Advanced FeCl3-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial Scale-up
Advanced FeCl3-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial Scale-up
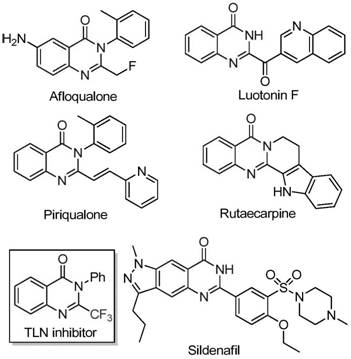
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic routes for nitrogen-containing heterocycles due to their prevalence in bioactive molecules. A significant breakthrough in this domain is documented in patent CN111675662B, which discloses a highly efficient preparation method for 2-trifluoromethyl substituted quinazolinone compounds. These scaffolds are critical in medicinal chemistry, often serving as key intermediates for drugs exhibiting anti-cancer, anticonvulsant, and anti-inflammatory activities. The introduction of the trifluoromethyl group specifically enhances electronegativity, metabolic stability, and lipophilicity, making these compounds highly desirable for drug discovery programs. This report analyzes the technical merits of this iron-catalyzed approach and its implications for reliable pharmaceutical intermediate supplier partnerships.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinazolinone derivatives bearing trifluoromethyl functional groups has relied on cyclization reactions involving specific synthons such as anthranilamide, anthranilic acid, or isatoic anhydride. While effective in laboratory settings, these conventional methodologies suffer from significant drawbacks that hinder commercial viability. Common trifluoromethylating agents like trifluoroacetic anhydride or ethyl trifluoroacetate are often expensive and require stringent handling conditions. Furthermore, traditional routes frequently necessitate severe reaction conditions, including extreme temperatures or pressures, which pose safety risks and increase energy consumption. The narrow substrate scope of many legacy methods limits the ability to generate diverse libraries of analogs, restricting the exploration of structure-activity relationships essential for modern drug development.
The Novel Approach
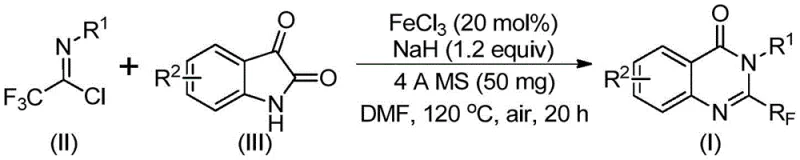
In contrast, the methodology outlined in CN111675662B offers a transformative solution by utilizing readily available trifluoroethylimidoyl chloride and isatin as starting raw materials. This novel route employs a cheap metal iron catalyst, specifically ferric chloride, to drive a series of cyclization reactions under relatively mild conditions. The process eliminates the need for precious metal catalysts, which are not only costly but also introduce challenges regarding residual metal contamination in the final active pharmaceutical ingredient. By shifting to an iron-based system, the reaction achieves high efficiency and broad applicability. The operational simplicity allows for easier post-treatment procedures, making this approach particularly attractive for cost reduction in pharmaceutical intermediate manufacturing where margin compression is a constant pressure.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The core of this technological advancement lies in the unique mechanistic pathway facilitated by the iron catalyst. During the reaction, an alkali-promoted carbon-nitrogen bond formation occurs between the trifluoroethylimidoyl chloride and the isatin substrate. This initial step generates a trifluoroacetamidine intermediate, which is crucial for the subsequent transformation. The presence of ferric chloride then catalyzes a decarbonylation and cyclization sequence. This iron-catalyzed step is pivotal as it drives the isomerization required to form the stable quinazolinone ring system. The use of sodium hydride as a base ensures the deprotonation necessary for nucleophilic attack, while the 4A molecular sieves play a critical role in sequestering moisture, thereby preventing hydrolysis of the sensitive imidoyl chloride species and driving the equilibrium towards product formation.
Impurity control is another critical aspect addressed by this mechanism. The high selectivity of the iron catalyst minimizes the formation of side products that typically plague complex heterocyclic syntheses. The reaction conditions, specifically the temperature gradient from 40°C to 120°C, allow for controlled kinetics. The initial lower temperature phase facilitates the formation of the intermediate without excessive decomposition, while the subsequent heating to 120°C ensures complete cyclization. This two-stage thermal profile is essential for maintaining high purity specifications. By avoiding harsh reagents that might degrade sensitive functional groups on the aryl rings, the process ensures that the final impurity profile is manageable, reducing the burden on downstream purification teams and ensuring consistent quality for high-purity pharmaceutical intermediates.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
Implementing this synthesis route requires precise adherence to the optimized parameters defined in the patent data. The process begins with the careful charging of ferric chloride and sodium hydride into a reaction vessel, followed by the addition of 4A molecular sieves to maintain anhydrous conditions. Trifluoroethylimidoyl chloride and isatin are then introduced into an organic solvent, with DMF being the preferred medium due to its superior solubility characteristics for these polar substrates. The reaction is initially stirred at 40°C for 8-10 hours to allow for the formation of the key intermediate. Subsequently, the temperature is raised to 120°C for 18-20 hours to complete the cyclization. Detailed standardized synthesis steps see the guide below.
- Mix ferric chloride, sodium hydride, 4A molecular sieves, trifluoroethylimidoyl chloride, and isatin in an organic solvent such as DMF.
- React the mixture at 40°C for 8-10 hours, then heat to 120°C for an additional 18-20 hours under air atmosphere.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the target compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this iron-catalyzed protocol represents a strategic opportunity to optimize the supply base for complex heterocycles. The primary driver for value creation here is the substitution of expensive noble metal catalysts with commodity-grade iron salts. This shift fundamentally alters the cost structure of the manufacturing process. Additionally, the starting materials, specifically the aromatic amines used to prepare the imidoyl chloride and the isatin derivatives, are widely available in the global chemical market. This abundance mitigates the risk of supply disruptions that often occur with specialized fluorinating agents. The robustness of the reaction conditions also implies a higher success rate in batch production, reducing the waste associated with failed runs and contributing to substantial cost savings.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts like palladium or rhodium removes the necessity for expensive heavy metal removal steps, which are often required to meet strict regulatory limits for API intermediates. This simplification of the downstream processing workflow directly translates to lower operational expenditures. Furthermore, the use of DMF as a solvent, while requiring careful handling, is economically favorable compared to specialized fluorinated solvents. The overall atom economy of the reaction is improved by the direct cyclization strategy, minimizing the generation of stoichiometric byproducts that require disposal. These factors combine to create a manufacturing process that is inherently more cost-efficient without compromising on the quality of the final output.
- Enhanced Supply Chain Reliability: The reliance on commercially available starting materials such as isatin and simple aromatic amines ensures a stable supply chain. Unlike proprietary reagents that may be sourced from a single vendor, these commodities can be procured from multiple qualified suppliers globally. This diversification reduces lead time for high-purity pharmaceutical intermediates and protects against geopolitical or logistical bottlenecks. The reaction's tolerance to air, as indicated by the procedure conducted under air atmosphere, further simplifies the engineering controls required for production. This ease of operation allows for more flexible scheduling and faster turnaround times, enabling supply chain teams to respond more agilely to fluctuating demand from R&D or commercial production units.
- Scalability and Environmental Compliance: The patent explicitly notes the potential for industrial scale application, having been validated from gram level upwards. The use of iron, a non-toxic and environmentally benign metal, aligns well with green chemistry principles and increasingly stringent environmental regulations. Waste streams generated from iron-catalyzed reactions are generally easier to treat compared to those containing heavy metals. The simplified work-up procedure, involving filtration and standard column chromatography or crystallization, facilitates scaling without the need for exotic equipment. This scalability ensures that the commercial scale-up of complex pharmaceutical intermediates can proceed smoothly from pilot plant to multi-ton production, securing long-term supply continuity for downstream drug manufacturers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their specific pipeline projects. The answers provided reflect the objective capabilities of the method as disclosed, ensuring transparency for all stakeholders involved in the sourcing and development process.
Q: What are the primary advantages of using FeCl3 over precious metal catalysts?
A: Using ferric chloride significantly reduces raw material costs compared to palladium or rhodium catalysts. It also simplifies the purification process by eliminating the need for expensive heavy metal scavenging steps, thereby enhancing overall process efficiency.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the patent explicitly states the method can be expanded from gram level to industrial scale application. The use of readily available starting materials and standard solvents like DMF supports robust commercial scale-up.
Q: What is the substrate scope for this reaction?
A: The method demonstrates good functional group tolerance. It accommodates various substituents on the aryl group of the imidoyl chloride and different positions on the isatin ring, allowing for the design of diverse quinazolinone derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in accelerating drug development timelines. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is well-versed in optimizing iron-catalyzed processes to meet stringent purity specifications required by global regulatory bodies. We operate rigorous QC labs equipped with advanced analytical instrumentation to ensure that every batch of 2-trifluoromethyl quinazolinone delivered meets the highest standards of quality and consistency. Our commitment to technical excellence ensures that your supply chain remains robust and compliant.
We invite you to collaborate with us to leverage this advanced technology for your upcoming projects. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact our technical procurement team to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a reliable supply of high-value intermediates backed by deep process knowledge and a dedication to commercial success.
