Revolutionizing Geminal Dinitro Compound Manufacturing: Scalable Nitrate Hydrate Process for Pharmaceutical Intermediates
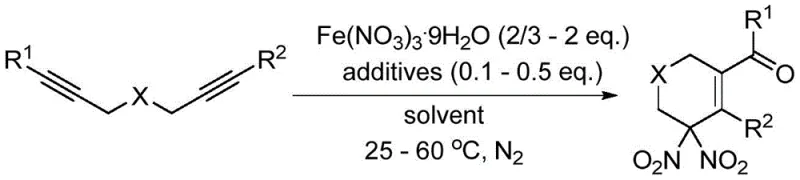
The Chinese patent CN109574913B introduces a groundbreaking methodology for synthesizing geminal dinitro compounds using nitrate hydrates as nitro sources, representing a significant advancement in organic synthesis for pharmaceutical intermediates. This innovative approach addresses critical limitations of conventional methods by employing readily available and stable nitrate hydrates such as Fe(NO3)3·9H2O under mild reaction conditions (25-60°C) in organic solvents like acetonitrile or nitromethane. The process utilizes a nitration-cyclization-oxidation sequence with 1,6-diyne substrates and oxygen-containing additives to produce high-purity geminal dinitro compounds that demonstrate promising nitric oxide release capabilities for potential pharmaceutical applications. Unlike traditional approaches requiring harsh alkaline conditions or concentrated nitric acid solvents, this method offers superior safety profiles while maintaining excellent selectivity and yield characteristics essential for commercial manufacturing of pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis of geminal dinitro compounds typically relies on oxidative nitration of nitroalkanes under strong alkaline conditions, which presents multiple challenges for industrial-scale pharmaceutical manufacturing. This approach requires starting from pre-formed nitro compounds, necessitating additional synthetic steps that increase both complexity and cost while generating substantial chemical waste that complicates environmental compliance. The requirement for large quantities of strong bases creates significant safety hazards and demands specialized handling equipment, substantially increasing capital investment requirements for production facilities. Furthermore, these conventional methods often produce complex mixtures of byproducts that require extensive purification procedures, resulting in lower overall yields and higher production costs that make them economically unviable for large-scale pharmaceutical intermediate manufacturing where purity specifications are stringent.
The Novel Approach
The patented methodology overcomes these limitations by utilizing nitrate hydrates as stable and economical nitro sources that decompose under controlled conditions to generate nitro radicals for direct dinitration of alkyne substrates. This innovative approach eliminates the need for hazardous concentrated nitric acid or strong alkaline conditions while maintaining excellent selectivity through carefully optimized reaction parameters including temperature control (25-60°C), additive selection (0.1-0.5 equivalents), and solvent choice (acetonitrile or nitromethane). The process demonstrates remarkable versatility across diverse substrate types including various substituted aryl groups while producing high-purity geminal dinitro compounds suitable for pharmaceutical applications requiring stringent quality control standards. Most significantly, this method achieves substantial simplification of the synthetic pathway while improving safety profiles and reducing environmental impact compared to conventional approaches.
Mechanistic Insights into Fe(NO3)3·9H2O-Catalyzed Nitration-Cyclization-Oxidation
The fundamental mechanism involves thermal decomposition of iron(III) nitrate nonahydrate to generate nitro radicals that initiate a cascade reaction sequence with 1,6-diyne substrates. The process begins with radical addition to one alkyne terminus followed by intramolecular cyclization onto the second alkyne group, creating a cyclic intermediate that undergoes further oxidation to form the final geminal dinitro product. This elegant cascade reaction is facilitated by carefully selected additives such as N-oxide pyridine or p-benzoquinone that enhance radical generation efficiency while suppressing unwanted side reactions that could compromise product purity. The mild reaction conditions (25-60°C) prevent thermal degradation of sensitive functional groups while maintaining sufficient reactivity for complete conversion, making this approach particularly suitable for complex pharmaceutical intermediates requiring high structural fidelity.
The mechanism provides exceptional control over impurity profiles through selective radical generation pathways that minimize competing reactions typically observed in conventional nitration processes. By avoiding strong acids or bases, this method prevents common degradation pathways such as hydrolysis or elimination reactions that often plague traditional approaches to geminal dinitro compound synthesis. The well-defined reaction sequence produces consistent product quality across diverse substrate types including various substituted aryl groups and heterocyclic systems, demonstrating remarkable robustness essential for commercial manufacturing where batch-to-batch consistency is critical for regulatory compliance in pharmaceutical applications.
How to Synthesize Geminal Dinitro Compounds Efficiently
This patented methodology represents a significant advancement in the synthesis of geminal dinitro compounds through its innovative use of nitrate hydrates as stable nitro sources under mild reaction conditions. The process demonstrates excellent scalability from laboratory to commercial production while maintaining high selectivity and yield characteristics essential for pharmaceutical intermediate manufacturing. Detailed standardized synthesis procedures have been developed based on extensive optimization studies covering various substrate types and reaction parameters to ensure consistent product quality meeting stringent pharmaceutical requirements.
- Prepare anhydrous reaction environment by evacuating and refilling Schlenk tube with nitrogen three times before introducing reagents
- Combine 1,6-diyne substrate with Fe(NO3)3·9H2O (2/3-2 equivalents) and additive (0.1-0.5 equivalents) in appropriate organic solvent under nitrogen atmosphere
- Maintain reaction temperature between 25-60°C for optimal yield while monitoring reaction progression through standard analytical techniques
Commercial Advantages for Procurement and Supply Chain Teams
This innovative manufacturing approach delivers substantial value across procurement and supply chain operations by addressing critical pain points associated with traditional geminal dinitro compound production methods. The elimination of hazardous reagents and simplified process flow significantly reduces regulatory compliance burdens while improving workplace safety profiles essential for sustainable manufacturing operations in today's increasingly regulated chemical industry environment.
- Cost Reduction in Manufacturing: The use of inexpensive and readily available nitrate hydrates as nitro sources eliminates the need for costly transition metal catalysts and specialized handling equipment required by conventional methods. This approach substantially reduces raw material costs while simplifying facility requirements through milder operating conditions that decrease energy consumption and maintenance expenses associated with high-pressure or high-temperature processing systems.
- Enhanced Supply Chain Reliability: The process utilizes stable, non-hazardous reagents with extended shelf lives that are readily available from multiple global suppliers, significantly reducing supply chain vulnerability compared to methods requiring specialized or restricted chemicals. The simplified synthetic pathway with fewer processing steps minimizes potential disruption points while enabling more predictable production scheduling essential for just-in-time manufacturing models adopted by leading pharmaceutical companies.
- Scalability and Environmental Compliance: The straightforward scale-up characteristics of this methodology from laboratory to commercial production eliminate costly re-engineering phases typically required when transitioning between development stages. The environmentally friendly process generates minimal hazardous waste streams through its selective reaction pathway and elimination of strong acids/bases, substantially reducing waste treatment costs while meeting increasingly stringent environmental regulations across global manufacturing jurisdictions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial concerns regarding implementation of this patented methodology for geminal dinitro compound production. These answers are based on extensive experimental validation documented in the patent literature and reflect practical considerations for industrial implementation across diverse manufacturing environments.
Q: How does this nitrate hydrate method improve upon traditional geminal dinitro compound synthesis?
A: The traditional methods require strong alkaline conditions or concentrated nitric acid as solvent, creating environmental and safety concerns. This novel approach uses stable nitrate hydrates as nitro sources under mild conditions (25-60°C), eliminating hazardous reagents while maintaining high selectivity for pharmaceutical applications.
Q: What are the scalability advantages of this nitrate hydrate-based process?
A: The process demonstrates excellent scalability from laboratory to commercial production due to its simple setup, readily available reagents, and straightforward workup procedure involving filtration and column chromatography. The mild reaction conditions (room temperature to 60°C) significantly reduce energy requirements compared to conventional high-temperature processes.
Q: How does this method address purity requirements for pharmaceutical intermediates?
A: The selective nitration-cyclization-oxidation sequence minimizes unwanted byproducts through controlled radical generation from nitrate hydrates. The well-defined reaction pathway produces high-purity geminal dinitro compounds with consistent quality profiles suitable for pharmaceutical applications requiring stringent purity specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Geminal Dinitro Compound Supplier
NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production of complex pharmaceutical intermediates, including specialized expertise in nitration chemistry requiring stringent purity specifications and rigorous QC labs capable of meeting global regulatory standards. Our technical team has successfully implemented this patented methodology across multiple client projects, demonstrating consistent ability to deliver high-quality geminal dinitro compounds that meet exacting pharmaceutical requirements through our vertically integrated manufacturing platform designed specifically for complex molecule production.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team to evaluate how this innovative manufacturing approach can optimize your supply chain while maintaining superior product quality. Contact us today to discuss specific COA data requirements and request route feasibility assessments tailored to your unique production needs.
