Scalable Total Synthesis of Morphine Derivatives via Advanced Catalytic Strategies
The pharmaceutical industry has long grappled with the supply chain vulnerabilities inherent in the production of morphine derivatives, a class of essential analgesics that includes oxycodone, hydrocodone, and naloxone. Traditionally dependent on the cultivation and extraction of opium poppies, the global supply of these critical medications is susceptible to geopolitical instability, climate change, and regulatory fluctuations. Patent CN113845478A introduces a transformative biomimetic synthesis strategy that circumvents these agricultural bottlenecks by constructing the complex morphine skeleton through advanced organic chemistry. This innovation leverages transition metal-catalyzed asymmetric hydrogenation and intramolecular Heck reactions to achieve high efficiency and selectivity, marking a significant shift towards a more secure and sustainable manufacturing paradigm for high-value opioid intermediates.
This technological advancement is particularly relevant for stakeholders seeking a reliable pharmaceutical intermediate supplier who can guarantee continuity of supply without the risks associated with botanical sourcing. The patent outlines a comprehensive route that not only replicates the natural biosynthetic logic but enhances it through modern catalytic tools, resulting in a process that is chemically robust and operationally simpler than previous total synthesis attempts. By decoupling production from crop yields, this method offers a strategic advantage for securing the global supply of pain management therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of morphine-like drugs has been inextricably linked to the agricultural sector, requiring vast tracts of land for poppy cultivation under strict international control. This reliance introduces significant volatility; crop failures due to pests or adverse weather can lead to immediate shortages, while political tensions in key growing regions can disrupt logistics and inflate costs. Furthermore, the extraction and semi-synthesis processes are often laden with complex purification steps to remove plant-derived impurities, leading to lower overall yields and higher environmental burdens. The existing total synthesis methods reported in academic literature, while scientifically elegant, have frequently failed to translate to commercial viability due to prohibitive costs, low yields in key coupling steps, and the use of hazardous reagents that complicate scale-up.
The Novel Approach
In stark contrast, the methodology disclosed in CN113845478A utilizes a biomimetic strategy that constructs the core structure from readily available chemical building blocks rather than plant extracts. The core innovation lies in the sequential application of an iridium-catalyzed asymmetric hydrogenation to set critical chiral centers followed by a palladium-catalyzed intramolecular Heck reaction to forge the rigid ring system. This approach eliminates the variability of biological sources and allows for precise control over reaction parameters. The process is designed for scalability, utilizing standard industrial solvents and catalysts that facilitate easier downstream processing. By achieving high conversion rates and excellent stereoselectivity in these key steps, the new route significantly streamlines the manufacturing workflow, offering a viable path for cost reduction in pharmaceutical manufacturing while ensuring a consistent quality profile.
Mechanistic Insights into Iridium-Catalyzed Asymmetric Hydrogenation and Heck Cyclization
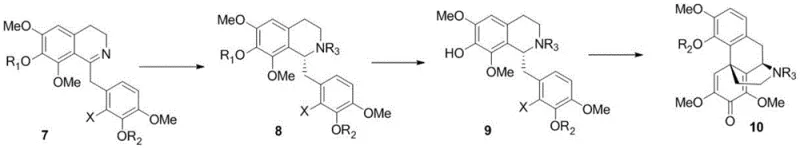
The heart of this synthetic breakthrough is the precise control of stereochemistry during the formation of the tetrahydroisoquinoline core. The process employs an Iridium catalyst complex, specifically generated from [Ir(cod)Cl]2 and chiral ligands such as (R)-(+)-BINAP or its derivatives, to effect the asymmetric hydrogenation of the imine intermediate. This step is critical as it establishes the configuration of the chiral centers that define the biological activity of the final opioid. The reaction proceeds under mild conditions, typically around 0°C and 500 psi of hydrogen pressure, in solvents like chloroform, which balances solubility and reaction rate. The inclusion of additives like tetraethylammonium iodide further enhances the catalytic turnover and selectivity, ensuring that the desired enantiomer is produced with an ee value exceeding 99%, thereby minimizing the formation of inactive or harmful isomers.
Following the establishment of chirality, the synthesis advances to the construction of the morphine skeleton via an intramolecular Heck reaction. This carbon-carbon bond-forming reaction is catalyzed by palladium complexes, such as Pd(PPh tBu2)2Cl2, in the presence of a base like potassium tert-butoxide. The reaction effectively closes the ring system, creating the complex bridged structure characteristic of morphine derivatives. The choice of ligand and catalyst in this step is paramount to overcoming the steric hindrance inherent in forming such a congested molecular architecture. The patent data indicates that optimizing the ligand environment allows for high yields (up to 87% in specific examples) and clean conversion, avoiding the formation of by-products that would otherwise necessitate extensive chromatographic purification. This mechanistic efficiency is what renders the total synthesis commercially competitive against semi-synthetic routes.
How to Synthesize Oxycodone Intermediate Efficiently
The synthesis of the key intermediate involves a multi-step sequence starting from simple aromatic precursors, progressing through amide coupling, cyclization, and the aforementioned catalytic transformations. The process is designed to minimize isolation steps, with several intermediates carried forward as crude materials to maximize throughput. Detailed operational parameters, including specific molar ratios of catalysts to substrates and precise temperature controls, are critical for reproducing the high yields reported in the patent examples. For a comprehensive understanding of the specific reaction conditions and workup procedures required to implement this technology, please refer to the standardized synthesis guide below.
- Perform asymmetric hydrogenation on compound 7 using an Iridium catalyst and chiral ligand to establish chirality.
- Deprotect the hydroxyl group to generate compound 9 suitable for cyclization.
- Execute an intramolecular Heck reaction using a Palladium catalyst to close the ring and form intermediate 10.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift from agricultural extraction to chemical synthesis represents a fundamental improvement in risk management and cost predictability. The traditional model is plagued by long lead times associated with crop cycles and the logistical complexities of handling controlled substances from farm to factory. This new synthetic route offers a decentralized production model where intermediates can be manufactured in standard chemical facilities anywhere in the world, provided the necessary regulatory licenses are in place. This geographic flexibility drastically reduces the risk of supply disruption caused by regional instabilities and allows for a more responsive inventory management strategy that can adapt to fluctuating market demand without the lag time of agricultural production.
- Cost Reduction in Manufacturing: The streamlined nature of this synthetic route directly translates to lower operational expenditures. By achieving high yields in the key bond-forming steps and reducing the number of purification operations required, the process minimizes solvent consumption and waste generation. The elimination of expensive and variable plant raw materials removes a major source of cost volatility. Furthermore, the use of robust catalysts that can be optimized for turnover means that the cost of goods sold (COGS) can be significantly lowered compared to semi-synthetic methods that suffer from low recovery rates during extraction. The ability to run reactions at moderate temperatures and pressures also reduces energy consumption, contributing to overall economic efficiency.
- Enhanced Supply Chain Reliability: Dependence on a single agricultural source creates a single point of failure in the supply chain. This chemical synthesis method diversifies the supply base, allowing for multiple manufacturing sites to produce the same high-quality intermediate. This redundancy is crucial for maintaining the continuity of essential pain medications. The synthetic route uses commercially available starting materials and reagents, ensuring that the supply chain is not bottlenecked by niche or scarce resources. This reliability allows pharmaceutical companies to plan long-term production schedules with greater confidence, knowing that the supply of active ingredients is not subject to the whims of nature or international trade disputes regarding agricultural commodities.
- Scalability and Environmental Compliance: The patent explicitly demonstrates the scalability of the process, with examples showing successful execution on scales relevant to commercial production. The reaction conditions are compatible with standard stainless steel reactors used in the fine chemical industry, facilitating a smooth transition from pilot plant to full-scale manufacturing. Additionally, the reduction in solvent usage and waste streams aligns with increasingly stringent environmental regulations. By avoiding the large volumes of water and organic solvents typically required for plant extraction and purification, this method offers a greener alternative that simplifies compliance with environmental, health, and safety (EHS) standards, thereby reducing the regulatory burden on the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this biomimetic synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on the feasibility and advantages of adopting this route for the production of opioid analgesics.
Q: How does this synthesis method improve supply chain security compared to traditional extraction?
A: Unlike traditional methods relying on opium poppy cultivation which are subject to geopolitical and climatic instability, this total synthesis route utilizes stable chemical starting materials and standard catalytic processes, ensuring a consistent and controllable supply of high-purity intermediates independent of agricultural variables.
Q: What represents the key technological breakthrough in this patent regarding stereoselectivity?
A: The process employs a highly efficient transition metal-catalyzed asymmetric hydrogenation step using specific Iridium complexes and chiral ligands like (R)-(+)-BINAP, achieving excellent enantiomeric excess (ee > 99%) which is critical for the biological activity and safety profile of the final opioid analgesic.
Q: Is this synthetic route suitable for large-scale industrial manufacturing?
A: Yes, the patent explicitly demonstrates scalability with reaction conditions optimized for larger batches, including the use of robust catalysts and solvents that allow for simplified purification processes, significantly reducing the operational complexity typically associated with complex alkaloid synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Oxycodone Intermediate Supplier
The transition to a fully synthetic supply chain for morphine derivatives requires a partner with deep expertise in complex organic synthesis and process development. NINGBO INNO PHARMCHEM stands at the forefront of this technological shift, possessing the technical capability to adapt and optimize such advanced catalytic routes for commercial production. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of intermediate meets the exacting standards required for pharmaceutical applications, providing our partners with the confidence needed to integrate new supply sources into their global networks.
We invite procurement leaders and R&D directors to engage with us to explore how this innovative synthesis route can be integrated into your supply chain. By leveraging our CDMO capabilities, you can secure a stable, high-quality source of critical intermediates that mitigates the risks of traditional sourcing. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how we can support your commitment to delivering essential pain relief medications to patients worldwide without interruption.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
