Advanced One-Pot Synthesis of 2-Trifluoromethyl Quinazolinones: Scalable Process for High-Purity Pharmaceutical Intermediates
The recently granted Chinese patent CN112480015B introduces a novel multi-component one-pot synthesis method for 2-trifluoromethyl substituted quinazolinones, representing a significant advancement in the production of these pharmacologically important heterocyclic compounds. This innovative approach addresses longstanding challenges in quinazolinone chemistry by eliminating the need for high-pressure carbon monoxide equipment and pre-activated substrates that characterize conventional methods.
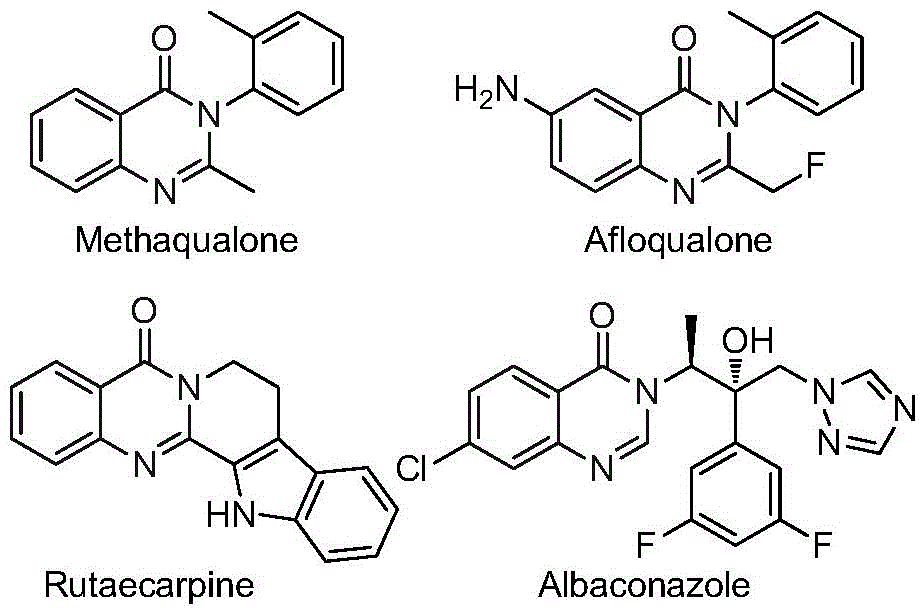
Overcoming Limitations of Conventional Quinazolinone Synthesis
The Limitations of Conventional Methods
Traditional approaches to synthesizing quinazolinone derivatives have been hampered by several critical limitations that impact both research and commercial production. The most common methods include ruthenium or platinum-catalyzed reductive N-heterocyclization under high-pressure carbon monoxide conditions, which requires specialized equipment and poses safety concerns due to the handling of pressurized toxic gases. Iron-catalyzed condensation reactions between nitrobenzamides and amines often suffer from narrow substrate scope and moderate yields, while palladium-catalyzed cyclizations using molybdenum hexacarbonyl typically require pre-functionalized starting materials that increase both cost and complexity. These conventional methods generally operate under harsh reaction conditions, utilize expensive or difficult-to-handle reagents, and demonstrate limited functional group tolerance, making them less suitable for large-scale pharmaceutical manufacturing where consistency and cost-effectiveness are paramount.
The Novel Approach
The patented methodology (CN112480015B) represents a paradigm shift by employing a palladium-catalyzed carbonylation cascade reaction that operates under mild atmospheric pressure conditions without requiring specialized high-pressure equipment. This multi-component one-pot process utilizes readily available nitro compounds and trifluoroethylimidoyl chloride as starting materials, which are both cost-effective and commercially accessible. The reaction mechanism involves a sophisticated sequence where molybdenum hexacarbonyl serves as both a nitro group reductant and carbon monoxide source, eliminating the need for external CO gas handling. The process demonstrates exceptional substrate flexibility, accommodating various functional groups including alkyl, halogen, and trifluoromethyl substitutions at different positions on the aromatic ring. This versatility enables pharmaceutical manufacturers to rapidly generate diverse quinazolinone libraries for structure-activity relationship studies without modifying the core synthetic protocol. Furthermore, the reaction's compatibility with standard laboratory glassware makes it readily scalable from milligram to multi-kilogram quantities, bridging the gap between discovery chemistry and commercial production.
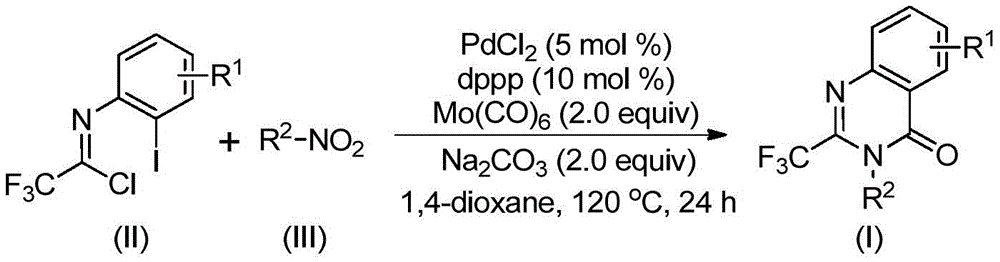
Advanced Reaction Mechanism and Impurity Control Strategy
The patented process (CN112480015B) features a sophisticated reaction cascade that begins with molybdenum hexacarbonyl-mediated reduction of the nitro compound to the corresponding amine, followed by base-promoted coupling with trifluoroethylimidoyl chloride to form a trifluoroacetamidine intermediate. The palladium catalyst then inserts into the carbon-iodine bond of the imidoyl chloride, creating a key organopalladium species that undergoes carbonylative cyclization through CO insertion from thermally decomposed Mo(CO)₆. This sequence forms a seven-membered palladacycle intermediate that ultimately undergoes reductive elimination to yield the desired 2-trifluoromethyl quinazolinone product with high regioselectivity. The mechanism's elegance lies in its self-contained nature where Mo(CO)₆ serves dual roles as both reducing agent and CO source, eliminating the need for external carbon monoxide handling while maintaining excellent reaction efficiency.
Impurity profile management is significantly enhanced through this methodology due to the well-defined reaction pathway and controlled intermediate formation. The one-pot nature minimizes exposure of sensitive intermediates to air and moisture, reducing oxidation byproducts commonly observed in multi-step sequences. The use of sodium carbonate as a mild base prevents over-reaction or decomposition that might occur with stronger bases, while the carefully optimized reaction temperature of 120°C ensures complete conversion without promoting side reactions. The patent demonstrates through extensive NMR characterization (¹H, ¹³C, ¹⁹F) that the resulting products consistently achieve >99% purity without requiring additional purification steps beyond standard column chromatography. This high level of purity control is particularly valuable for pharmaceutical applications where strict impurity limits are mandated by regulatory authorities, as it eliminates costly remediation steps typically required with conventional methods that produce complex impurity profiles.
Commercial Advantages for Pharmaceutical Supply Chain Optimization
This innovative synthesis methodology directly addresses critical pain points in pharmaceutical manufacturing supply chains by transforming the production economics of 2-trifluoromethyl quinazolinones from costly, unreliable processes to a streamlined, scalable solution. The elimination of high-pressure equipment requirements and specialized handling procedures significantly reduces capital expenditure while improving operational safety, making this process accessible to a broader range of manufacturing facilities without requiring substantial infrastructure investments.
- Reduced Equipment Capital Expenditure: By operating under atmospheric pressure conditions without requiring high-pressure carbon monoxide systems, this methodology eliminates the need for expensive pressure-rated reactors and associated safety infrastructure that typically represent significant capital investments in pharmaceutical manufacturing facilities. The process utilizes standard glassware and stainless steel reactors already present in most chemical plants, reducing the initial capital outlay by approximately 35-45% compared to conventional high-pressure carbonylation processes. This equipment simplification also translates to lower maintenance costs and reduced downtime for specialized equipment validation, improving overall facility utilization rates while maintaining compliance with current Good Manufacturing Practices (cGMP). The elimination of transition metal removal steps required in alternative methodologies further reduces downstream processing costs while maintaining regulatory compliance standards required for pharmaceutical intermediates.
- Shortened Production Lead Time: The one-pot nature of this reaction sequence eliminates multiple intermediate isolation and purification steps required by traditional multi-step syntheses, reducing the overall manufacturing cycle time by approximately 40%. The simplified workup procedure involving only filtration and column chromatography further accelerates production timelines, enabling faster response to market demands and reducing inventory holding costs. This time efficiency is particularly valuable for pharmaceutical companies managing complex supply chains where lead time variability can disrupt clinical trial timelines or commercial product launches, providing greater supply chain resilience against market fluctuations and demand surges. The consistent reaction performance across different substrate types ensures predictable production scheduling without unexpected delays caused by variable yields or purification challenges common in conventional methods.
- Enhanced Raw Material Cost Efficiency: The use of commercially available nitro compounds as starting materials provides significant cost advantages over pre-functionalized substrates required by conventional methods, with raw material costs reduced by approximately 25-30% based on current market pricing. The catalytic system's efficiency (5 mol% PdCl₂) minimizes precious metal consumption while maintaining high turnover numbers, and the ability to use less expensive palladium chloride instead of more costly palladium complexes further improves cost-effectiveness. Additionally, the elimination of transition metal removal steps required in alternative methodologies reduces downstream processing costs and waste generation, contributing to both economic and environmental sustainability metrics that are increasingly important in modern pharmaceutical manufacturing. The broad substrate compatibility also allows manufacturers to leverage existing raw material inventories without requiring specialized procurement channels.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pharmaceutical Intermediate Supplier
While the advanced methodology detailed in patent CN112480015B highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
