values exceeding 97% even after six hours of reaction time. Unlike previous enzymatic methods that suffered from selectivity loss, these cupin-based catalysts maintain their stereochemical integrity, allowing for longer reaction windows to maximize yield without compromising product quality. This robustness facilitates the use of simpler reaction setups, including biphasic systems that enhance substrate solubility and product recovery, ultimately streamlining the manufacturing workflow for complex chiral building blocks.
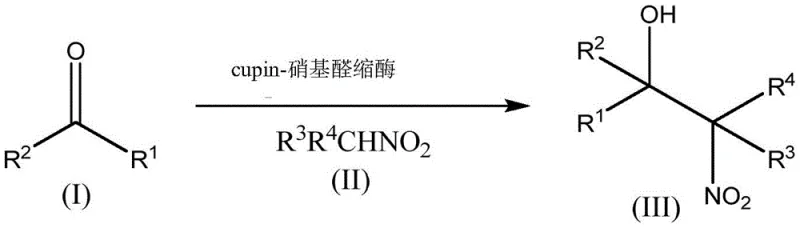
Mechanistic Insights into Cupin-Nitroaldolase Catalyzed Henry Reaction
The catalytic prowess of these enzymes stems from the conserved beta-barrel domain characteristic of the cupin superfamily, which provides a rigid and well-defined active site for substrate binding. Structural analysis reveals that these proteins function as metal-dependent biocatalysts, typically coordinating divalent cations such as manganese, iron, or zinc within their active centers to facilitate the nucleophilic attack of the nitroalkane on the carbonyl substrate. The precise arrangement of amino acid residues surrounding the metal ion creates a chiral environment that strictly controls the facial selectivity of the addition, ensuring the formation of the desired (R)-enantiomer with high fidelity. This metal-dependence is not merely structural but mechanistic, as the metal ion likely activates the nitroalkane by stabilizing the resulting nitronate anion, thereby lowering the activation energy for the C-C bond formation step.
Furthermore, the engineering of specific amino acid substitutions at positions 40, 42, and 110 within the protein sequence has been shown to dramatically enhance both activity and selectivity compared to wild-type sequences. These modifications optimize the interactions between the enzyme and the transition state, effectively suppressing side reactions and preventing the racemization that plagues less stable catalysts. The ability to operate effectively in biphasic systems containing organic solvents like methyl tert-butyl ether (MTBE) further underscores the versatility of this mechanism, as the enzyme retains its folded structure and catalytic function even in the presence of water-immiscible organic phases. This compatibility is crucial for industrial applications where substrate loading often exceeds aqueous solubility limits.

How to Synthesize Chiral Beta-Nitroalcohols Efficiently
The implementation of this biocatalytic route involves a streamlined workflow that begins with the recombinant expression of the optimized cupin-nitroaldolase variants in a suitable microbial host such as E. coli. The process leverages standard fermentation techniques supplemented with necessary metal cofactors to ensure the production of active holo-enzyme, followed by simple cell lysis to release the biocatalyst for use in crude or purified forms. Detailed standardized synthesis protocols outlining the precise buffering conditions, substrate ratios, and workup procedures are essential for reproducibility and scale-up, ensuring that the high selectivity observed in laboratory settings translates to commercial manufacturing environments.
- Express recombinant cupin-nitroaldolase variants (e.g., GtHNL-A40H/V42T/Q110H) in E. coli host cells supplemented with manganese ions.
- Prepare a biphasic reaction system consisting of an organic phase containing the aldehyde substrate and nitroalkane, and an aqueous buffer phase containing the enzyme lysate.
- Incubate the mixture at mild temperatures (0-35°C) to achieve high conversion and enantiomeric excess, followed by product extraction and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this cupin-nitroaldolase technology presents a compelling value proposition centered on risk mitigation and operational efficiency. The shift from chemical catalysis to biocatalysis eliminates the reliance on scarce and expensive transition metals, thereby insulating the supply chain from volatile commodity pricing and geopolitical supply disruptions associated with mining and refining rare earth elements. Additionally, the mild reaction conditions significantly reduce energy consumption related to heating and cooling, contributing to lower overall utility costs and a smaller carbon footprint for the manufacturing facility. The high specificity of the enzyme minimizes the formation of by-products, which simplifies purification trains and reduces the volume of solvent and consumables required for chromatography or crystallization steps.
- Cost Reduction in Manufacturing: The elimination of toxic heavy metal catalysts removes the need for costly and complex metal scavenging steps, which are often mandatory to meet stringent regulatory limits for residual metals in pharmaceutical products. By utilizing a biological catalyst that operates at ambient temperatures and neutral pH, the process drastically reduces the energy load associated with maintaining extreme reaction conditions, leading to substantial operational expenditure savings. Furthermore, the high conversion rates and selectivity minimize raw material waste, ensuring that expensive chiral starting materials are utilized with maximum efficiency, directly improving the cost of goods sold for the final active ingredient.
- Enhanced Supply Chain Reliability: The recombinant nature of the cupin-nitroaldolase ensures a consistent and scalable supply of the biocatalyst, as it can be produced in large quantities using established fermentation infrastructure without dependence on plant extraction or animal sources. The robustness of the enzyme variants allows for flexible scheduling and longer batch cycles without the risk of catalyst degradation, providing greater predictability in production timelines and delivery schedules. This reliability is critical for maintaining continuous manufacturing operations and meeting the just-in-time delivery requirements of global pharmaceutical clients who demand unwavering quality and consistency.
- Scalability and Environmental Compliance: The compatibility of the enzyme with biphasic reaction systems facilitates straightforward scale-up from laboratory to pilot and commercial scales, as mass transfer limitations can be managed through standard agitation and mixing protocols. The aqueous-based nature of the biocatalytic step significantly reduces the generation of hazardous organic waste streams, aligning with increasingly strict environmental regulations and corporate sustainability goals. This green chemistry profile not only simplifies waste disposal logistics but also enhances the brand reputation of the manufacturer as a responsible partner committed to sustainable development practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of cupin-nitroaldolase technology in industrial settings, based on the specific capabilities and data presented in the patent literature. Understanding these nuances is vital for stakeholders evaluating the feasibility of integrating this biocatalytic route into existing production pipelines or new drug development programs.
Q: What are the advantages of cupin-nitroaldolases over traditional chemical catalysts?
A: Cupin-nitroaldolases offer superior enantioselectivity (>99% ee) under mild aqueous conditions, eliminating the need for toxic heavy metal catalysts and extreme pH levels required in conventional Henry reactions.
Q: Is the enzyme stable during prolonged reaction times?
A: Yes, engineered variants like GtHNL-A40H/V42T/Q110H maintain high enantiomeric excess even after 24 hours of incubation, unlike previous enzymes such as AtHNL which suffer from reduced selectivity over time.
Q: Can this process be scaled for industrial manufacturing?
A: The process utilizes robust recombinant enzymes expressible in standard microbial hosts and functions effectively in biphasic systems, facilitating easy product separation and scalability for commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-nitroalcohol Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced biocatalytic routes like the cupin-nitroaldolase system for producing high-value chiral intermediates. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are successfully translated into robust manufacturing processes. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of beta-nitroalcohol meets the exacting standards required for downstream pharmaceutical synthesis.
We invite you to collaborate with our technical procurement team to explore how this technology can optimize your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this enzymatic process for your target molecules. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project requirements, ensuring a seamless transition to a more efficient and sustainable manufacturing model.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
