Advanced Manufacturing of High-Purity 2,7-Dibromocarbazole for Commercial Scale-Up
The rapid evolution of the organic electronics sector has placed immense pressure on the supply chain for high-performance hole transport materials and luminescent precursors. Among these critical building blocks, 2,7-dibromocarbazole stands out as a pivotal intermediate for constructing rigid, conjugated polymer backbones essential for next-generation OLED displays and organic photovoltaics. A groundbreaking technical disclosure found in patent CN102875447B details a robust, two-step synthetic methodology that fundamentally redefines the production economics and safety profile of this valuable compound. By shifting away from hazardous, high-energy conventional protocols, this innovation introduces a streamlined pathway utilizing inexpensive iron or aluminum salt catalysts and mild nitration conditions. For R&D directors and procurement strategists alike, this represents a significant opportunity to secure a reliable 2,7-dibromocarbazole supplier capable of delivering consistent quality while mitigating the environmental and operational risks associated with traditional manufacturing.
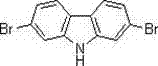
The structural integrity of the final product is paramount for its performance in electronic applications, where even trace impurities can quench luminescence or degrade charge mobility. The patented approach ensures high purity through a controlled nitration sequence followed by a selective cyclization, avoiding the formation of complex byproduct mixtures often seen in harsher acidic environments. This level of control is not merely a laboratory curiosity but a commercial necessity for manufacturers aiming to reduce lead time for high-purity electronic chemical intermediates. As the demand for flexible displays and efficient solar cells grows, the ability to produce such precursors with minimal environmental footprint and maximum atom economy becomes a decisive competitive advantage in the global fine chemicals market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of carbazole derivatives has been plagued by severe operational constraints that hinder scalability and increase total cost of ownership. Prior art, such as the methods described by Freeman and Urvoy, typically relies on the use of o-dichlorobenzene as a solvent and triphenylphosphine as a deoxygenating agent, necessitating reaction temperatures as high as 180°C. Such extreme thermal requirements impose heavy burdens on reactor design, demanding specialized heating systems and robust cooling capabilities to manage exotherms safely. Furthermore, the nitration step in traditional routes often employs glacial acetic acid and fuming nitric acid, creating a highly corrosive environment that accelerates equipment degradation and poses significant safety hazards to personnel. The combination of high boiling point solvents and aggressive reagents makes downstream processing arduous, as removing residual solvents requires substantial energy input, thereby inflating utility costs and extending batch cycle times significantly.
The Novel Approach
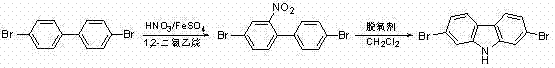
In stark contrast, the methodology outlined in the patent data introduces a paradigm shift towards milder, more sustainable processing conditions that directly address these historical pain points. By utilizing 1,2-dichloroethane as the solvent for the nitration step and maintaining temperatures between 80°C and 84°C, the process drastically reduces thermal stress on the system while maintaining excellent conversion rates. The subsequent cyclization step is even more remarkable, proceeding efficiently at room temperature in dichloromethane, which eliminates the need for energy-intensive heating during the critical ring-closing phase. This novel approach not only simplifies the operational workflow but also enhances the safety profile by replacing fuming nitric acid with dilute nitric acid and utilizing cost-effective iron or aluminum salts as catalysts. The result is a streamlined synthesis that facilitates cost reduction in electronic chemical manufacturing by minimizing energy consumption and simplifying waste treatment protocols.
Mechanistic Insights into Fe-Catalyzed Nitration and Phosphine-Mediated Cyclization
The success of this synthetic route lies in the precise orchestration of electrophilic aromatic substitution and intramolecular cyclization mechanisms. In the first stage, the presence of iron or aluminum salts acts as a Lewis acid catalyst, activating the dilute nitric acid to generate the active nitronium ion species required for the nitration of 4,4'-dibromobiphenyl. This catalytic activation allows the reaction to proceed rapidly at moderate temperatures, ensuring regioselectivity that favors the formation of the 2-nitro-4,4'-dibromobiphenyl intermediate. The choice of 1,2-dichloroethane as the solvent is critical here, as it provides an optimal polarity balance that stabilizes the transition state while remaining immiscible with the aqueous acid phase, facilitating easy separation and recycling of the organic layer. This mechanistic efficiency translates directly into higher yields, with experimental data indicating conversion rates exceeding 90%, thereby maximizing raw material utilization.
The second stage involves a sophisticated deoxygenative cyclization driven by specialized phosphine reagents such as 2-diphenylphosphine-biphenyl or tri(o-methylphenyl)phosphine. Unlike traditional triphenylphosphine, these advanced deoxygenating agents exhibit superior nucleophilicity and oxygen affinity, enabling them to abstract the oxygen atom from the nitro group under ambient conditions. This abstraction triggers an intramolecular attack by the adjacent aromatic ring, closing the carbazole skeleton without the need for external thermal activation. The mechanism effectively bypasses the high-energy barriers associated with conventional thermal cyclization, resulting in a cleaner reaction profile with fewer side products. For quality control teams, this means a simplified purification process, often requiring only standard silica gel chromatography to achieve the stringent purity specifications demanded by the optoelectronics industry.
How to Synthesize 2,7-Dibromocarbazole Efficiently
Implementing this synthesis protocol requires careful attention to reagent stoichiometry and phase separation techniques to ensure optimal outcomes. The process begins with the nitration of the biphenyl precursor, where maintaining the temperature window of 80-84°C is crucial to prevent over-nitration or decomposition. Following the isolation of the nitro-intermediate, the cyclization step demands an inert atmosphere, typically nitrogen, to protect the sensitive phosphine reagents from oxidation before they can react with the substrate. Detailed standardized operating procedures regarding mixing speeds, addition rates, and workup protocols are essential for transferring this chemistry from the bench to the pilot plant.
- Nitration of 4,4'-dibromobiphenyl using dilute nitric acid and iron/aluminum salts in 1,2-dichloroethane at 80-84°C.
- Isolation of 2-nitro-4,4'-dibromobiphenyl intermediate via extraction and recrystallization.
- Cyclization using specialized phosphine deoxygenating agents in dichloromethane at room temperature to yield 2,7-dibromocarbazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers tangible benefits that extend far beyond simple yield improvements. The shift to milder reaction conditions and cheaper catalysts fundamentally alters the cost structure of production, making the supply of this critical intermediate more resilient against market volatility. By eliminating the need for high-temperature operations and hazardous fuming acids, manufacturers can significantly reduce their reliance on specialized, high-maintenance equipment, leading to lower capital expenditure and maintenance overheads. Furthermore, the use of common, recyclable solvents like dichloromethane and 1,2-dichloroethane simplifies logistics and waste management, ensuring compliance with increasingly strict environmental regulations without compromising throughput.
- Cost Reduction in Manufacturing: The replacement of expensive or hazardous reagents with inexpensive iron salts and dilute nitric acid creates a direct pathway for substantial cost savings in raw material procurement. Additionally, the elimination of high-temperature heating requirements for the cyclization step results in drastic reductions in energy consumption, as reactors do not need to be maintained at 180°C for extended periods. This energy efficiency, combined with the high yields reported in the patent examples, ensures that the overall cost per kilogram of the final product is significantly optimized, providing a competitive edge in pricing negotiations.
- Enhanced Supply Chain Reliability: The use of readily available starting materials and catalysts reduces the risk of supply disruptions caused by the scarcity of specialized reagents. Since the process operates under milder conditions, the risk of unplanned shutdowns due to equipment failure or safety incidents is minimized, ensuring a more consistent and predictable production schedule. This reliability is crucial for downstream customers in the display and pharmaceutical sectors who depend on just-in-time delivery models to maintain their own production lines without interruption.
- Scalability and Environmental Compliance: The simplified workup procedures, involving straightforward phase separations and solvent recovery, make this process highly amenable to scale-up from kilogram to multi-ton quantities. The reduced generation of hazardous waste, particularly the avoidance of heavy metal contaminants and aggressive acid residues, streamlines the environmental permitting process and lowers waste disposal costs. This alignment with green chemistry principles not only enhances the corporate sustainability profile but also future-proofs the manufacturing operation against tightening global environmental standards.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its potential for integration into existing supply chains. The following questions address common concerns regarding process safety, scalability, and product quality, drawing directly from the technical disclosures and experimental data provided in the patent documentation. These insights are intended to clarify the operational advantages and assist decision-makers in assessing the feasibility of adopting this technology for commercial production.
Q: What are the key advantages of this synthesis method over conventional high-temperature routes?
A: This method eliminates the need for extreme temperatures (180°C) and hazardous fuming nitric acid, utilizing mild conditions (80°C and room temperature) which significantly enhances operational safety and reduces energy consumption.
Q: How does the choice of deoxygenating agent impact the reaction efficiency?
A: The use of specialized phosphines like 2-diphenylphosphine-biphenyl provides superior deoxygenation capability compared to traditional triphenylphosphine, facilitating easier ring closure and higher overall yields without requiring harsh thermal activation.
Q: Is this process suitable for large-scale industrial production of electronic chemicals?
A: Yes, the process utilizes common solvents like 1,2-dichloroethane and dichloromethane which are easily recoverable, and the mild reaction conditions simplify heat management, making it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,7-Dibromocarbazole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to industrial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising metrics observed in patent literature are faithfully reproduced at scale. We are committed to maintaining stringent purity specifications through our rigorous QC labs, employing advanced analytical techniques to verify that every batch of 2,7-dibromocarbazole meets the exacting standards required for high-performance OLED and pharmaceutical applications.
We invite you to collaborate with us to explore how this optimized synthesis route can enhance your product portfolio and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation and quality can support your long-term strategic goals in the fine chemicals sector.
