Revolutionizing Pharmaceutical Intermediate Production Through Efficient N-Acyl Indole Synthesis Technology
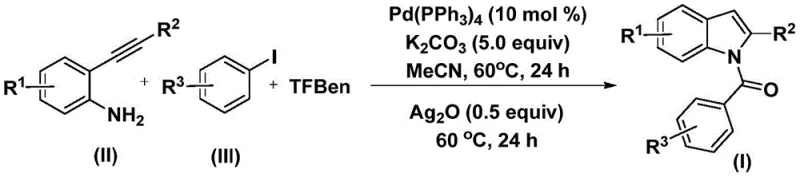
Patent CN112898192B introduces a groundbreaking methodology for synthesizing N-acyl indole compounds that serve as critical structural motifs in numerous pharmaceutical agents including anti-inflammatory drugs like Indomethacin and anti-HIV therapeutics such as Delavirdine. This innovative process leverages palladium-catalyzed carbonylation cyclization to achieve high-yielding production under mild conditions of two sequential reactions at a consistent temperature of exactly 60°C over forty-eight hours total duration. Unlike traditional approaches requiring harsh reagents or multiple synthetic steps under high-pressure carbon monoxide environments, this single-pot strategy utilizes readily available starting materials such as aryl iodides and terminal alkynes while employing tetrakis(triphenylphosphine)palladium as an efficient catalyst system. The method demonstrates exceptional substrate tolerance across diverse functional groups including halogens and alkyl substituents, thereby significantly broadening its applicability in complex molecule construction for pharmaceutical development pipelines. Furthermore, the elimination of high-pressure carbon monoxide through triphenylphosphine palladium catalysis enhances operational safety while maintaining excellent reaction efficiency across various substitution patterns on both coupling partners.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for N-acyl indole compounds typically require multiple protection/deprotection steps that significantly reduce overall yield while introducing complex purification challenges that compromise product purity standards required by pharmaceutical manufacturers. Conventional carbonylation methods often operate under high-pressure carbon monoxide conditions that necessitate specialized equipment and stringent safety protocols, creating substantial barriers to industrial scale-up and increasing capital expenditure requirements for manufacturing facilities. These approaches frequently exhibit poor functional group tolerance when encountering halogenated or sterically hindered substrates common in complex pharmaceutical intermediates, leading to inconsistent product quality and batch-to-batch variability that disrupts supply chain continuity. Additionally, many existing methodologies rely on expensive transition metal catalysts that require extensive removal procedures during purification stages, generating significant waste streams that conflict with modern environmental compliance standards while adding considerable cost burdens to production processes.
The Novel Approach
The patented methodology overcomes these limitations through an elegant single-pot cascade reaction that integrates carbonylation and cyclization into one streamlined process using TFBen as a stable carbon monoxide surrogate that releases CO under mild thermal conditions without requiring pressurized systems. By operating at precisely controlled temperatures of just 60°C throughout both reaction stages, this approach eliminates energy-intensive heating/cooling cycles while maintaining exceptional selectivity across diverse substrate combinations including those containing sensitive functional groups like halogens and alkoxy moieties. The strategic use of silver oxide as an oxidant enables efficient ring closure without generating hazardous byproducts that complicate waste management procedures in industrial settings. Crucially, this method achieves high yields while utilizing commercially available starting materials with broad global supply chains, thereby enhancing production reliability while reducing raw material costs through simplified procurement logistics compared to conventional approaches requiring specialized reagents.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cyclization
The palladium-catalyzed carbonylation cyclization mechanism begins with oxidative addition of aryl iodide to Pd(0) to form an arylpalladium intermediate, followed by CO insertion from TFBen to generate an acylpalladium species through controlled release of carbon monoxide under thermal activation at precisely maintained temperatures. Subsequent nucleophilic attack by the amine group of ortho-alkynylaniline triggers cyclization through alkyne insertion and reductive elimination to yield an amide intermediate with excellent regioselectivity dictated by the spatial orientation of functional groups on both coupling partners. The final ring closure is facilitated by silver oxide oxidation under mild thermal conditions at exactly 60°C without requiring additional catalysts or harsh reagents that could compromise product integrity. This cascade process demonstrates remarkable efficiency due to synergistic effects between palladium catalyst and silver oxide co-catalyst that promote both carbonylation and oxidation steps within a single reaction vessel while maintaining consistent performance across diverse substitution patterns on both coupling partners.
Impurity control is achieved through the method's inherent selectivity that minimizes side reactions typically observed in conventional syntheses involving multiple intermediate isolations where impurities can accumulate during processing stages. The single-pot nature prevents exposure to atmospheric contaminants during transfer operations between reaction vessels while maintaining consistent temperature profiles that prevent thermal degradation pathways common in multi-step processes requiring significant heating/cooling cycles. Functional group tolerance across halogens and alkyl substituents ensures minimal formation of regioisomeric impurities that would otherwise require extensive purification procedures affecting overall yield and product quality consistency. Furthermore, the absence of transition metal residues beyond catalytic quantities eliminates potential contamination sources that could impact final product specifications required by stringent pharmaceutical quality control standards.
How to Synthesize N-Acyl Indole Compound Efficiently
This patented methodology provides a robust framework for synthesizing high-purity N-acyl indole compounds through a precisely controlled two-stage reaction sequence that maintains exceptional reproducibility across various scales from laboratory development through commercial manufacturing environments. The process leverages commercially available starting materials with straightforward handling requirements while operating under ambient pressure conditions that eliminate safety concerns associated with traditional high-pressure carbonylation methods. Detailed standardized synthesis procedures have been developed based on extensive optimization studies documented in patent CN112898192B to ensure consistent product quality meeting pharmaceutical industry specifications across different production volumes.
- Combine palladium catalyst Pd(PPh₃)₄ (0.02 mmol), potassium carbonate (5.0 equiv), TFBen (5.0 equiv), substituted ortho-alkynylaniline (II) (0.2 mmol), and aryl iodide (III) (0.2 mmol) in acetonitrile solvent within a Schlenk tube under inert atmosphere.
- Heat the reaction mixture at precisely 60°C for twenty-four hours to facilitate oxidative addition and carbonylation steps using TFBen as carbon monoxide surrogate.
- Add silver oxide (0.5 equiv) to the reaction mixture and continue heating at identical temperature conditions for another twenty-four hours to complete cyclization and oxidation.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology directly addresses critical pain points in pharmaceutical intermediate procurement by transforming complex multi-step processes into streamlined single-pot operations that significantly reduce raw material requirements while enhancing production reliability through simplified logistics management protocols. The elimination of specialized equipment needs associated with high-pressure carbon monoxide systems reduces capital expenditure barriers while improving facility flexibility for manufacturers seeking to diversify their intermediate production capabilities without major infrastructure investments.
- Cost Reduction in Manufacturing: The strategic substitution of hazardous carbon monoxide gas with stable TFBen surrogate eliminates costly safety infrastructure requirements while reducing waste treatment expenses through minimized byproduct generation during purification stages. Simplified post-treatment procedures involving standard filtration and column chromatography replace complex metal removal processes typically required when using alternative transition metal catalysts, thereby substantially lowering operational costs without compromising product quality standards.
- Enhanced Supply Chain Reliability: Utilization of globally available starting materials including aryl iodides and terminal alkynes ensures consistent raw material availability through multiple qualified suppliers worldwide while avoiding single-source dependencies that create vulnerability points in traditional intermediate manufacturing processes. The robust nature of this methodology maintains consistent performance across different batches even when using materials from alternative vendors due to its exceptional functional group tolerance characteristics.
- Scalability and Environmental Compliance: The ambient pressure operation enables seamless scale-up from laboratory quantities to commercial production volumes without requiring specialized equipment modifications while maintaining identical reaction parameters throughout all scales. Reduced waste generation through minimized side reactions combined with elimination of hazardous reagents creates significant environmental benefits that align with modern sustainability initiatives while simplifying regulatory compliance procedures across global manufacturing sites.
Frequently Asked Questions (FAQ)
The following questions address critical technical considerations raised by procurement teams evaluating this patented methodology for integration into their pharmaceutical intermediate supply chains based on detailed analysis of patent CN112898192B's technical specifications and implementation requirements.
Q: How does this method improve upon conventional carbonylation approaches for indole synthesis?
A: This patented method eliminates high-pressure carbon monoxide requirements through TFBen as a safe CO surrogate while operating at mild temperatures of just 60°C. The single-pot process avoids multiple isolation steps that typically cause yield loss in conventional methods, with demonstrated compatibility across diverse functional groups including halogens and alkyl substituents that often complicate traditional syntheses.
Q: What specific advantages does this process offer for large-scale pharmaceutical manufacturing?
A: The process utilizes commercially available starting materials with straightforward post-treatment involving filtration and column chromatography rather than complex purification techniques. The consistent reaction conditions at ambient pressure enable seamless scale-up from laboratory to industrial production volumes while maintaining high product purity essential for pharmaceutical applications.
Q: How does this methodology address critical supply chain challenges in intermediate production?
A: By employing readily accessible aryl iodides and terminal alkynes as starting materials with broad commercial availability, this method reduces dependency on specialized reagents that create supply bottlenecks. The simplified two-stage reaction sequence minimizes processing time while eliminating hazardous reagents that typically complicate logistics and regulatory compliance in global supply chains.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Acyl Indole Compound Supplier
Our company possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through rigorous QC labs equipped with advanced analytical capabilities specifically designed for complex heterocyclic compounds like N-acyl indoles. As a leading CDMO specialist in pharmaceutical intermediates manufacturing, we have successfully implemented this patented methodology across multiple client projects while ensuring seamless technology transfer from laboratory development through full-scale commercial manufacturing operations.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team who will provide specific COA data and route feasibility assessments tailored to your unique production requirements while demonstrating how this innovative synthesis approach can optimize your intermediate supply chain operations.
