Advanced Four-Step Synthesis Of Dydrogesterone For Commercial Scale-Up And Procurement Efficiency
Advanced Four-Step Synthesis Of Dydrogesterone For Commercial Scale-Up And Procurement Efficiency
The pharmaceutical industry continuously seeks robust manufacturing pathways for critical hormonal therapies, and the synthesis of dydrogesterone represents a significant area of innovation for reducing dependency on single-source suppliers. Patent CN112812146A introduces a transformative methodology that streamlines the production of this vital progestogen by utilizing pregnenolone as a cost-effective starting material rather than traditional progesterone. This novel approach addresses long-standing inefficiencies in steroid synthesis by condensing the reaction sequence into merely four strategic steps, thereby enhancing overall process throughput and minimizing cumulative yield losses associated with linear synthesis. By leveraging photochemical isomerization and aluminum-mediated oxidation, the technology achieves a total yield of 16.4 percent while maintaining a purity profile exceeding 99.5 percent, which is critical for meeting stringent regulatory standards in global markets. For procurement leaders and technical directors, understanding this shift from multi-step transition metal catalysis to a greener organic pathway offers substantial opportunities for supply chain optimization and risk mitigation. The following analysis details how this patented route redefines the economic and technical landscape for producing high-purity dydrogesterone at an industrial scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the manufacturing of dydrogesterone has been plagued by inefficient synthetic routes that rely heavily on progesterone as the initial feedstock, leading to prohibitively low overall yields and complex purification challenges. As illustrated in prior art such as world patent WO 2018/109622, traditional methods often necessitate a nine-step linear synthesis that involves multiple oxidation stages using harsh chemical oxidants, resulting in significant environmental burden and waste disposal costs. 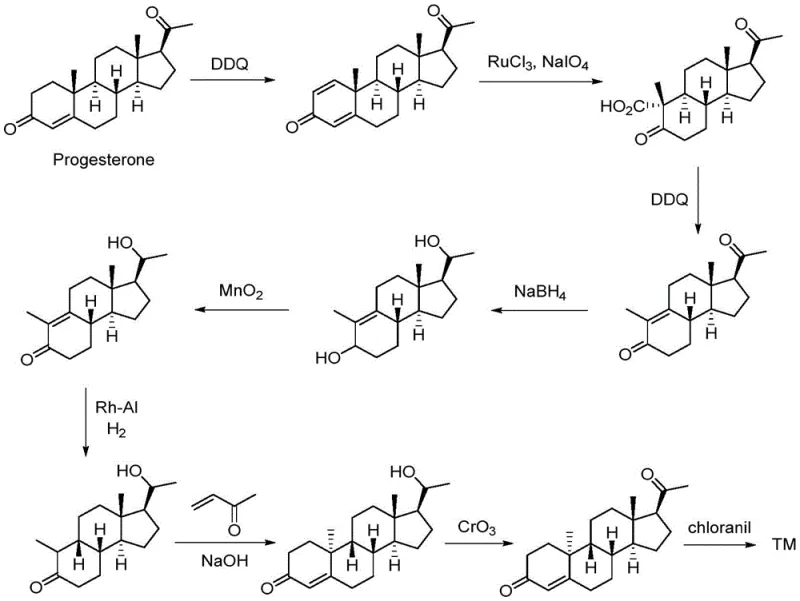 Furthermore, these legacy processes frequently require high-pressure hydrogenation steps involving transition metal catalysts, which introduce potential safety hazards and necessitate expensive heavy metal removal protocols to ensure patient safety. The cumulative effect of these lengthy sequences is a total yield that often falls below 5 percent, making the final active pharmaceutical ingredient economically vulnerable to raw material price fluctuations and production bottlenecks. Additionally, the reliance on scarce or expensive starting materials like trans-progesterone in certain one-step attempts further complicates the supply chain, as seen in US patent 3198792 where key precursors are not commercially viable. These structural inefficiencies create a fragile supply network that struggles to meet the growing global demand for hormonal treatments without incurring excessive operational expenditures.
Furthermore, these legacy processes frequently require high-pressure hydrogenation steps involving transition metal catalysts, which introduce potential safety hazards and necessitate expensive heavy metal removal protocols to ensure patient safety. The cumulative effect of these lengthy sequences is a total yield that often falls below 5 percent, making the final active pharmaceutical ingredient economically vulnerable to raw material price fluctuations and production bottlenecks. Additionally, the reliance on scarce or expensive starting materials like trans-progesterone in certain one-step attempts further complicates the supply chain, as seen in US patent 3198792 where key precursors are not commercially viable. These structural inefficiencies create a fragile supply network that struggles to meet the growing global demand for hormonal treatments without incurring excessive operational expenditures.
The Novel Approach
In stark contrast to these cumbersome legacy methods, the innovative process disclosed in CN112812146A leverages pregnenolone, a widely available and inexpensive steroid precursor, to construct the dydrogesterone backbone with remarkable efficiency. This modernized route eliminates the need for transition metal catalysts entirely, substituting them with small organic molecules and aluminum-based reagents that are both safer to handle and easier to remove from the final product matrix. By reducing the synthetic timeline to just four distinct chemical transformations, the process drastically cuts down on solvent consumption, energy usage, and labor hours required for intermediate isolation and purification. The strategic use of allylic halogenation followed by elimination allows for the rapid construction of the necessary diene system, setting the stage for the critical photochemical rearrangement that defines the molecule's biological activity. This streamlined architecture not only improves the total yield to 16.4 percent but also ensures a much cleaner reaction profile, reducing the burden on downstream processing units. Consequently, this approach represents a paradigm shift towards sustainable manufacturing that aligns with modern green chemistry principles while delivering superior economic performance for commercial producers.
Mechanistic Insights into Photo-Isomerization and Oppenauer Oxidation
The core technical breakthrough of this synthesis lies in the precise execution of the photo-isomerization step, which converts the initial diene intermediate into the crucial 9β,10α-Pregna-5,7-dien-3-ol-20-one configuration required for biological efficacy. This transformation is achieved by irradiating the reaction mixture in tetrahydrofuran with a high-pressure mercury lamp within a wavelength range of 210 to 490 nanometers, facilitating a radical-mediated rearrangement that would be difficult to achieve through thermal means alone. 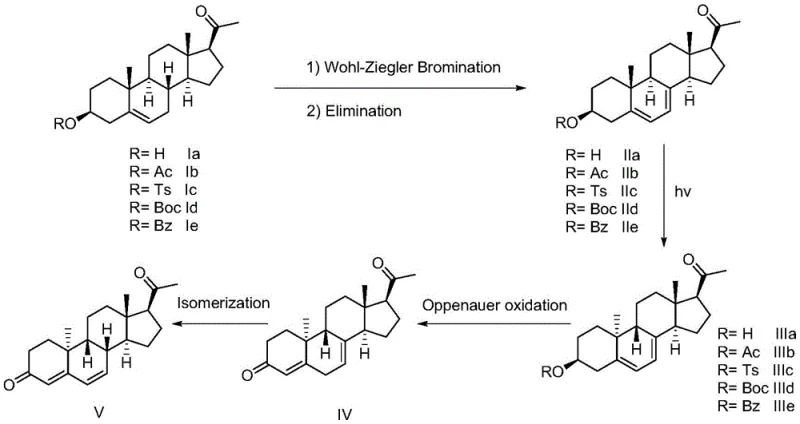 The careful control of light intensity, typically between 500W and 2000W, ensures that the reaction proceeds with high selectivity, minimizing the formation of unwanted stereoisomers that could compromise the purity of the final drug substance. Following this photochemical event, the process employs an Oppenauer oxidation utilizing aluminum alkoxides or aluminum-based molecular sieves in the presence of a ketone acceptor like N-methyl-4-piperidone. This oxidation step is particularly elegant as it avoids the use of toxic chromium or manganese reagents, instead relying on the reversible transfer of hydride ions to convert the alcohol functionality into the necessary ketone group with high fidelity. The combination of these two key mechanistic steps ensures that the stereochemical integrity of the steroid nucleus is maintained throughout the synthesis, resulting in a product that meets rigorous pharmacopeial specifications without the need for extensive chromatographic separation.
The careful control of light intensity, typically between 500W and 2000W, ensures that the reaction proceeds with high selectivity, minimizing the formation of unwanted stereoisomers that could compromise the purity of the final drug substance. Following this photochemical event, the process employs an Oppenauer oxidation utilizing aluminum alkoxides or aluminum-based molecular sieves in the presence of a ketone acceptor like N-methyl-4-piperidone. This oxidation step is particularly elegant as it avoids the use of toxic chromium or manganese reagents, instead relying on the reversible transfer of hydride ions to convert the alcohol functionality into the necessary ketone group with high fidelity. The combination of these two key mechanistic steps ensures that the stereochemical integrity of the steroid nucleus is maintained throughout the synthesis, resulting in a product that meets rigorous pharmacopeial specifications without the need for extensive chromatographic separation.
Beyond the primary transformation mechanisms, the process incorporates sophisticated impurity control strategies that are essential for scaling this chemistry to multi-ton production levels. The initial halogenation step utilizes specific initiators such as AIBN or peroxides in conjunction with halide sources like N-bromosuccinimide to ensure regioselective functionalization at the allylic position, preventing side reactions at other sensitive sites on the steroid ring. Subsequent acid-catalyzed isomerization in the final step is carefully tuned using solvents like isopropanol or hexafluoroisopropanol to drive the equilibrium towards the desired 6-dehydro product while suppressing the formation of conjugated byproducts. The absence of transition metals throughout the entire sequence means that the risk of metal leaching into the final API is virtually eliminated, simplifying the validation process for regulatory filings. Furthermore, the ability to recycle solvents such as petroleum ether and toluene across multiple steps contributes to a closed-loop manufacturing system that minimizes waste generation. This holistic attention to mechanistic detail and impurity profiling demonstrates a deep understanding of process chemistry that is vital for ensuring consistent batch-to-batch quality in a commercial setting.
How to Synthesize Dydrogesterone Efficiently
Implementing this advanced synthesis route requires a clear understanding of the operational parameters defined in the patent to ensure optimal yield and safety during production. The process begins with the preparation of the halogenated intermediate, followed by the critical photochemical step, oxidation, and final acid treatment, each requiring specific temperature and stoichiometric controls. Detailed standardized operating procedures for each unit operation are essential for maintaining consistency, particularly regarding the light source intensity and the purity of the aluminum reagents used in the oxidation phase. For technical teams looking to adopt this methodology, the following guide outlines the critical workflow necessary to replicate the patented success in a pilot or production environment.
- Perform allylic halogenation and elimination on pregnenolone derivatives using NBS or DBDMH with an azo initiator to form the diene intermediate.
- Execute photo-isomerization under UV light (210-490 nm) in tetrahydrofuran to achieve the critical 9-beta,10-alpha stereochemical configuration.
- Conduct Oppenauer oxidation using aluminum alkoxides followed by acid-catalyzed olefin translocation to finalize the dydrogesterone structure.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this four-step synthesis route offers profound benefits that extend far beyond simple chemical yield improvements, directly impacting the bottom line and supply resilience. By shifting the starting material to pregnenolone, manufacturers can tap into a more abundant and stable supply chain compared to the constrained availability of specialized progesterone derivatives required by older methods. This fundamental change in raw material strategy significantly reduces the risk of supply disruptions caused by upstream bottlenecks, ensuring a more reliable flow of intermediates for downstream formulation. Moreover, the elimination of expensive transition metal catalysts and high-pressure hydrogenation equipment lowers the capital expenditure barrier for production facilities, allowing for more flexible manufacturing arrangements. These structural advantages translate into a more competitive cost structure that can withstand market volatility while maintaining high margins for suppliers.
- Cost Reduction in Manufacturing: The removal of transition metal catalysts and the reduction of synthetic steps from nine to four fundamentally alters the cost equation by eliminating expensive reagent purchases and complex waste treatment protocols. Without the need for heavy metal scavengers or high-pressure reactors, the operational expenditure per kilogram of product is drastically simplified, leading to substantial cost savings in utility consumption and labor. The higher overall yield of 16.4 percent means that less raw material is wasted to produce the same amount of finished goods, effectively lowering the cost of goods sold. Additionally, the ability to recycle solvents like petroleum ether and toluene further enhances the economic efficiency of the process, making it highly attractive for cost-sensitive procurement strategies in generic drug manufacturing.
- Enhanced Supply Chain Reliability: Utilizing pregnenolone as a feedstock provides a significant advantage in terms of raw material security, as it is a widely produced natural steroid available from multiple global sources compared to proprietary intermediates. This diversification of supply sources reduces the dependency on single vendors, thereby mitigating the risk of price gouging or allocation during periods of high demand. The simplified process flow also shortens the production cycle time, allowing manufacturers to respond more rapidly to fluctuating market requirements and urgent orders from pharmaceutical clients. By avoiding complex steps that require specialized equipment or hazardous conditions, the process becomes more robust and less prone to unplanned downtime, ensuring consistent delivery schedules for partners relying on a reliable steroid intermediate supplier.
- Scalability and Environmental Compliance: The green nature of this synthesis, characterized by the absence of toxic heavy metals and the use of recyclable solvents, aligns perfectly with increasingly stringent environmental regulations governing pharmaceutical manufacturing. Scaling this process does not require massive investments in pollution control infrastructure for heavy metal waste, simplifying the permitting process for new production lines or facility expansions. The use of photochemistry and mild aluminum reagents presents a safer working environment for operators, reducing liability and insurance costs associated with hazardous chemical handling. This environmental compatibility ensures long-term operational sustainability, protecting the supply chain from future regulatory shocks that could disrupt less compliant manufacturing routes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the patent specifications and comparative data. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers provided reflect the specific advantages and operational parameters outlined in the intellectual property documentation.
Q: What are the primary advantages of the pregnenolone-based route over progesterone-based synthesis?
A: The pregnenolone-based route described in CN112812146A reduces the synthetic steps from nine to four, significantly improving total yield from less than 5 percent to 16.4 percent while eliminating the need for high-pressure hydrogenation and toxic transition metal catalysts.
Q: How does the photo-isomerization step impact impurity profiles?
A: The photochemical conversion allows for precise stereochemical control at the 9-beta,10-alpha position without requiring complex chiral resolution steps, resulting in a final product purity exceeding 99.5 percent after recrystallization.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process utilizes readily available raw materials like pregnenolone and avoids expensive transition metals, making it economically viable for commercial scale-up of complex steroid hormones with reduced environmental waste.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dydrogesterone Supplier
The technological potential of this four-step synthesis route underscores the importance of partnering with a CDMO expert capable of translating complex laboratory patents into robust industrial reality. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this green chemistry are fully realized in large-scale batches. Our facilities are equipped with stringent purity specifications and rigorous QC labs designed to handle sensitive photochemical reactions and maintain the high-quality standards required for hormonal APIs. We understand that moving from a legacy nine-step process to this optimized four-step method requires precise engineering and process validation, which our technical team is prepared to execute with speed and accuracy.
We invite procurement leaders and R&D directors to initiate a dialogue regarding your specific supply chain needs for this critical intermediate. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this more efficient manufacturing route for your organization. We encourage you to contact our technical procurement team to索取 specific COA data and route feasibility assessments tailored to your volume requirements. Let us help you secure a sustainable and cost-effective supply of high-purity dydrogesterone that supports your long-term business goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
