Advanced Catalyst-Free Synthesis of 3-Selenocyanoindoles for Commercial Pharmaceutical Applications
Advanced Catalyst-Free Synthesis of 3-Selenocyanoindoles for Commercial Pharmaceutical Applications
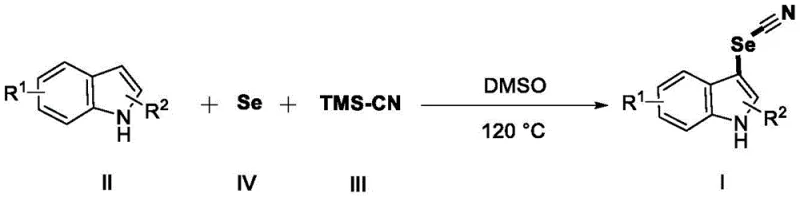
The landscape of heterocyclic chemistry is constantly evolving, driven by the need for more sustainable and cost-effective synthetic routes for bioactive scaffolds. A significant breakthrough in this domain is documented in Chinese patent CN109232363B, which discloses a novel method for synthesizing 3-selenocyanoindole compounds. This technology represents a paradigm shift by utilizing elemental selenium and trimethylsilyl cyanide (TMS-CN) as a unique selenocyano source, completely bypassing the need for transition metal catalysts or hazardous oxidants. For R&D directors and procurement specialists in the pharmaceutical and agrochemical sectors, this development offers a compelling value proposition: a streamlined pathway to access selenium-containing heterocycles that are pivotal in drug discovery for conditions ranging from leishmaniasis to viral infections. The simplicity of the protocol, operating at 120°C in dimethyl sulfoxide (DMSO), underscores its potential for immediate adoption in both laboratory screening and large-scale manufacturing environments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of selenocyano groups into indole frameworks has been fraught with synthetic challenges and economic inefficiencies. Traditional methodologies often rely heavily on the use of stoichiometric amounts of expensive oxidants like tert-butyl hydroperoxide (TBHP) or hypervalent iodine reagents such as N-iodosuccinimide (NIS). These reagents not only inflate the raw material costs but also introduce significant safety hazards due to their potential explosiveness and instability during storage and handling. Furthermore, many prior art methods necessitate the presence of transition metal catalysts, such as copper salts, which pose a severe contamination risk for pharmaceutical intermediates. The removal of trace metal residues to meet stringent regulatory standards requires additional purification steps, such as chelation or extensive chromatography, thereby extending the overall production timeline and reducing the final yield. Additionally, some existing routes involve multi-step preparations of specialized selenocyanating agents, adding unnecessary complexity and waste generation to the process.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the method described in patent CN109232363B introduces a remarkably direct and atom-economical strategy. By leveraging elemental selenium, an abundant and inexpensive commodity chemical, alongside TMS-CN, the process achieves direct C-H functionalization at the C-3 position of the indole ring. This catalyst-free approach operates under an air atmosphere, eliminating the need for inert gas protection and specialized pressure vessels often required for sensitive organometallic reactions. The reaction conditions are mild yet robust, utilizing DMSO as a dual solvent and potential promoter, which facilitates the dissolution of reactants and stabilizes the transition states. This innovation effectively decouples the synthesis of high-value selenocyanoindoles from the supply chain volatility associated with precious metal catalysts and hazardous peroxides, offering a greener and more economically viable alternative for the production of complex nitrogen heterocycles.
Mechanistic Insights into Catalyst-Free Selenocyanation
The mechanistic underpinning of this transformation is a subject of great interest for process chemists aiming to optimize reaction parameters. While the exact pathway may involve radical or ionic intermediates facilitated by the polar aprotic solvent, the key feature is the activation of elemental selenium by TMS-CN to generate an active selenocyanating species in situ. Unlike traditional electrophilic selenylation which often requires pre-functionalized selenium reagents, this system likely generates a reactive Se-CN bond equivalent that attacks the electron-rich C-3 position of the indole nucleus. The absence of external oxidants suggests that the reaction might proceed through a unique redox-neutral manifold or utilize the solvent oxygen in a controlled manner, although the patent specifies operation in air without added oxidants. This mechanistic simplicity translates directly to operational robustness; without the need to fine-tune catalyst loading or manage exothermic oxidant addition, the process window is significantly widened, reducing the risk of batch-to-batch variability.
From an impurity control perspective, the catalyst-free nature of this reaction is a decisive advantage for pharmaceutical manufacturing. In conventional metal-catalyzed processes, side reactions often lead to metal-complexed byproducts or over-oxidized species that are difficult to separate. Here, the primary byproducts are likely volatile siloxanes or unreacted starting materials that can be easily removed during the aqueous workup and subsequent silica gel chromatography. The high regioselectivity observed across various substrates, including those with electron-withdrawing or electron-donating groups on the benzene ring, indicates a strong inherent preference for the C-3 position, minimizing the formation of regioisomers. This purity profile is critical for downstream applications, as it reduces the burden on analytical quality control teams and ensures that the final API intermediate meets the rigorous specifications required for clinical trials and commercial release.
How to Synthesize 3-Selenocyanoindole Efficiently
The practical execution of this synthesis is designed for ease of operation, making it accessible even to laboratories with standard equipment. The protocol involves a straightforward sequential addition of reagents followed by a single heating step, which simplifies the training requirements for operators and reduces the potential for human error. The detailed standardized synthesis steps below outline the precise molar ratios and workup procedures validated in the patent examples, ensuring reproducibility and high yields across a broad range of indole derivatives.
- Sequentially add indole compound, elemental selenium, and DMSO solvent into a reaction vessel and stir to dissolve.
- Add TMS-CN to the mixture and heat the reaction system to 120°C under air atmosphere until the starting material is consumed.
- Cool the reaction, extract with ethyl acetate, dry over sodium sulfate, and purify via column chromatography to obtain the target product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology translates into tangible strategic benefits that extend beyond mere technical feasibility. The elimination of costly catalysts and hazardous oxidants fundamentally alters the cost structure of producing selenocyanoindole intermediates. By relying on bulk commodities like elemental selenium and TMS-CN, manufacturers can insulate themselves from the price volatility of precious metals and specialty reagents. Furthermore, the simplified workup procedure, which avoids complex metal scavenging steps, leads to a drastic reduction in processing time and solvent consumption. This efficiency gain allows for faster turnover of production batches, enhancing the overall agility of the supply chain and enabling quicker response times to market demands for novel therapeutic candidates.
- Cost Reduction in Manufacturing: The most significant economic driver of this process is the complete removal of transition metal catalysts and expensive oxidants from the bill of materials. Traditional methods often incur high costs due to the purchase of palladium, copper, or iodine reagents, as well as the disposal of heavy metal waste. By switching to a catalyst-free system, the direct material costs are significantly lowered, and the indirect costs associated with waste treatment and environmental compliance are also minimized. This lean manufacturing approach ensures that the final product remains cost-competitive even when produced at smaller scales, facilitating early-stage drug development without prohibitive expenses.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, specifically elemental selenium and TMS-CN, are widely available from multiple global suppliers, reducing the risk of single-source dependency. Unlike specialized catalysts that may have long lead times or limited availability, these commoditized chemicals can be sourced reliably in large quantities. This abundance ensures continuity of supply, which is critical for maintaining uninterrupted production schedules for key pharmaceutical intermediates. Additionally, the stability of these reagents simplifies inventory management, as they do not require the stringent storage conditions often mandated for sensitive organometallic complexes or peroxides.
- Scalability and Environmental Compliance: The operational simplicity of running the reaction at 120°C in DMSO makes this process highly amenable to scale-up from gram to ton quantities. The absence of explosive oxidants removes a major safety barrier that often limits the batch size in conventional protocols. From an environmental standpoint, the reduction in hazardous waste generation aligns with green chemistry principles, helping companies meet increasingly strict regulatory standards for emissions and effluent discharge. This sustainability profile not only mitigates regulatory risk but also enhances the corporate reputation of manufacturers committed to eco-friendly production practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the method's capabilities and limitations for potential adopters.
Q: What are the advantages of this selenium cyanation method over traditional catalytic routes?
A: This method eliminates the need for expensive transition metal catalysts like copper or iodine-based oxidants such as TBHP, significantly reducing raw material costs and simplifying the purification process by avoiding metal residue removal.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the process utilizes commercially available elemental selenium and operates under relatively mild thermal conditions (120°C) in a common solvent (DMSO), making it highly suitable for scale-up without requiring specialized high-pressure equipment.
Q: What is the regioselectivity of the selenocyanation reaction?
A: The reaction exhibits excellent regioselectivity, specifically targeting the C-3 position of the indole ring, which is crucial for maintaining the structural integrity required for downstream biological activity in pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Selenocyanoindole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this catalyst-free synthesis route for the production of high-value selenium-containing heterocycles. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific thermal and solvent requirements of this process, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch of 3-selenocyanoindole meets the highest industry standards. We are committed to delivering consistent quality and reliability, serving as a trusted extension of your own R&D and manufacturing teams.
We invite you to explore how this innovative technology can optimize your supply chain and reduce your overall manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our expertise can accelerate your development timelines and secure your supply of critical pharmaceutical intermediates.
