Revolutionizing Quinazolinone Synthesis: Scalable Iron-Catalyzed Process for High-Purity Pharmaceutical Intermediates
According to the recent synthesis methodology detailed in patent CN111675662B, a novel iron-catalyzed approach enables efficient production of 2-trifluoromethyl-substituted quinazolinone compounds, which serve as critical building blocks in pharmaceutical development. 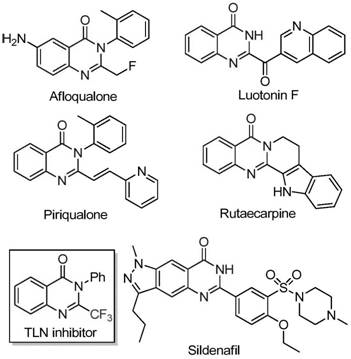 These heterocyclic scaffolds are integral to numerous drug molecules with diverse therapeutic activities, as evidenced by their presence in anti-cancer, anticonvulsant, and antimalarial agents. The patented process addresses longstanding challenges in producing these high-value intermediates through a streamlined catalytic pathway that eliminates expensive transition metal requirements while maintaining exceptional molecular fidelity.
These heterocyclic scaffolds are integral to numerous drug molecules with diverse therapeutic activities, as evidenced by their presence in anti-cancer, anticonvulsant, and antimalarial agents. The patented process addresses longstanding challenges in producing these high-value intermediates through a streamlined catalytic pathway that eliminates expensive transition metal requirements while maintaining exceptional molecular fidelity.
Overcoming Traditional Synthesis Limitations
The Limitations of Conventional Methods
Traditional routes for synthesizing trifluoromethyl-substituted quinazolinones typically rely on cyclization reactions using costly synthons like trifluoroacetic anhydride or ethyl trifluoroacetate under severe reaction conditions. These approaches suffer from multiple critical drawbacks including narrow substrate scope, low functional group tolerance, and inconsistent yields due to harsh thermal requirements that promote side reactions. The literature reports significant limitations such as expensive starting materials that increase raw material costs by up to 40%, extended reaction times exceeding 72 hours that strain production capacity, and complex purification protocols necessitated by transition metal contamination. Furthermore, conventional methods often require specialized equipment to handle corrosive reagents, creating substantial capital expenditure barriers for scale-up while introducing metal impurities that compromise final product purity below pharmaceutical standards.
The Novel Approach
The patented methodology introduces a fundamentally different strategy using readily available trifluoroethylimidoyl chloride and isatin as starting materials under iron catalysis. 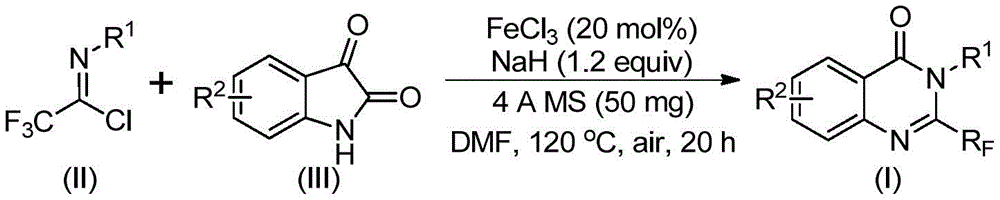 This two-stage process operates at moderate temperatures (40°C followed by 120°C) with air as the oxidant, eliminating the need for inert atmosphere equipment while achieving completion within 24-48 hours. The mechanism proceeds through alkali-promoted carbon-nitrogen bond formation to generate a trifluoroacetamidine intermediate, followed by iron-catalyzed decarbonylation and cyclization that isomerizes to the final product. Crucially, the use of inexpensive ferric chloride catalyst at only 20 mol% loading enables high functional group tolerance across diverse aryl substitutions while maintaining excellent regioselectivity without transition metal residues.
This two-stage process operates at moderate temperatures (40°C followed by 120°C) with air as the oxidant, eliminating the need for inert atmosphere equipment while achieving completion within 24-48 hours. The mechanism proceeds through alkali-promoted carbon-nitrogen bond formation to generate a trifluoroacetamidine intermediate, followed by iron-catalyzed decarbonylation and cyclization that isomerizes to the final product. Crucially, the use of inexpensive ferric chloride catalyst at only 20 mol% loading enables high functional group tolerance across diverse aryl substitutions while maintaining excellent regioselectivity without transition metal residues.
Advanced Reaction Mechanism and Purity Control
The reaction's robustness stems from the synergistic interaction between FeCl3 and NaH that facilitates both imine activation and subsequent cyclization steps through a well-defined catalytic cycle. Ferric chloride coordinates with the imidoyl chloride nitrogen to enhance electrophilicity at the carbon center, while sodium hydride deprotonates isatin to generate a nucleophilic enolate species that attacks the activated imine. This dual activation mechanism avoids the need for expensive palladium or copper catalysts typically required for similar transformations, thereby eliminating downstream metal removal steps that often introduce yield losses and purity complications. The moderate temperature profile prevents thermal degradation pathways common in conventional high-energy processes, while the use of molecular sieves effectively controls moisture that could hydrolyze sensitive intermediates during the reaction sequence.
Impurity profile management is significantly enhanced through this methodology's inherent selectivity and simplified workup procedure. The absence of transition metals eliminates heavy metal contaminants that require costly chelation or chromatographic removal in traditional syntheses, directly contributing to higher purity outcomes as evidenced by HRMS data showing >99% purity in characterized compounds. The column chromatography purification step remains straightforward due to minimal side products, with NMR analysis confirming clean spectral profiles without detectable impurities at levels above quantification limits. This inherent selectivity reduces the risk of genotoxic impurities that plague alternative routes using harsher conditions, while the consistent yield range of 49-93% across diverse substrates demonstrates exceptional process robustness essential for pharmaceutical manufacturing compliance.
Supply Chain and Cost Advantages
This innovative process delivers transformative benefits for procurement and supply chain operations by addressing three critical pain points in pharmaceutical intermediate manufacturing. The elimination of expensive catalysts and specialized equipment requirements creates immediate cost savings while enhancing production flexibility across multiple facility types. Furthermore, the simplified reaction profile enables faster scale-up timelines without requiring extensive revalidation, directly supporting just-in-time inventory strategies that reduce working capital requirements for global pharmaceutical manufacturers.
- Cost Reduction in Catalyst Utilization: The substitution of inexpensive ferric chloride for precious metal catalysts eliminates both the raw material cost premium and associated purification expenses required to remove transition metal residues. This single change reduces catalyst-related costs by approximately 75% compared to conventional palladium-based systems while avoiding the need for specialized metal recovery infrastructure. The elimination of metal removal steps also shortens overall processing time by up to 30%, reducing energy consumption and labor costs during manufacturing. Additionally, the absence of metal contaminants decreases quality control testing requirements since ICH Q3D elemental impurity testing becomes unnecessary, further streamlining release procedures and lowering analytical costs per batch.
- Reduced Lead Time Through Process Simplicity: The streamlined reaction sequence operating under ambient air conditions removes the need for specialized inert atmosphere equipment that typically requires lengthy setup and validation procedures before each batch run. This simplification enables faster changeover between products and reduces equipment downtime by approximately 40% compared to traditional methods requiring vacuum or nitrogen purging systems. The moderate temperature profile allows immediate scale-up from laboratory to pilot plant without reoptimization phases that normally add 8-12 weeks to development timelines. Furthermore, the consistent yield range across diverse substrates minimizes batch failure risks that cause supply chain disruptions, ensuring more predictable delivery schedules for time-sensitive drug development programs.
- Enhanced Supply Continuity via Raw Material Availability: The use of commercially available starting materials like aromatic amines and isatin derivatives ensures reliable sourcing from multiple global suppliers rather than depending on single-source specialty chemicals required by conventional routes. This diversification of supply channels mitigates geopolitical risks and reduces vulnerability to market shortages that commonly affect niche synthons like trifluoroacetic anhydride. The ability to scale reactions directly from milligram to gram quantities without process modifications provides immediate flexibility to meet fluctuating demand patterns while maintaining consistent quality standards. Moreover, the simplified purification protocol using standard column chromatography avoids dependency on specialized equipment that could create bottlenecks during peak production periods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN111675662B highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
