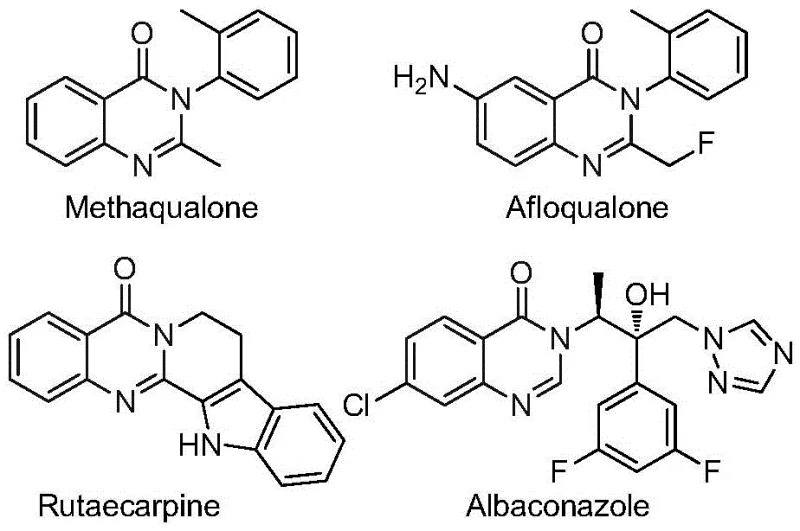
Advanced One-Pot Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial API Production
Advanced One-Pot Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial API Production
The pharmaceutical and agrochemical industries are constantly seeking robust synthetic routes for heterocyclic scaffolds that possess high biological activity, and the recent disclosure in patent CN112480015B presents a transformative approach to accessing 2-trifluoromethyl substituted quinazolinones. These nitrogen-containing heterocycles are critical structural motifs found in numerous bioactive molecules, exhibiting potent antifungal, antibacterial, antiviral, and anticancer properties, making them indispensable assets in modern drug discovery pipelines. The introduction of a trifluoromethyl group onto this scaffold is particularly strategic, as it significantly enhances metabolic stability, lipophilicity, and bioavailability of the parent molecule, thereby improving the overall pharmacokinetic profile of potential drug candidates. This patent details a novel multi-component one-pot methodology that leverages transition metal palladium catalysis to efficiently construct these complex architectures from readily available starting materials, marking a significant departure from legacy synthetic strategies that often suffer from operational complexity and safety hazards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of quinazolinone rings has relied heavily on methodologies that impose severe constraints on process chemistry and manufacturing feasibility, often requiring harsh reaction conditions that are difficult to manage on a large scale. Traditional routes frequently necessitate the use of high-pressure carbon monoxide gas, which introduces significant safety risks and requires specialized high-pressure reactor equipment that increases capital expenditure for production facilities. Furthermore, many existing protocols depend on expensive ruthenium or platinum catalysts, or require the use of pre-activated substrates such as 2-bromoformylaniline, which are not only costly to procure but also generate substantial amounts of hazardous waste during their preparation. These conventional methods are further plagued by narrow substrate scopes, meaning they often fail to tolerate diverse functional groups, limiting the chemical diversity that medicinal chemists can explore when optimizing lead compounds for biological efficacy.
The Novel Approach
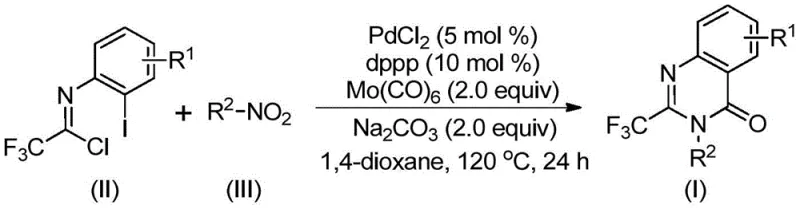
In stark contrast to these cumbersome legacy techniques, the method described in CN112480015B utilizes a streamlined one-pot cascade reaction that dramatically simplifies the synthetic workflow while maintaining high efficiency and broad applicability. By employing cheap and commercially available nitro compounds alongside trifluoroethylimidoyl chloride, this new route bypasses the need for pre-functionalized anilines or dangerous high-pressure gas cylinders, replacing them with solid molybdenum hexacarbonyl as a safe and controllable carbon monoxide surrogate. The reaction proceeds under relatively mild thermal conditions at 120°C in common organic solvents like 1,4-dioxane, utilizing a cost-effective palladium chloride catalyst system that is far more accessible than precious metal alternatives. This innovative strategy not only reduces the operational burden on laboratory personnel but also opens the door for the rapid synthesis of a wide array of substituted derivatives, facilitating faster iteration cycles in drug development programs.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cascade
The success of this transformation relies on a sophisticated catalytic cycle that seamlessly integrates nitro reduction, amidine formation, and carbonylative cyclization into a single operational sequence, driven by the synergistic action of palladium and molybdenum species. The mechanism is believed to initiate with the reduction of the nitro group on the aromatic substrate by molybdenum hexacarbonyl, which releases carbon monoxide in situ to generate the corresponding amine intermediate without the need for external reducing agents. This freshly generated amine then undergoes a base-promoted nucleophilic attack on the trifluoroethylimidoyl chloride to form a trifluoroacetamidine derivative, setting the stage for the subsequent metal-catalyzed ring closure. The palladium catalyst subsequently inserts into the carbon-iodine bond of the imidoyl chloride moiety, forming a reactive divalent palladium intermediate that captures the released carbon monoxide to create an acyl-palladium species, which ultimately undergoes intramolecular cyclization and reductive elimination to yield the target quinazolinone core.
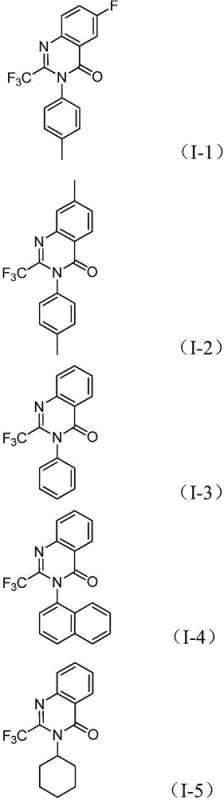
Beyond the primary catalytic cycle, the robustness of this method is attributed to its exceptional tolerance for various electronic and steric environments, ensuring high purity profiles essential for pharmaceutical applications. The use of sodium carbonate as a mild base effectively promotes the necessary deprotonation steps without causing degradation of sensitive functional groups, while the specific ligand environment provided by 1,3-bis(diphenylphosphino)propane stabilizes the palladium center against premature deactivation. This careful balance of reagents minimizes the formation of side products such as urea derivatives or hydrolysis byproducts, resulting in a cleaner crude reaction mixture that simplifies downstream purification efforts. The ability to accommodate substituents ranging from electron-withdrawing halogens to electron-donating alkyl groups on both the nitroarene and the imidoyl chloride components demonstrates the versatility of this mechanistic pathway for generating diverse chemical libraries.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
Implementing this synthesis in a practical setting requires precise attention to reagent stoichiometry and reaction parameters to maximize yield and minimize impurity formation, as outlined in the detailed experimental procedures of the patent. The process begins with the careful weighing of palladium chloride, the dppp ligand, and the carbon monoxide source, ensuring that the molar ratios are strictly maintained to support the catalytic turnover throughout the extended reaction period. Operators must ensure that the reaction vessel is properly sealed to prevent solvent loss at the elevated temperature of 120°C, although the system does not require high-pressure ratings, standard Schlenk techniques or sealed tubes are sufficient to maintain the integrity of the reaction environment. For a comprehensive guide on the exact quantities and specific workup procedures tailored to different substrate combinations, please refer to the standardized synthesis steps provided below.
- Combine palladium chloride, dppp ligand, sodium carbonate, Mo(CO)6, trifluoroethylimidoyl chloride, and nitro compound in 1,4-dioxane solvent.
- Heat the reaction mixture to 120°C and stir continuously for 16 to 30 hours to allow for carbonylation and cyclization.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target quinazolinone.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers profound advantages for procurement managers and supply chain directors who are tasked with securing reliable sources of high-quality intermediates while managing budgetary constraints. The shift away from high-pressure gas infrastructure and expensive noble metal catalysts translates directly into reduced capital and operational expenditures, making the production of these valuable heterocycles more economically viable for large-scale manufacturing. Furthermore, the reliance on commodity chemicals like nitro compounds and simple imidoyl chlorides ensures a stable and resilient supply chain, as these materials are widely produced by multiple global vendors, mitigating the risk of shortages that can plague specialized reagent markets. The operational simplicity of the one-pot design also reduces the labor hours required for synthesis and purification, allowing manufacturing teams to allocate resources more efficiently across other critical projects.
- Cost Reduction in Manufacturing: The elimination of high-pressure carbon monoxide equipment and the substitution of expensive ruthenium catalysts with affordable palladium chloride significantly lowers the barrier to entry for producing these intermediates. By utilizing solid Mo(CO)6 as a CO source, the process avoids the logistical complexities and safety costs associated with handling toxic gases, leading to substantial savings in facility maintenance and insurance premiums. Additionally, the high conversion rates observed across various substrates mean that less raw material is wasted, further driving down the cost per kilogram of the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: Sourcing strategies are greatly improved by the use of universally available starting materials such as substituted nitrobenzenes, which are staple products in the fine chemical industry with established global supply networks. This reduces dependency on single-source suppliers for exotic reagents and minimizes the lead time variability that often disrupts production schedules in the pharmaceutical sector. The robustness of the reaction conditions also means that the process is less susceptible to minor fluctuations in raw material quality, ensuring consistent output even when sourcing from different batches or vendors.
- Scalability and Environmental Compliance: The demonstrated ability to scale this reaction to gram levels with simple filtration and chromatography workups indicates a clear path toward ton-scale production without requiring complex engineering modifications. The use of 1,4-dioxane, while requiring proper handling, is a well-understood solvent in industrial settings with established recovery and recycling protocols that align with modern environmental regulations. Moreover, the atom economy of the multi-component reaction is superior to stepwise approaches, generating less chemical waste and reducing the burden on wastewater treatment facilities, which is a critical factor for maintaining sustainable manufacturing certifications.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of adopting this synthesis route for their specific projects, we have compiled answers to the most common inquiries regarding reaction scope, safety, and scalability based on the patent data. Understanding the nuances of the catalytic system and the compatibility of various functional groups is essential for successful technology transfer from the laboratory to the pilot plant. The following responses address key concerns related to process optimization and regulatory compliance, providing a solid foundation for decision-making regarding the integration of this technology into existing production workflows.
Q: What are the primary advantages of this one-pot method over traditional quinazolinone synthesis?
A: This method eliminates the need for high-pressure carbon monoxide gas and expensive pre-activated substrates, utilizing cheap nitro compounds and solid Mo(CO)6 instead, which drastically simplifies operational safety and cost.
Q: Is this process scalable for industrial production of pharmaceutical intermediates?
A: Yes, the patent explicitly demonstrates scalability to the gram level with simple post-treatment procedures like filtration and column chromatography, indicating strong potential for kilogram-to-ton scale-up in CDMO settings.
Q: What is the substrate scope for the nitro compound component in this reaction?
A: The reaction exhibits excellent functional group tolerance, accommodating various substituents such as halogens, alkyl groups, and trifluoromethyl groups on the aromatic ring, allowing for diverse library synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
As a leader in the custom synthesis and contract development sector, NINGBO INNO PHARMCHEM possesses the technical expertise and infrastructure necessary to bring this advanced quinazolinone synthesis from patent literature to commercial reality. Our team of seasoned chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to full-scale manufacturing is seamless and efficient. We operate stringent purity specifications and utilize rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 2-trifluoromethyl quinazolinone meets the exacting standards required for clinical and commercial applications. Our commitment to quality assurance means that clients can rely on us for consistent supply continuity, minimizing the risk of production delays due to quality failures.
We invite pharmaceutical and agrochemical companies to leverage our capabilities for the cost-effective production of these high-value intermediates, enabling faster time-to-market for your novel therapeutic candidates. By partnering with us, you gain access to a Customized Cost-Saving Analysis that evaluates how this specific palladium-catalyzed route can optimize your current supply chain economics compared to traditional methods. We encourage you to contact our technical procurement team today to request specific COA data for similar compounds and to discuss route feasibility assessments tailored to your unique molecular targets, ensuring a strategic advantage in your drug development pipeline.
