Advanced Synthetic Strategy for Clausenamide: Scalable Production for Pharmaceutical Applications
Advanced Synthetic Strategy for Clausenamide: Scalable Production for Pharmaceutical Applications
The pharmaceutical industry continuously seeks robust and scalable methods for producing bioactive natural products, and the synthesis of clausenamide represents a significant breakthrough in this domain. As detailed in patent CN1015710B, a novel synthetic route has been developed that overcomes the severe limitations of traditional extraction methods. This patent describes a highly efficient preparation method for (±)3(S*), 4(R*), 5(R*), 7(S*)-3-hydroxy-5-α-hydroxybenzyl-1-methyl-4-phenyl-pyrrolidin-2-one, commonly known as clausenamide. The innovation lies in a stereoselective oxidation process that ensures the formation of the biologically active C3-C4 trans configuration with high fidelity. For R&D directors and procurement specialists, this technology offers a viable pathway to secure high-purity API intermediates without the supply chain volatility associated with botanical sourcing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the acquisition of clausenamide relied heavily on extraction from the leaves of Clausena plants, a method fraught with inefficiency and economic instability. The patent data highlights a critical bottleneck: extracting merely 1.5 grams of clausenamide requires processing approximately 4 kilograms of dried leaves. This abysmal yield renders plant extraction commercially unviable for large-scale pharmacological trials or mass market production. Furthermore, botanical extracts are inherently complex matrices containing numerous secondary metabolites that are structurally similar to the target compound, making purification arduous and costly. The reliance on agricultural cycles introduces significant supply chain risks, where crop failure or seasonal variations can disrupt the availability of raw materials, leading to unpredictable lead times and price fluctuations for downstream manufacturers.
The Novel Approach
In stark contrast, the synthetic methodology outlined in the patent provides a deterministic and controllable alternative to nature-dependent extraction. By constructing the pyrrolidinone core through a series of well-defined organic transformations, manufacturers can achieve consistent quality and quantity regardless of external environmental factors. The core of this innovation is the ability to synthesize the key intermediate, formula II, and subsequently oxidize it to the final product, formula I, with precise stereochemical control. This approach not only bypasses the low-yield extraction bottleneck but also simplifies the purification process by avoiding the co-extraction of unrelated plant actives. The result is a streamlined production workflow that significantly enhances the reliability of the supply chain for this valuable hepatoprotective agent.
Mechanistic Insights into Stereoselective Oxidation and Grignard Addition
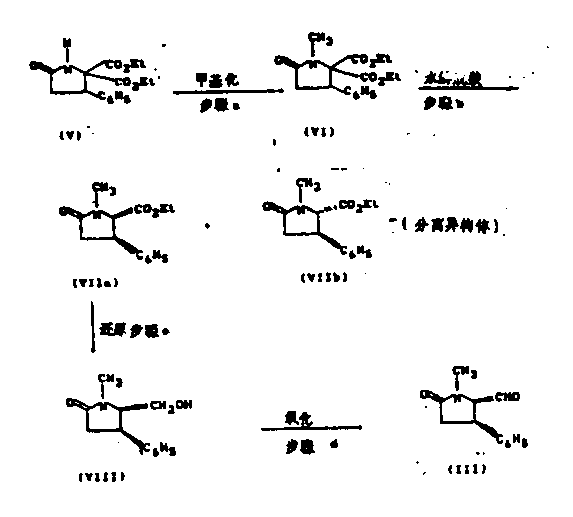
The chemical elegance of this process is best understood through its critical mechanistic steps, particularly the final oxidation and the preceding Grignard addition. The synthesis begins with the construction of the pyrrolidinone scaffold, where stereochemistry is carefully managed. A pivotal moment occurs during the introduction of the hydroxybenzyl group at the C5 position. The patent describes reacting a formyl-pyrrolidinone intermediate with organometallic reagents, such as phenylmagnesium bromide. While this step can initially produce the incorrect 7(R*) configuration (Formula IIa), the process includes a robust isomerization or oxidation-reduction sequence to ensure the desired 7(S*) configuration is achieved. This flexibility in handling stereoisomers demonstrates the resilience of the synthetic route against minor variations in reaction conditions.
The culmination of the synthesis is the oxidation of the 5-α-hydroxybenzyl-1-methyl-4-phenylpyrrolidin-2-one (Formula II) to the final clausenamide (Formula I). This transformation is achieved by generating an enolate salt using strong bases like lithium diisopropylamide (LDA) in inert solvents such as tetrahydrofuran or hexamethylphosphoric triamide. Subsequent exposure to dry oxygen, often in the presence of a phosphite auxiliary like triethyl phosphite, facilitates the insertion of the hydroxyl group at the C3 position. Crucially, this reaction conditions favor the formation of the C3-C4 trans configuration exclusively. This high degree of stereocontrol is surprising and advantageous, as it minimizes the formation of diastereomeric impurities that would otherwise require resource-intensive chromatographic separation, thereby directly impacting the cost of goods sold.
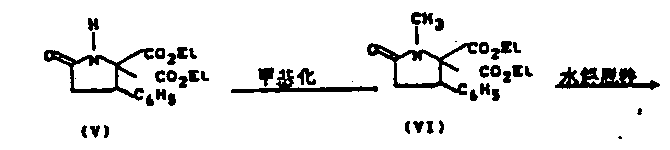
How to Synthesize Clausenamide Efficiently
Executing this synthesis requires strict adherence to anhydrous conditions and precise temperature control, particularly during the enolization and oxidation phases. The process is designed to be modular, allowing for the isolation and characterization of key intermediates such as the 5-formyl and 5-benzoyl derivatives. This modularity is beneficial for quality control, as each stage can be verified before proceeding to the next, ensuring that any deviations are caught early. The use of common laboratory reagents like iodomethane for N-methylation and standard reducing agents like lithium triethylborohydride ensures that the barrier to entry for manufacturing is low. For a detailed breakdown of the operational parameters, stoichiometry, and workup procedures required to implement this route in a GMP environment, please refer to the standardized guide below.
- Methylation of 5,5-diethoxycarbonyl-4-phenylpyrrolidin-2-one using methyl iodide and sodium hydride to form the N-methyl derivative.
- Hydrolysis and decarboxylation followed by reduction to generate the 5-hydroxymethyl intermediate.
- Oxidation of the hydroxymethyl group to an aldehyde, followed by Grignard addition to introduce the hydroxybenzyl moiety with stereocontrol.
- Final stereoselective oxidation of the pyrrolidinone ring using lithium diisopropylamide and oxygen to yield Clausenamide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from extraction to total synthesis represents a strategic opportunity to de-risk the supply of clausenamide. The traditional reliance on plant material creates a fragile supply chain susceptible to geopolitical and environmental disruptions. By adopting this synthetic route, organizations can secure a stable, year-round supply of the API intermediate. The elimination of agricultural sourcing removes the variability associated with crop yields and harvest seasons, allowing for more accurate demand forecasting and inventory planning. Furthermore, the synthetic route produces a product that is chemically identical to the natural isolate but free from the complex matrix of plant-derived impurities, reducing the burden on quality assurance teams.
- Cost Reduction in Manufacturing: The economic implications of this synthetic method are profound, primarily driven by the drastic improvement in material efficiency. Unlike the extraction process which consumes massive quantities of biomass for negligible output, the chemical synthesis utilizes high-yield reactions that maximize atom economy. The avoidance of expensive chromatographic purifications needed to separate plant impurities further drives down processing costs. Additionally, the reagents employed, such as oxygen and common organic solvents, are commodity chemicals with stable pricing, shielding the production budget from the volatility often seen in specialty botanical extracts. This structural shift in the cost base allows for substantial savings that can be passed down the value chain or reinvested in further R&D.
- Enhanced Supply Chain Reliability: Supply continuity is a critical metric for pharmaceutical manufacturing, and this synthetic route offers superior reliability compared to botanical sourcing. Chemical synthesis can be performed in controlled reactor environments independent of weather patterns or agricultural pests. This independence ensures that production schedules can be maintained consistently, reducing the risk of stockouts that could halt downstream formulation lines. The ability to scale production simply by increasing reactor volume, rather than securing more land for cultivation, provides a linear and predictable path to meeting increased market demand. This reliability is essential for maintaining the trust of downstream partners and regulatory bodies.
- Scalability and Environmental Compliance: From an environmental and operational perspective, the synthetic route is highly amenable to scale-up. The process avoids the generation of massive amounts of solid plant waste associated with extraction, replacing it with liquid waste streams that are easier to treat and manage using standard industrial wastewater protocols. The reactions operate at moderate temperatures and pressures, reducing energy consumption and safety risks associated with high-pressure hydrogenation or extreme thermal processes. This alignment with green chemistry principles not only simplifies regulatory compliance but also enhances the sustainability profile of the final product, a growing priority for global pharmaceutical buyers.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its adoption. The following questions address common concerns regarding the feasibility, purity, and scalability of the described method. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. By clarifying these points, we aim to facilitate a smoother transition from laboratory evaluation to commercial procurement.
Q: How does the synthetic route ensure the correct C3-C4 trans configuration?
A: The process utilizes a specific oxidation step on the enolate salt of the pyrrolidinone precursor using oxygen and a phosphite auxiliary, which selectively yields the thermodynamically stable trans-hydroxylated product.
Q: What are the advantages of this synthesis over plant extraction?
A: Synthetic production eliminates the dependency on seasonal plant availability, avoids complex purification of plant impurities, and significantly increases yield compared to the negligible recovery rates from dried leaves.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the reaction conditions utilize standard organic solvents and reagents compatible with industrial reactors, allowing for scalable production from kilogram to metric ton quantities without requiring exotic catalysts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Clausenamide Supplier
The technical potential of this synthetic route is immense, offering a clear path to high-volume production of this hepatoprotective agent. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this chemistry to life. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of handling the sensitive oxidation steps and stereochemical controls demanded by this process. We understand that consistency is key in API manufacturing, and our team is dedicated to ensuring that every batch meets the highest international standards for pharmaceutical intermediates.
We invite you to discuss how this advanced synthesis can optimize your supply chain and reduce your overall manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities align with your project goals. Let us partner with you to secure a reliable and cost-effective supply of clausenamide for your pharmaceutical applications.
