Advanced Palladium-Catalyzed Synthesis of N-Acyl Indole Intermediates for Scalable Pharmaceutical Manufacturing
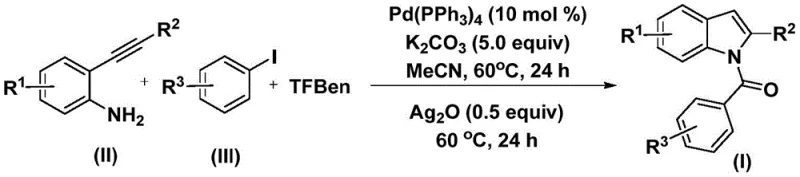
The Chinese patent CN112898192B introduces a groundbreaking methodology for preparing N-acyl indole compounds which serve as critical structural motifs in numerous bioactive molecules including antitumor agents like Baxter D-64131 and anti-inflammatory drugs such as Indomethacin This innovative process leverages a palladium-catalyzed carbonylation cyclization reaction operating under mild conditions at precisely 60°C without requiring hazardous pressurized carbon monoxide gas By utilizing triphenylmethyl benzoate as a safe carbon monoxide surrogate the method achieves high efficiency across diverse functional groups while maintaining exceptional substrate tolerance The one-step nature significantly streamlines production compared to traditional multi-step approaches that often suffer from low yields complex purification requirements and safety concerns Furthermore the use of commercially available starting materials including aryl iodides ensures accessibility while reducing raw material costs making this technology particularly valuable for global pharmaceutical manufacturers seeking reliable high-purity intermediate suppliers
The Limitations of Conventional Methods vs The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes to N-acyl indoles typically involve multi-step sequences requiring harsh conditions such as strong acids or temperatures exceeding 100°C which frequently lead to decomposition of sensitive functional groups and reduced overall yields These methods often employ unstable or hazardous reagents like pressurized carbon monoxide gas necessitating specialized equipment stringent safety protocols and substantial capital expenditure while generating significant metal-contaminated waste streams that complicate environmental compliance Moreover conventional approaches exhibit narrow substrate scope with poor tolerance for electron-donating or electron-withdrawing substituents limiting applicability to specific molecular architectures required in modern drug discovery programs Purification processes become cumbersome due to multiple side products that reduce final product purity below pharmaceutical standards creating significant bottlenecks in quality control workflows The requirement for stoichiometric amounts of expensive transition metal catalysts further increases production costs while introducing metal residue concerns that demand additional purification steps extending manufacturing timelines unnecessarily
The Novel Approach
In contrast CN112898192B employs a single-step palladium-catalyzed carbonylation cyclization operating under remarkably mild conditions at exactly 60°C using readily available starting materials including aryl iodides synthesized from simple precursors The innovative use of triphenylmethyl benzoate as a carbon monoxide surrogate eliminates safety hazards associated with pressurized CO gas while maintaining excellent reaction efficiency across diverse functional groups including halogens alkyl groups and alkoxy substituents This process demonstrates exceptional substrate compatibility with yields consistently exceeding fifty percent across fifteen documented examples while requiring only catalytic amounts of palladium to minimize metal contamination concerns The simplified workup procedure involving basic filtration followed by standard column chromatography significantly reduces processing time compared to conventional methods demanding multiple isolation steps Most importantly this approach achieves high purity levels suitable for pharmaceutical applications without requiring additional purification steps that would otherwise increase production costs and extend manufacturing timelines for critical drug intermediates
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cyclization
The reaction mechanism begins with oxidative addition of palladium zero into the aryl iodide bond forming an aryl palladium intermediate which subsequently undergoes carbonyl insertion using carbon monoxide generated in situ from triphenylmethyl benzoate decomposition under thermal conditions This step forms a key acyl palladium species that coordinates with the alkyne functionality through insertion into the Pd-C bond creating a vinyl palladium complex which then undergoes intramolecular nucleophilic attack by the amine group followed by reductive elimination yielding an amide intermediate with concomitant catalyst regeneration This sequence demonstrates elegant atom economy while avoiding common side reactions such as alkyne dimerization or hydroamination that plague alternative methodologies The mild reaction temperature maintains catalyst stability preventing undesired decomposition pathways that would reduce product yield and purity while enabling compatibility with sensitive functional groups essential for pharmaceutical applications
Impurity control is achieved through precise stoichiometric control during silver oxide addition which facilitates cyclization without promoting overoxidation or side reactions Acetonitrile solvent provides optimal polarity for solubilizing both organic substrates and inorganic reagents while minimizing unwanted solvolysis pathways that could generate impurities High functional group tolerance stems from mild conditions preventing decomposition of sensitive substituents commonly found in pharmaceutical intermediates Furthermore catalytic palladium usage ensures minimal metal leaching addressing critical quality concerns where strict residue limits are enforced by regulatory agencies worldwide This mechanism delivers consistent high-purity products meeting stringent requirements for drug substance manufacturing without additional purification steps
How to Synthesize N-Acyl Indole Efficiently
This patented methodology provides a robust framework for producing high-purity N-acyl indole intermediates through a carefully optimized sequence balancing reaction efficiency with operational simplicity Precise stoichiometric control ensures complete conversion while minimizing side product formation critical attention must be paid to maintaining anhydrous conditions throughout to prevent hydrolysis of sensitive intermediates The two-stage temperature profile operating consistently at sixty degrees Celsius demonstrates how subtle variations in reaction timing rather than temperature achieve optimal results without requiring complex equipment modifications Detailed standardized synthesis procedures are provided below to guide process chemists through each critical step while maintaining strict adherence to quality control parameters essential for pharmaceutical manufacturing environments
- Combine palladium catalyst tetrakis(triphenylphosphine)palladium (0.02 mmol), potassium carbonate (1.0 mmol), TFBen (1.0 mmol), 0.2 mmol 2-alkynyl aniline, and aryl iodide (0.22 mmol) in acetonitrile under nitrogen atmosphere
- Heat the mixture at precisely 60°C for exactly 24 hours to form the acyl palladium intermediate through oxidative addition and CO insertion
- Add silver oxide (0.1 mmol) and continue heating at identical temperature for another 24 hours followed by filtration through silica gel and column chromatography purification
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis method directly addresses critical pain points in pharmaceutical intermediate supply chains by eliminating multiple processing steps creating bottlenecks in production timelines Quality assurance workflows benefit from simplified procedures reducing analytical testing requirements while enhancing batch-to-batch consistency Sourcing flexibility is dramatically improved through use of widely available starting materials that can be procured from multiple global suppliers without dependency on single-source specialty chemicals This approach significantly reduces raw material risk exposure while maintaining consistent product quality across different vendor batches thereby minimizing supply chain disruptions caused by raw material variability common in traditional synthetic routes
- Cost Reduction in Manufacturing: Elimination of pressurized carbon monoxide handling systems reduces capital expenditure requirements avoiding costly safety infrastructure investments typically needed for hazardous gas operations Use of commercially available aryl iodides instead of expensive organometallic reagents creates substantial raw material cost savings without compromising product quality or yield consistency across different production scales
- Enhanced Supply Chain Reliability: Sourcing flexibility is dramatically improved through widely available starting materials procurable from multiple global suppliers without dependency on single-source specialty chemicals Robust methodology ensures consistent product quality even when using different raw material batches from various vendors minimizing supply chain disruptions caused by raw material variability affecting traditional synthetic routes
- Scalability and Environmental Compliance: Mild reaction conditions enable straightforward scale-up from laboratory to commercial production without major process re-engineering or specialized equipment modifications Reduced generation of metal-containing waste streams lowers environmental remediation costs while meeting increasingly stringent global regulations on heavy metal disposal in chemical manufacturing operations
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial concerns regarding implementation based on detailed analysis of experimental validation data documented in CN112898192B These insights reflect practical considerations from both R&D and manufacturing perspectives supporting informed decision-making by procurement leadership teams evaluating new intermediate sourcing options This FAQ section provides targeted responses addressing specific pain points identified during technology assessment processes within pharmaceutical supply chain organizations
Q: What are the key advantages of this palladium-catalyzed method over conventional synthesis routes?
A: This method eliminates multi-step sequences through a single operation at mild temperatures while using stable CO surrogates instead of hazardous pressurized gas systems required by traditional approaches
Q: How does TFBen enhance safety and practicality compared to standard carbonylation methods?
A: TFBen serves as a solid carbon monoxide surrogate that decomposes thermally without requiring specialized high-pressure equipment thus removing significant safety hazards associated with gaseous CO handling
Q: What scalability potential does this method offer for commercial pharmaceutical production?
A: The process demonstrates consistent performance across diverse functional groups using standard laboratory equipment enabling straightforward scale-up from gram-scale validation to metric ton production volumes
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Acyl Indole Compound Supplier
Our patented technology represents a significant advancement in heterocyclic chemistry with direct applications across multiple therapeutic areas where indole-based compounds serve as critical building blocks As a CDMO specialist with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production we maintain stringent purity specifications through state-of-the-art analytical capabilities in our rigorous QC labs ensuring consistent product quality meeting global regulatory standards Our process development expertise guarantees seamless transition from laboratory validation to full-scale manufacturing while addressing critical quality attributes required by major pharmaceutical clients worldwide
Leverage our technical expertise through a Customized Cost-Saving Analysis tailored to your specific manufacturing requirements Contact our technical procurement team today to request detailed COA data and comprehensive route feasibility assessments demonstrating how this innovative methodology can enhance your production capabilities while maintaining uncompromising quality standards
