Scalable Catalyst-Free Synthesis of 3-Selenocyanoindole Compounds for Advanced Drug Discovery
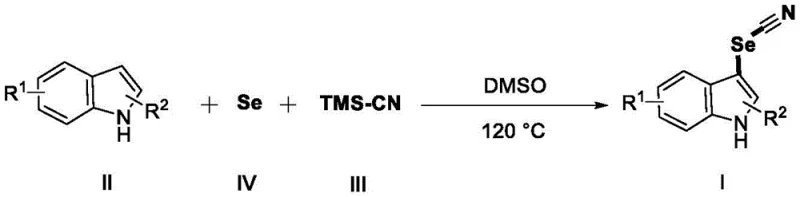
The landscape of heterocyclic chemistry is constantly evolving, driven by the demand for efficient routes to bioactive scaffolds. Patent CN109232363B introduces a transformative approach to synthesizing 3-selenocyanoindole compounds, a class of molecules with significant potential in biomedicine and agrochemicals. This technology leverages elemental selenium and trimethylsilyl cyanide (TMS-CN) as a novel selenocyano source, operating under catalyst-free conditions at 120°C in dimethyl sulfoxide (DMSO). Unlike traditional methods that rely on expensive transition metals or hazardous oxidants, this process utilizes commodity chemicals to achieve high-yielding C-SeCN bond formation. For R&D directors and procurement specialists, this represents a paradigm shift towards greener, more cost-effective manufacturing of complex nitrogen-containing heterocycles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of selenocyano groups onto indole frameworks has been fraught with synthetic challenges and safety concerns. Previous literature, such as the work by Muniraj et al. in 2016, relied on N-iodosuccinimide (NIS) as a catalyst and tert-butyl hydroperoxide (TBHP) as an oxidant, creating potential explosion hazards and requiring careful handling of peroxides. Other approaches utilized copper promoters or iodine catalysts alongside potassium selenocyanate, which often necessitated complex additive systems like tetrabutylammonium iodide. These metal-catalyzed routes introduce the risk of heavy metal contamination, a critical failure point for pharmaceutical intermediates that must meet stringent purity specifications. Furthermore, methods employing triselenium dicyanide involve multi-step reagent preparation, driving up costs and complicating the supply chain for large-scale production.
The Novel Approach
The methodology disclosed in CN109232363B circumvents these issues by employing a direct, catalyst-free strategy. By utilizing elemental selenium, an abundant and inexpensive raw material, combined with TMS-CN, the process eliminates the need for transition metal catalysts entirely. The reaction proceeds smoothly in DMSO at 120°C under ambient air, removing the requirement for inert gas protection or specialized pressure equipment beyond standard heating capabilities. This simplicity translates directly to operational efficiency; the absence of metal catalysts means there is no need for expensive scavenging resins or complex purification steps to remove trace metals. The broad substrate scope, accommodating various substituents on the indole ring, further underscores the robustness of this method for generating diverse libraries of selenocyano-substituted compounds for drug discovery programs.
Mechanistic Insights into Catalyst-Free Selenocyanation
The core of this innovation lies in the unique activation of elemental selenium by TMS-CN in a polar aprotic solvent. While the exact mechanistic pathway may involve radical or ionic intermediates facilitated by the high temperature and the polarity of DMSO, the outcome is a highly regioselective functionalization at the C3 position of the indole nucleus. The electron-rich nature of the indole C3 position makes it the preferred site for electrophilic attack or radical addition, ensuring that the selenocyano group is installed precisely where it is needed for biological activity. This inherent selectivity minimizes the formation of regioisomers, which are often difficult to separate and can compromise the efficacy of the final active pharmaceutical ingredient. The use of TMS-CN serves not only as a cyanide source but likely facilitates the solubilization and activation of the elemental selenium, creating a reactive selenocyanating species in situ without external promoters.
From an impurity control perspective, the absence of metal catalysts is a decisive advantage for regulatory compliance. In conventional metal-catalyzed cross-couplings or functionalizations, residual metals like copper or palladium must be reduced to parts-per-million levels, often requiring additional processing steps that lower overall yield. In this catalyst-free protocol, the primary impurities are likely unreacted starting materials or minor side products that are easily removed via standard aqueous workup and silica gel chromatography. The use of DMSO, a high-boiling solvent, ensures that the reaction kinetics are favorable at 120°C, driving the conversion to completion within 16 to 24 hours. This thermal stability allows for a straightforward reaction profile that is easily monitored and controlled, reducing the risk of batch-to-batch variability in a commercial setting.
How to Synthesize 3-Selenocyanoindole Efficiently
The synthesis protocol described in the patent is remarkably straightforward, designed for ease of execution in both laboratory and pilot plant environments. The process begins with the sequential addition of the indole substrate, elemental selenium powder, and DMSO into a reaction vessel, followed by the introduction of TMS-CN. The mixture is then heated to 120°C and stirred until the starting material is consumed, typically requiring between 16 and 24 hours depending on the specific substrate electronics. Following the reaction, a simple aqueous workup involving extraction with ethyl acetate and drying over anhydrous sodium sulfate is sufficient to isolate the crude product, which is then purified by column chromatography. Detailed standardized synthesis steps are provided in the guide below.
- Charge indole substrate, elemental selenium, and DMSO solvent into a pressure-resistant reaction vessel under air atmosphere.
- Add trimethylsilyl cyanide (TMS-CN) to the mixture and heat the reaction system to 120°C for 16 to 24 hours.
- Upon completion, cool the mixture, extract with ethyl acetate, dry over sodium sulfate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the economic implications of this synthetic route are profound. The shift from specialized, expensive reagents to commodity chemicals like elemental selenium and TMS-CN drastically reduces the raw material cost base. Elemental selenium is widely available globally, ensuring supply continuity and shielding the manufacturing process from the volatility associated with precious metal catalysts. Additionally, the elimination of hazardous oxidants like TBHP reduces the costs associated with safety compliance, storage, and waste disposal. The simplified workflow, which does not require inert atmosphere techniques or complex additive packages, lowers the barrier to entry for contract manufacturing organizations, allowing for faster technology transfer and quicker time-to-market for new drug candidates.
- Cost Reduction in Manufacturing: The most significant driver of cost savings in this process is the complete removal of transition metal catalysts and expensive oxidants. Traditional methods often require stoichiometric or near-stoichiometric amounts of oxidants and catalytic loads of noble or base metals, both of which add substantial cost to the bill of materials. By replacing these with cheap elemental selenium, the direct material cost is significantly lowered. Furthermore, the downstream processing costs are reduced because there is no need for metal scavenging technologies or extensive testing for heavy metal residues, streamlining the quality control workflow and reducing the overall cost of goods sold for high-purity intermediates.
- Enhanced Supply Chain Reliability: Reliance on specialized reagents like triselenium dicyanide or specific organocatalysts can create bottlenecks in the supply chain, especially during periods of global chemical shortages. In contrast, elemental selenium and TMS-CN are bulk commodities produced by multiple suppliers worldwide, ensuring a robust and resilient supply chain. The ability to run the reaction in air without sensitive equipment further enhances reliability, as it reduces the dependency on specialized infrastructure like nitrogen generators or gloveboxes. This robustness ensures that production schedules can be maintained consistently, reducing lead times for high-purity pharmaceutical intermediates and preventing delays in clinical trial material supply.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental risks, particularly when peroxides or volatile solvents are involved. This method operates in DMSO, a solvent with a high boiling point and low volatility, which minimizes evaporative losses and fire hazards during scale-up. The absence of explosive peroxides makes the process inherently safer for large-scale reactors, reducing insurance premiums and safety monitoring costs. Moreover, the waste stream is simpler, consisting primarily of organic residues and inorganic selenium byproducts that are easier to manage than complex metal-laden waste, facilitating better environmental compliance and sustainability metrics for the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalyst-free selenocyanation technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on reaction conditions, substrate scope, and purification strategies. Understanding these details is essential for process chemists evaluating the feasibility of adopting this route for commercial production.
Q: What are the advantages of using elemental selenium over potassium selenocyanate?
A: Elemental selenium is significantly cheaper and more readily available than specialized selenocyanate salts. Furthermore, this novel method eliminates the need for toxic oxidants like TBHP or expensive metal catalysts required when using traditional selenocyanate sources, resulting in a cleaner reaction profile and reduced downstream purification costs.
Q: Does this synthesis method require an inert atmosphere?
A: No, one of the key operational advantages of this patented process is that it proceeds efficiently in an air atmosphere. This removes the need for rigorous degassing or nitrogen protection, simplifying the equipment requirements and making the process highly suitable for large-scale commercial manufacturing.
Q: What is the regioselectivity of the selenocyanation reaction?
A: The reaction exhibits excellent regioselectivity for the C3 position of the indole ring. This specificity is crucial for pharmaceutical applications where precise substitution patterns are required for biological activity, avoiding the formation of difficult-to-separate isomers common in less selective electrophilic substitutions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Selenocyanoindole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of efficient, scalable synthetic routes like the one described in CN109232363B for advancing your pipeline. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to industrial manufacturing is seamless. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3-selenocyanoindole intermediate delivered meets the highest standards required for pharmaceutical and agrochemical applications. We are committed to leveraging our technical expertise to optimize this catalyst-free process for your specific needs.
We invite you to collaborate with us to explore the full potential of this technology for your projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this novel route can improve your margins. Please contact us to request specific COA data for related analogues or to discuss route feasibility assessments for your target molecules. Let us help you secure a reliable supply of high-quality intermediates while optimizing your manufacturing costs.
