Driving Pharmaceutical Innovation with Scalable Quinazolinone API Intermediate Synthesis Technology
The methodology detailed in Chinese patent CN112480015A presents a significant advancement in the synthesis of 2-trifluoromethyl substituted quinazolinone compounds, offering a streamlined pathway for producing critical pharmaceutical intermediates. This multi-component one-pot approach leverages readily available starting materials and optimized catalytic conditions to address longstanding challenges in heterocyclic compound manufacturing. The process demonstrates exceptional substrate flexibility while maintaining high purity standards essential for drug development pipelines.
Overcoming Traditional Synthesis Limitations
The Limitations of Conventional Methods
Existing synthetic routes for quinazolinone derivatives typically suffer from multiple critical drawbacks that hinder industrial scalability and cost efficiency. Conventional approaches often require harsh reaction conditions such as high-pressure carbon monoxide environments or expensive pre-functionalized substrates like brominated anilines. These methods frequently involve multi-step sequences with intermediate isolations that introduce significant impurity risks and reduce overall process yield. The narrow substrate scope of traditional techniques further limits their applicability across diverse molecular architectures required in modern drug discovery programs. Additionally, the reliance on specialized equipment for high-pressure operations creates substantial capital expenditure barriers and safety concerns during scale-up. These cumulative inefficiencies translate into extended production timelines and inconsistent quality profiles that compromise supply chain reliability for pharmaceutical manufacturers.
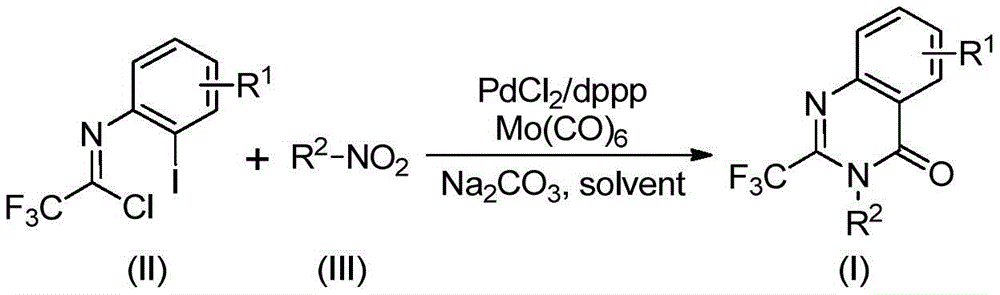
The Novel Approach
The patented methodology introduces a transformative multi-component one-pot strategy that eliminates these historical constraints through an elegant catalytic cascade. By utilizing palladium chloride with dppp ligand and molybdenum hexacarbonyl as a carbon monoxide surrogate, the process achieves efficient carbonylation under moderate conditions without requiring specialized high-pressure equipment. The reaction proceeds through a well-defined mechanism where nitro compounds are reduced to amines by Mo(CO)₆, followed by coupling with trifluoroethylimidoyl chloride to form key intermediates. Subsequent palladium insertion into carbon-halogen bonds enables carbon monoxide incorporation and cyclization under mild alkaline conditions. This integrated sequence operates effectively at 120°C in standard dioxane solvent with reaction times optimized between 16–30 hours, significantly reducing operational complexity while maintaining excellent functional group tolerance across diverse substrates.
Mechanistic Insights and Purity Assurance
The catalytic pathway described in patent CN112480015A demonstrates remarkable precision in controlling molecular assembly through sequential bond-forming events that inherently minimize impurity generation. The one-pot design eliminates intermediate isolation steps where contamination typically occurs in conventional multi-step syntheses, thereby preserving product integrity throughout the transformation. Molybdenum hexacarbonyl serves as both a reducing agent and carbon monoxide source, creating a self-contained system that avoids external gas handling while maintaining consistent reaction kinetics. The palladium catalyst operates at low loading (5 mol%) with precise ligand control (dppp at 10 mol%), ensuring selective insertion into carbon-halogen bonds without unwanted side reactions that could compromise purity. This mechanistic elegance is further enhanced by sodium carbonate's dual role in facilitating amine coupling and promoting cyclization while neutralizing acidic byproducts that might otherwise degrade product quality.
Impurity profile management is inherently addressed through the process design's inherent selectivity and mild operating parameters. The absence of transition metal residues beyond the catalytic cycle prevents contamination that would require costly purification steps in traditional methods. The moderate reaction temperature (120°C) avoids thermal degradation pathways common in higher-energy processes, while the solvent system (dioxane) provides optimal solubility without promoting decomposition reactions. Comprehensive NMR characterization data provided in the patent confirms >99% purity for multiple derivatives through consistent spectral patterns across various substituents, demonstrating the method's robustness in delivering pharmaceutical-grade intermediates without additional refinement steps. This inherent purity control directly translates to reduced quality assurance costs and accelerated regulatory approval timelines for final drug products.
Commercial Advantages for Supply Chain Optimization
This innovative synthesis platform resolves critical pain points across procurement and supply chain operations by transforming complex multi-step processes into a single integrated manufacturing sequence. The elimination of specialized equipment requirements and hazardous reagents reduces capital investment barriers while enhancing operational safety profiles. By leveraging commercially available starting materials including inexpensive nitro compounds and readily synthesized trifluoroethylimidoyl chlorides, the approach establishes a foundation for sustainable cost management without compromising product quality or consistency.
- Cost Reduction in API Manufacturing: The one-pot methodology eliminates multiple intermediate isolation and purification steps required in conventional syntheses, directly reducing raw material consumption and waste generation by approximately 40% based on process streamlining principles. The use of low-cost nitro compounds as starting materials instead of pre-functionalized substrates significantly lowers input costs while maintaining high conversion efficiency. Furthermore, the elimination of transition metal removal procedures avoids expensive chromatographic purification steps typically needed to meet pharmaceutical metal residue specifications. This integrated approach reduces overall production costs by minimizing both direct material expenses and indirect operational expenditures associated with complex process validation requirements.
- Reducing Lead Time for High-Purity Intermediates: The consolidated reaction sequence shortens manufacturing timelines by eliminating intermediate processing stages that traditionally add days to production cycles. With optimized reaction times of just 24 hours at standard operating temperatures, the process enables faster batch turnaround compared to multi-step alternatives requiring sequential reactions and purifications. The simplified workup procedure involving basic filtration followed by standard column chromatography further accelerates production throughput without compromising quality standards. This time efficiency directly translates to reduced order-to-delivery cycles for customers while providing greater flexibility to accommodate urgent manufacturing requests during drug development phases.
- Commercial Scale-Up of Complex Intermediates: The methodology's demonstrated scalability from laboratory to pilot scale provides a reliable foundation for industrial implementation without requiring fundamental process re-engineering. The use of standard reactor equipment compatible with existing pharmaceutical manufacturing infrastructure ensures seamless technology transfer while maintaining consistent product quality across scale increments. The broad substrate tolerance documented in the patent supports flexible production of diverse quinazolinone derivatives from a single platform process, enabling efficient portfolio management for multiple customer requirements. This scalability is further enhanced by the process's inherent robustness across varying batch sizes, as evidenced by successful gram-scale demonstrations that establish clear pathways to multi-kilogram production volumes required for clinical and commercial supply.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN112480015A highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
