Revolutionizing Opioid Manufacturing: Advanced Biomimetic Synthesis for High-Purity Morphine Intermediates
The pharmaceutical industry is currently witnessing a paradigm shift in the production of controlled substances, driven by the urgent need for secure and sustainable supply chains. Patent CN114423764B discloses a groundbreaking biomimetic total synthesis method for morphine derivatives, specifically targeting the efficient preparation of oxycodone and codeine. This technology leverages a sophisticated understanding of biogenic pathways to construct the complex morphine skeleton without relying on the traditional extraction from opium poppies. By utilizing transition metal-catalyzed asymmetric hydrogenation and intramolecular oxidative dearomatization Heck reactions as key steps, this method achieves superior chemical and enantioselectivity. The strategic importance of this innovation lies in its ability to bypass the agricultural bottlenecks and regulatory complexities associated with poppy cultivation, offering a chemically defined route that ensures consistent quality and availability for global healthcare markets.
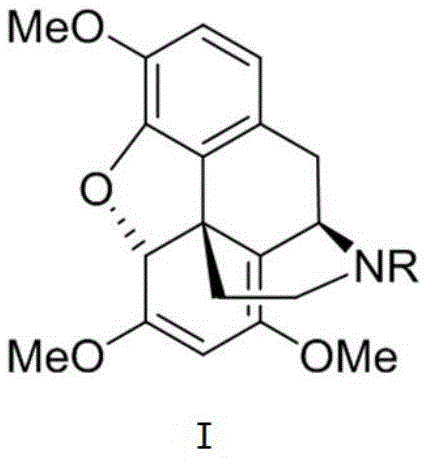
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of morphine-like drugs has been inextricably linked to the agricultural cultivation of opium poppies, a process fraught with significant vulnerabilities and inefficiencies. The reliance on natural extraction means that supply volumes are subject to the whims of weather patterns, pest infestations, and geopolitical instability in growing regions, leading to fluctuating market prices and potential shortages. Furthermore, the semi-synthetic processes derived from extracted morphine or thebaine often involve lengthy control procedures and complex regulatory oversight to prevent diversion, which adds substantial administrative costs and lead times to the manufacturing lifecycle. From a chemical perspective, traditional methods struggle with the inherent variability of natural products, where impurity profiles can vary batch to batch, necessitating rigorous and costly purification steps to meet stringent pharmacopeial standards for active pharmaceutical ingredients.
The Novel Approach
In stark contrast, the novel biomimetic synthesis route described in the patent data offers a robust, chemically controlled alternative that decouples production from agricultural constraints. This approach utilizes precise organic synthesis techniques to build the morphine core from simpler, commercially available starting materials, ensuring a consistent and reproducible impurity profile that simplifies quality control. The method is designed with scalability in mind, employing reaction conditions that are mild and operationally simple, allowing for seamless translation from laboratory benchtop to large-scale commercial reactors. By focusing on key catalytic transformations that high-yieldingly construct the complex ring systems, this new methodology significantly reduces the number of synthetic steps and waste generation, aligning with modern green chemistry principles while enhancing the overall economic viability of producing essential pain management medications.
Mechanistic Insights into Ir-Catalyzed Asymmetric Hydrogenation and Heck Cyclization
The cornerstone of this synthetic breakthrough is the implementation of a highly stereoselective asymmetric hydrogenation reaction, which sets the critical chiral centers required for the biological activity of the final opioid product. This step employs an Iridium-based catalyst system coordinated with specialized chiral ligands, such as BINAP derivatives, to differentiate between enantiotopic faces of the substrate molecule under hydrogen pressure. The precision of this catalytic cycle ensures that the resulting tetrahydroisoquinoline intermediate possesses the correct absolute configuration, thereby eliminating the need for difficult resolution steps later in the synthesis. The optimization of reaction parameters, including solvent choice, temperature, and additive concentration, allows for near-quantitative conversion rates while maintaining exceptional enantiomeric excess, which is paramount for ensuring the safety and efficacy of the downstream pharmaceutical products.
Following the establishment of stereochemistry, the synthesis proceeds through a pivotal intramolecular oxidative dearomatization Heck reaction to construct the characteristic five-ring fused structure of the morphine skeleton. This transformation involves the activation of a phenolic precursor using a Palladium catalyst, which facilitates the formation of a new carbon-carbon bond through an oxidative mechanism that disrupts the aromaticity of the ring system. 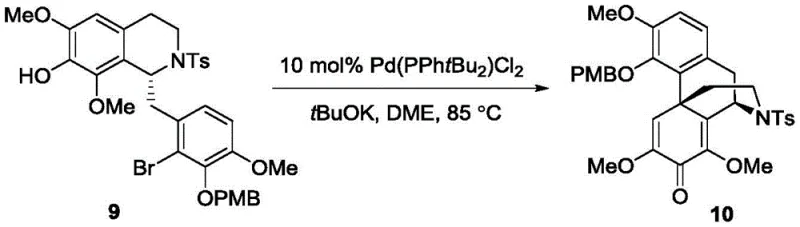
The efficiency of this cyclization is remarkable, with reported yields reaching up to 87 percent, a significant improvement over historical biomimetic attempts that often suffered from poor regioselectivity and low conversion. The mechanistic pathway is carefully tuned to minimize side reactions and byproduct formation, ensuring that the complex polycyclic framework is assembled with high fidelity. This step effectively mimics nature's biosynthetic machinery but does so under controlled laboratory conditions that allow for rigorous monitoring and optimization, providing a reliable foundation for the subsequent functional group manipulations needed to reach the final active drug substances.
How to Synthesize Morphine Intermediate I Efficiently
The preparation of the key Intermediate I involves a sequential series of catalytic transformations that prioritize atom economy and operational simplicity to facilitate industrial adoption. The process begins with the assembly of the linear precursor followed by the critical cyclization events that lock in the three-dimensional architecture of the molecule. Detailed standardized synthesis steps see the guide below.
- Prepare the tetrahydroisoquinoline precursor via asymmetric hydrogenation using an Iridium catalyst system with chiral ligands to establish stereochemistry.
- Execute the intramolecular oxidative dearomatization Heck reaction using a Palladium catalyst to construct the core five-ring morphine skeleton efficiently.
- Perform downstream functional group modifications including reduction and cyclization to yield the final Intermediate I suitable for Oxycodone conversion.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this total synthesis technology represents a strategic opportunity to de-risk the sourcing of critical opioid intermediates and stabilize long-term supply agreements. By shifting away from agriculture-dependent raw materials, manufacturers can insulate their production schedules from external environmental shocks and regulatory changes affecting crop cultivation, thereby ensuring a more predictable and continuous flow of materials. This synthetic route also offers substantial cost reduction in pharmaceutical intermediate manufacturing by streamlining the purification process; the patent highlights that the entire synthetic route requires minimal chromatographic separation, relying instead on crystallization and filtration which are far more scalable and cost-effective unit operations in a commercial plant setting.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal removal steps and the reduction in solvent consumption through optimized reaction concentrations directly contribute to a lower cost of goods sold. By avoiding the need for extensive agricultural processing and the associated logistics of handling controlled plant materials, the overall operational expenditure is significantly reduced, allowing for more competitive pricing structures in the global market without compromising on quality standards.
- Enhanced Supply Chain Reliability: Synthetic production facilities can be located in diverse geographic regions independent of specific climatic zones required for poppy growth, diversifying the supplier base and reducing single-point-of-failure risks. The ability to produce key intermediates on demand using stable chemical feedstocks ensures that inventory levels can be maintained consistently, preventing stockouts that could disrupt the manufacturing of finished dosage forms for patients relying on these essential medicines for pain management.
- Scalability and Environmental Compliance: The process has been demonstrated to operate effectively at scales exceeding 100 grams with clear pathways for expansion to multi-ton production, supported by mild reaction conditions that reduce energy consumption. Furthermore, the simplified workup procedures result in reduced waste discharge and lower environmental impact, facilitating easier compliance with increasingly stringent global environmental regulations and sustainability goals mandated by corporate governance frameworks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this biomimetic synthesis technology in a GMP manufacturing environment. These answers are derived directly from the experimental data and beneficial effects outlined in the patent documentation to provide clarity on feasibility and performance metrics.
Q: How does this synthetic route improve upon traditional opium poppy extraction?
A: This biomimetic total synthesis eliminates dependence on agricultural cultivation, mitigating risks associated with climate, pests, and geopolitical instability while ensuring consistent chemical purity and supply continuity.
Q: What is the enantioselectivity performance of the key hydrogenation step?
A: The patented asymmetric hydrogenation process utilizes optimized Iridium catalysts and chiral ligands to achieve excellent enantiomeric excess, ensuring the production of the correct stereoisomer required for biological activity.
Q: Is this manufacturing process scalable for industrial production?
A: Yes, the methodology has been validated at scales exceeding 100 grams with simplified purification protocols, demonstrating robust feasibility for commercial scale-up from 100 kgs to 100 MT annual production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Morphine Intermediate Supplier
NINGBO INNO PHARMCHEM stands at the forefront of adopting such advanced synthetic methodologies to deliver high-value pharmaceutical intermediates to the global market. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes are successfully translated into robust industrial manufacturing lines. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch meets the highest international standards, providing our partners with the confidence needed to integrate these complex intermediates into their own supply chains.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this cutting-edge technology for their opioid portfolios. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your supply chain resilience and drive down overall production costs.
