Advanced Palladium-Catalyzed Synthesis of Substituted 2,3-Dihydroquinolone Intermediates for Commercial Scale-Up
Advanced Palladium-Catalyzed Synthesis of Substituted 2,3-Dihydroquinolone Intermediates for Commercial Scale-Up
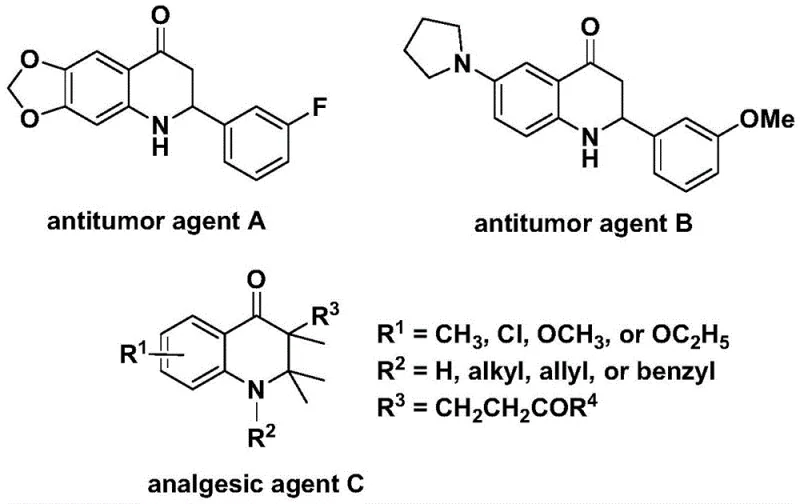
The development of efficient synthetic routes for nitrogen-containing heterocycles remains a cornerstone of modern medicinal chemistry, particularly for scaffolds exhibiting potent biological activities. Patent CN112239456A discloses a robust preparation method for substituted 2,3-dihydroquinolone compounds, a structural motif prevalent in numerous bioactive molecules ranging from antitumor agents to analgesics. This technology leverages a transition metal palladium-catalyzed carbonylation reaction, utilizing N-pyridine sulfonyl-o-iodoaniline and olefins as key starting materials. By employing a solid carbon monoxide substitute rather than toxic CO gas, the process addresses significant safety and scalability concerns inherent in traditional carbonylation methods. The methodology offers a versatile platform for generating diverse 2-aryl and 3-alkyl substituted derivatives, making it an invaluable asset for the production of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 2,3-dihydroquinolone skeleton has relied heavily on classical cyclization strategies or direct carbonylation using carbon monoxide gas. While effective in laboratory settings, these conventional approaches present substantial hurdles for industrial application. The use of gaseous CO requires specialized high-pressure equipment and rigorous safety protocols due to its extreme toxicity and flammability, which drastically increases capital expenditure and operational complexity. Furthermore, traditional methods often suffer from limited substrate scope, struggling to accommodate sensitive functional groups or sterically hindered olefins without compromising yield. The reliance on harsh reaction conditions can also lead to the formation of complex impurity profiles, necessitating costly and time-consuming purification steps that erode overall process efficiency and economic viability for large-scale manufacturing.
The Novel Approach
The innovative strategy outlined in the patent data circumvents these challenges by introducing a palladium-catalyzed system that utilizes TFBen as a safe, solid carbon monoxide surrogate. This approach allows the reaction to proceed under relatively mild thermal conditions (110 °C) in standard atmospheric pressure vessels, effectively democratizing access to carbonylation chemistry for broader manufacturing facilities. The method exhibits exceptional functional group tolerance, successfully integrating various aryl and alkyl substituents without the need for protective group manipulations. By streamlining the reaction setup and eliminating the hazards associated with gas handling, this novel route not only enhances operator safety but also significantly reduces the barrier to entry for cost reduction in pharmaceutical intermediate manufacturing. The versatility of this system enables the rapid generation of diverse chemical libraries, accelerating the drug discovery pipeline.
Mechanistic Insights into Palladium-Catalyzed Carbonylation
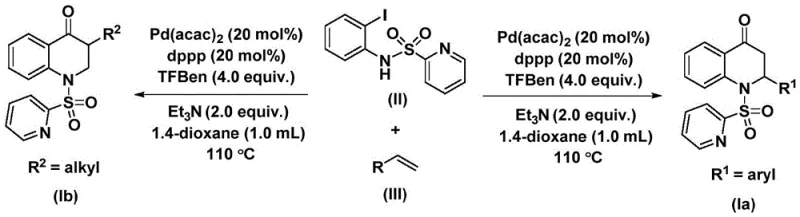
The catalytic cycle driving this transformation is a sophisticated orchestration of organometallic steps initiated by the oxidative addition of the palladium catalyst into the carbon-iodine bond of the N-pyridine sulfonyl-o-iodoaniline substrate. This critical step generates a reactive aryl-palladium intermediate, which serves as the foundation for subsequent bond-forming events. Following oxidative addition, the carbon monoxide molecule, released in situ from the decomposition of the TFBen additive, inserts into the palladium-carbon bond to form an acyl-palladium species. This insertion is the key carbonylation event that introduces the ketone functionality essential to the quinolone core. The precision of this step ensures high regioselectivity and minimizes side reactions such as homocoupling, which are common pitfalls in palladium chemistry.
Subsequent coordination and migratory insertion of the olefin substrate into the acyl-palladium bond generate a new alkyl-palladium intermediate, setting the stage for ring closure. The final step involves an intramolecular reductive elimination that forms the carbon-nitrogen bond, releasing the substituted 2,3-dihydroquinolone product and regenerating the active palladium(0) catalyst to continue the cycle. This mechanistic pathway is highly efficient, as evidenced by the high yields observed across a wide range of substrates, including those with electron-withdrawing and electron-donating groups. The use of the dppp ligand is crucial in stabilizing the palladium center throughout these transitions, preventing catalyst deactivation and ensuring consistent performance over the 48-hour reaction duration, thereby guaranteeing a clean impurity profile suitable for stringent pharmaceutical specifications.
How to Synthesize Substituted 2,3-Dihydroquinolone Efficiently
The practical execution of this synthesis is designed for reproducibility and ease of operation, making it ideal for both process development and production environments. The protocol involves charging a reaction vessel with the palladium catalyst, ligand, base, CO surrogate, and substrates in an aprotic solvent like 1,4-dioxane. The detailed standardized synthesis steps for implementing this high-efficiency route are provided in the guide below.
- Combine Pd(acac)2 catalyst, dppp ligand, TFBen, triethylamine, N-pyridine sulfonyl-o-iodoaniline, and olefin in 1,4-dioxane solvent.
- Heat the reaction mixture to 110 °C and stir for 48 hours under inert atmosphere to facilitate carbonylation and cyclization.
- Filter the reaction mixture, adsorb onto silica gel, and purify via column chromatography to isolate the target 2,3-dihydroquinolone product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology translates into tangible strategic benefits regarding cost structure and supply reliability. The shift away from hazardous gas infrastructure to a solution-phase reaction using solid reagents simplifies the supply chain logistics and reduces the regulatory burden associated with storing and transporting toxic gases. This operational simplification directly correlates to lower overhead costs and reduced risk of production stoppages due to safety audits or equipment failures. Furthermore, the high substrate compatibility means that a single manufacturing line can be utilized to produce a wide variety of analogues, maximizing asset utilization and flexibility in responding to market demands for different API intermediates.
- Cost Reduction in Manufacturing: The elimination of high-pressure CO gas infrastructure represents a significant capital saving, as facilities do not require specialized autoclaves or gas detection systems. Additionally, the use of commercially available starting materials like olefins and iodoanilines, combined with a catalyst system that operates efficiently at standard loading levels, drives down the raw material cost per kilogram. The high conversion rates and clean reaction profiles minimize the loss of valuable intermediates during purification, leading to substantial cost savings in downstream processing and waste disposal.
- Enhanced Supply Chain Reliability: By utilizing stable, solid reagents such as TFBen instead of compressed gases, the risk of supply disruption due to transportation restrictions or storage limitations is drastically minimized. The robustness of the reaction conditions (110 °C, 48 hours) ensures consistent batch-to-batch quality, reducing the likelihood of failed batches that could delay delivery timelines. This reliability is critical for maintaining continuous production schedules for key pharmaceutical intermediates, ensuring that downstream API manufacturing is never compromised by upstream supply volatility.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work effectively from gram to potential multi-kilogram scales without significant re-optimization. The use of 1,4-dioxane, while requiring careful handling, is a well-understood solvent in the industry with established recovery and recycling protocols, supporting green chemistry initiatives. The high atom economy of the carbonylation step and the avoidance of stoichiometric heavy metal oxidants contribute to a reduced environmental footprint, facilitating easier compliance with increasingly strict global environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis route. These insights are derived directly from the experimental data and mechanistic understanding provided in the patent documentation, ensuring that potential partners have a clear view of the technology's capabilities and limitations.
Q: What is the primary advantage of using TFBen in this synthesis?
A: TFBen serves as a solid carbon monoxide substitute, eliminating the need for handling hazardous high-pressure CO gas, thereby significantly enhancing operational safety and simplifying reactor requirements for industrial scale-up.
Q: What types of substituents are tolerated on the olefin substrate?
A: The process demonstrates excellent substrate compatibility, successfully accommodating both aryl groups (with methyl, chloro, fluoro, or methoxy substituents) and alkyl groups (linear, branched, or silyl-substituted), allowing for diverse library synthesis.
Q: What are the typical reaction conditions for this transformation?
A: Optimal results are achieved using Pd(acac)2 (20 mol%) and dppp (20 mol%) in 1,4-dioxane at 110 °C for 48 hours, yielding products with high conversion rates up to 88%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Substituted 2,3-Dihydroquinolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug development and securing supply chains. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries like this palladium-catalyzed carbonylation can be seamlessly transitioned to industrial reality. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our expertise in managing complex catalytic cycles allows us to optimize these processes for maximum yield and minimal impurity generation.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific project needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your supply chain resilience and drive down your overall production costs.
