Advanced Synthesis of Geminal Dinitro Compounds: Scalable Production for Pharmaceutical Intermediates Market
The Chinese patent CN109574913B introduces a groundbreaking methodology for synthesizing geminal dinitro compounds through a novel nitration-cyclization-oxidation sequence utilizing nitrate hydrates as sustainable nitro sources. This innovative approach represents a significant advancement over conventional techniques that typically require harsh alkaline conditions or concentrated nitric acid solvents, which present substantial environmental and safety challenges. The patent demonstrates how iron(III) nitrate nonahydrate [Fe(NO3)3·9H2O] serves as an exceptionally effective and economical nitro source when combined with specific additives in organic solvents under mild reaction conditions. This development holds particular importance given the growing pharmaceutical interest in geminal dinitro compounds due to their demonstrated nitric oxide release capabilities and potential therapeutic applications as antihypertensive agents. The methodology's simplicity, safety profile, and scalability position it as a highly attractive option for manufacturers seeking reliable [precise industry noun] supplier capabilities in the specialty chemical sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing geminal dinitro compounds typically involve oxidative nitration of nitroalkanes under strongly alkaline conditions, which presents multiple significant challenges for industrial implementation. These methods require starting from pre-existing nitro compounds rather than simpler precursors, necessitating additional synthetic steps that increase overall process complexity and cost. The substantial quantities of strong bases required create serious environmental concerns due to difficult waste stream management and neutralization requirements. Furthermore, these conventional processes often generate significant impurities that complicate downstream purification and compromise final product quality, particularly problematic for pharmaceutical applications where stringent purity specifications must be met. Another common approach involves sequential nitrosation-oxidation of oximes using large volumes of concentrated nitric acid as solvent, which is both economically inefficient and poses serious safety hazards due to the highly corrosive nature of concentrated acids at elevated temperatures. These limitations collectively restrict the commercial viability and scalability of traditional methods for producing high-purity geminal dinitro compounds needed for advanced pharmaceutical development.
The Novel Approach
The patented methodology overcomes these limitations through an elegant process that directly converts readily available 1,6-diynes into geminal dinitro compounds using nitrate hydrates as the primary nitro source under remarkably mild conditions. This innovative approach eliminates the need for strong bases or concentrated acids while maintaining excellent reaction efficiency and product quality. The process operates effectively at temperatures ranging from room temperature to 60°C in standard organic solvents such as acetonitrile or nitromethane, significantly improving operational safety compared to conventional high-temperature or strongly acidic processes. By utilizing iron(III) nitrate nonahydrate as the preferred nitro source with carefully selected additives like N-oxide pyridine or p-benzoquinone, the method achieves excellent yields while minimizing unwanted side reactions that typically generate impurities. This streamlined approach not only reduces environmental impact but also simplifies manufacturing workflows, making it particularly suitable for commercial scale-up of complex [precise industry noun] while maintaining high-purity standards required by pharmaceutical clients.
Mechanistic Insights into Fe(NO3)3-Catalyzed Cyclization
The fundamental mechanism underlying this innovative synthesis involves a sophisticated sequence where nitrate hydrate decomposes to generate nitro radicals that selectively react with the alkyne moieties in 1,6-diyne substrates. This radical-based process initiates with homolytic cleavage of the nitrate species under mild thermal conditions, producing reactive nitrogen dioxide radicals that add across the triple bonds in a regioselective manner. The resulting radical intermediates then undergo intramolecular cyclization followed by oxidation steps to form the characteristic geminal dinitro functionality within a six-membered ring structure. This cascade reaction is facilitated by carefully selected additives that modulate radical reactivity and prevent undesired side reactions, ensuring high selectivity toward the target products. The mechanism operates efficiently at ambient temperatures due to the favorable thermodynamics of radical addition to alkynes when properly catalyzed by transition metal nitrates.
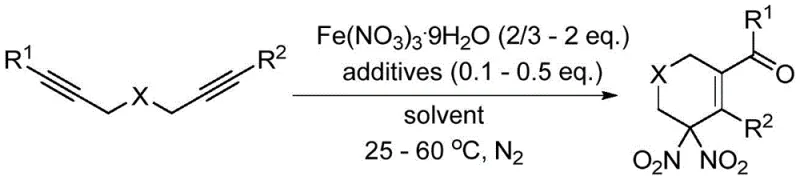
The process demonstrates exceptional control over impurity formation through its carefully balanced reaction parameters and additive selection. By operating at moderate temperatures between 25-60°C with precise stoichiometric control of both the nitrate hydrate (2/3-2 equivalents) and additives (0.1-0.5 equivalents), the method minimizes competing side reactions that typically generate unwanted byproducts in conventional syntheses. The use of oxygen-containing additives such as N-methylmorpholine-N-oxide or p-benzoquinone serves dual purposes: they facilitate the oxidation step while also acting as radical traps that prevent over-nitration or decomposition pathways. This precise mechanistic control results in products with significantly improved purity profiles compared to traditional methods, making them particularly suitable for pharmaceutical applications where impurity levels must be strictly controlled to meet regulatory requirements for drug substances.
How to Synthesize Geminal Dinitro Compounds Efficiently
This patented methodology provides a robust framework for synthesizing high-purity geminal dinitro compounds through a carefully optimized sequence that begins with substrate preparation and concludes with product isolation. The process leverages readily available starting materials including variously substituted 1,6-diynes that can be tailored to produce diverse product libraries meeting specific pharmaceutical requirements. By following precise stoichiometric ratios between substrate, iron(III) nitrate nonahydrate (typically at 2/3 to 2 equivalents), and selected additives (0.1 to 0.5 equivalents), manufacturers can achieve consistent results across different production scales. The reaction proceeds efficiently in common organic solvents such as acetonitrile or nitromethane under nitrogen atmosphere at temperatures ranging from room temperature to 60°C, depending on substrate reactivity. Detailed standardized synthesis steps are provided below to ensure successful implementation of this innovative methodology.
- Prepare reaction mixture with 1,6-diyne substrate, nitrate hydrate (2/3-2 eq.), and additives (0.1-0.5 eq.) in organic solvent under nitrogen atmosphere
- Maintain reaction temperature between 25-60°C for optimal yield while monitoring reaction progress
- Perform post-reaction processing including filtration, solvent removal, and column chromatography purification
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology delivers substantial commercial benefits that directly address critical pain points faced by procurement and supply chain professionals in specialty chemical manufacturing. The elimination of hazardous reagents and extreme reaction conditions significantly reduces both operational risks and associated compliance costs throughout the manufacturing process. By utilizing readily available iron(III) nitrate nonahydrate instead of expensive or difficult-to-handle nitrating agents, manufacturers can achieve substantial cost savings while improving workplace safety profiles. The simplified process flow reduces equipment requirements and maintenance costs compared to traditional high-pressure or high-temperature systems needed for conventional syntheses. These advantages collectively enhance supply chain resilience by creating more reliable production pathways less susceptible to raw material shortages or regulatory restrictions.
- Cost Reduction in Manufacturing: The substitution of expensive transition metal catalysts with iron-based nitrates provides significant raw material cost savings while eliminating complex catalyst removal steps required in alternative methodologies. The use of ambient temperature processing reduces energy consumption substantially compared to high-temperature alternatives, while simplified workup procedures minimize solvent usage and waste generation. This streamlined approach creates multiple avenues for cost optimization without compromising product quality or yield consistency.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials including common organic solvents and iron-based nitrates creates more resilient supply chains less vulnerable to single-source dependencies or geopolitical disruptions. The elimination of hazardous reagents reduces transportation restrictions and storage requirements, enabling more flexible manufacturing scheduling and inventory management practices. This enhanced supply chain flexibility directly translates to improved on-time delivery performance while maintaining consistent quality standards across production batches.
- Scalability and Environmental Compliance: The mild reaction conditions enable straightforward scale-up from laboratory to commercial production without requiring specialized equipment modifications typically needed for high-pressure or cryogenic processes. The reduced environmental footprint through minimized waste generation and elimination of hazardous reagents aligns with increasingly stringent global regulations while supporting corporate sustainability initiatives. This environmentally responsible approach facilitates regulatory approval processes and enhances market positioning as a forward-thinking supplier committed to sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common concerns raised by technical procurement teams regarding implementation of this innovative synthesis methodology. These answers are based on detailed analysis of the patent's technical disclosures and experimental results demonstrating process robustness across diverse substrate types.
Q: How does this method improve upon traditional approaches for synthesizing geminal dinitro compounds?
A: This method eliminates the need for strong alkaline conditions or concentrated nitric acid solvents required by conventional methods. The use of nitrate hydrate as a nitro source provides a safer, more environmentally friendly process with simpler operation and better scalability while maintaining high product purity essential for pharmaceutical applications.
Q: What are the key advantages of using nitrate hydrate as a nitro source in this synthesis?
A: Nitrate hydrate is significantly cheaper and more readily available than traditional nitration reagents. It provides a stable nitro source that enables milder reaction conditions (25-60°C) without requiring hazardous strong acids or bases, resulting in improved safety profile and reduced environmental impact while maintaining excellent product quality.
Q: How does this process ensure high purity of the final geminal dinitro compounds?
A: The controlled reaction mechanism using nitrate hydrate minimizes side reactions that typically produce impurities in conventional methods. The specific combination of additives and mild conditions allows for selective formation of the desired geminal dinitro products with excellent purity profiles suitable for pharmaceutical applications requiring stringent quality standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Geminal Dinitro Compounds Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through rigorous QC labs dedicated to specialty chemical manufacturing. We have successfully implemented this patented methodology across multiple production scales while ensuring consistent product quality that meets exacting pharmaceutical industry standards. Our technical team possesses deep expertise in optimizing reaction parameters for specific substrate requirements while maintaining cost-effective production economics through continuous process improvement initiatives.
We invite you to request our Customized Cost-Saving Analysis tailored specifically to your production needs by contacting our technical procurement team directly. They will provide comprehensive information including specific COA data for relevant product variants along with detailed route feasibility assessments demonstrating how our manufacturing capabilities can support your supply chain requirements while delivering significant value through optimized production economics.
