Scaling High-Purity Linalool Production via Continuous Isomerization Technology
The chemical landscape of terpene alcohol synthesis is undergoing a significant transformation, driven by the need for more efficient and scalable manufacturing processes. A pivotal development in this field is documented in patent CN1264789C, which details a robust method for the isomerization of reactant allylic alcohols, specifically focusing on the conversion of geraniol and nerol into linalool. This technology represents a departure from traditional batch processing, introducing semi-continuous and continuous reactor systems that utilize specialized tungsten-based catalysts to achieve superior space-time yields. For industry stakeholders, this innovation addresses critical bottlenecks in the production of high-purity fragrance intermediates, offering a pathway to more consistent quality and reduced operational complexity. The ability to drive equilibrium reactions forward through simultaneous distillation marks a substantial technical advancement, ensuring that manufacturers can meet the rigorous demands of the global flavors and fragrances market with greater reliability and economic efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the isomerization of allyl alcohols such as geraniol has been constrained by the inherent inefficiencies of batch processing methods. In traditional setups, the reaction is allowed to proceed to equilibrium in a closed vessel, which often results in incomplete conversion and the accumulation of high-boiling byproducts that can inhibit catalyst activity over time. Furthermore, the necessity to interrupt the process for the removal of products or the addition of fresh catalyst leads to significant downtime and inconsistent batch-to-batch quality. These operational interruptions not only extend the overall lead time for production but also increase the energy consumption per unit of product, as the reactor must be repeatedly heated and cooled. Additionally, conventional methods often require highly purified starting materials, necessitating expensive pre-treatment steps to remove impurities like iso-nerols that are commonly present in crude streams from citral hydrogenation, thereby inflating the overall cost of goods sold.
The Novel Approach
The novel approach described in the patent data overcomes these historical constraints by implementing a dynamic continuous or semi-continuous flow system. By continuously metering the reactant mixture into a reactor containing the catalyst while simultaneously distilling off the lower-boiling product, linalool, the system effectively shifts the chemical equilibrium towards the desired product. This continuous removal prevents the reverse reaction and minimizes the residence time of the product in the reaction zone, reducing the formation of degradation byproducts. The process is designed to handle crude feedstocks directly, tolerating impurities that would typically foul a batch reactor, which simplifies the upstream supply chain requirements. Moreover, the ability to continuously manage catalyst activity and remove high-boiling residues ensures a stable and prolonged operational cycle, significantly enhancing the overall productivity and economic viability of large-scale linalool manufacturing facilities.
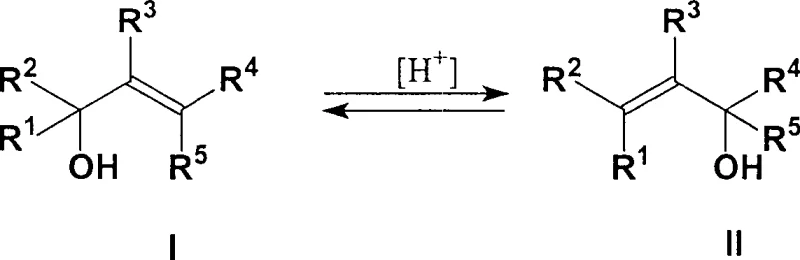
Mechanistic Insights into Tungsten-Catalyzed Isomerization
The core of this technological advancement lies in the specific catalytic mechanism employed, which utilizes dioxo tungsten (VI) complexes to facilitate the 1,3-migration of the hydroxyl group and the double bond. Unlike simple acid catalysts that may promote unwanted dehydration or cyclization side reactions, these tungsten complexes offer a controlled acidic environment that favors the rearrangement of the allylic system. The catalyst structure, characterized by ligands such as amino alcohols or amino phenols coordinated to the tungsten center, provides the necessary stability and activity at elevated temperatures ranging from 150°C to 200°C. This specific coordination chemistry ensures that the isomerization proceeds with high selectivity, minimizing the formation of undesirable terpene hydrocarbons or ethers that often contaminate products from less sophisticated catalytic systems. The robustness of the catalyst under continuous flow conditions is a critical factor, allowing for sustained activity over long operational periods without rapid deactivation.
Impurity control is another critical aspect of the mechanistic design, particularly when dealing with feedstocks derived from citral hydrogenation which contain isomers like iso-nerol 1 and iso-nerol 2. The continuous process allows for the strategic management of these byproducts; while they may accumulate in the reactor residue, the continuous distillation of linalool ensures they do not contaminate the final distillate. The system is engineered to separate these higher-boiling impurities from the catalyst and the product stream, often through the use of thin-film evaporators or specific discharge protocols. This separation capability is vital for maintaining the stringent purity specifications required by the fragrance industry, where even trace impurities can alter the olfactory profile of the final product. By understanding the boiling point differentials and the chemical stability of these impurities under reaction conditions, the process ensures a high-purity output suitable for direct use in fine fragrance formulations.

How to Synthesize Linalool Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize the efficiency of the isomerization process. The operation begins with the preparation of the reactor system, ensuring that the tungsten catalyst is properly dissolved and distributed within the reaction zone. The feedstock, typically a mixture of geraniol and nerol, is then metered into the reactor at a rate that matches the distillation rate of the product, maintaining a constant liquid volume and optimal residence time. Temperature and pressure must be carefully regulated, usually operating under reduced pressure to facilitate the removal of linalool at temperatures that do not compromise catalyst stability. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during scale-up operations.
- Prepare the reactor with a suitable tungsten-based catalyst solution and heat to 150-200°C under reduced pressure.
- Meter the geraniol/nerol mixture continuously into the reactor while simultaneously distilling off the formed linalool to drive equilibrium.
- Manage high-boiling byproducts by continuous removal via thin-film evaporation or periodic discharge to maintain catalyst activity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement and supply chain professionals, the adoption of this continuous isomerization technology translates into tangible strategic advantages regarding cost structure and supply reliability. The shift from batch to continuous processing inherently reduces the operational overhead associated with reactor turnover, cleaning, and setup, leading to a more streamlined manufacturing workflow. By enabling the use of unpurified raw materials directly from upstream hydrogenation units, the process eliminates the need for costly intermediate purification steps, thereby reducing the total cost of ownership for the raw materials. Furthermore, the continuous nature of the production allows for a more consistent output rate, which stabilizes inventory levels and reduces the risk of supply disruptions that are common with batch-dependent manufacturing schedules. This reliability is crucial for maintaining long-term contracts with downstream fragrance houses that demand consistent quality and uninterrupted supply.
- Cost Reduction in Manufacturing: The elimination of expensive purification steps for the starting material significantly lowers the input costs associated with production. Additionally, the continuous removal of the product drives the reaction to higher conversion rates without the need for excessive catalyst loading or prolonged reaction times, which reduces energy consumption and utility costs. The potential for catalyst recovery and reuse from the high-boiling residue further contributes to cost optimization by minimizing the consumption of precious metal complexes. These factors combine to create a more lean and cost-effective manufacturing process that enhances margin potential in a competitive market.
- Enhanced Supply Chain Reliability: Continuous processing capabilities allow for a steady and predictable production flow, which is essential for meeting the just-in-time delivery requirements of global supply chains. The ability to run the reactor for extended periods without interruption reduces the frequency of maintenance shutdowns and ensures a consistent availability of product. This stability mitigates the risk of stockouts and allows for better planning of logistics and distribution, ensuring that customers receive their orders on time and in full. The robustness of the process against feedstock variations also means that supply is less vulnerable to fluctuations in the quality of upstream raw materials.
- Scalability and Environmental Compliance: The design of the reactor system facilitates easy scale-up from pilot to commercial production volumes without the need for fundamental process changes, ensuring that technology transfer is smooth and efficient. The continuous removal of byproducts and the potential for catalyst recycling minimize the generation of hazardous waste, aligning the process with increasingly stringent environmental regulations. This compliance reduces the burden of waste disposal costs and enhances the sustainability profile of the manufacturing operation, which is a growing priority for corporate procurement strategies.
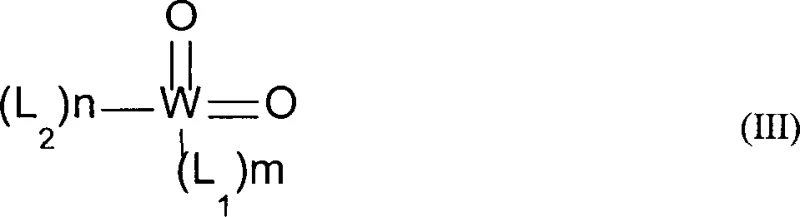
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this isomerization technology. They are derived from the specific operational parameters and benefits outlined in the patent documentation, providing clarity on how this method compares to existing industry standards. Understanding these details is essential for technical teams evaluating the feasibility of adopting this process for their specific production needs.
Q: How does the continuous process improve yield compared to batch methods?
A: The continuous process allows for the immediate removal of linalool via distillation as it forms. This shifts the chemical equilibrium towards the product side according to Le Chatelier's principle, preventing reverse reactions and significantly increasing space-time yield compared to static batch reactors.
Q: What catalyst is used for this isomerization and can it be recycled?
A: The process utilizes a dioxo tungsten (VI) complex catalyst. The patent describes methods to separate this catalyst from high-boiling residues, potentially allowing for recycling or reuse, which minimizes heavy metal waste and reduces raw material costs.
Q: Can unpurified geraniol from citral hydrogenation be used directly?
A: Yes, a key advantage of this method is its tolerance for impurities. It can process geraniol/nerol mixtures directly obtained from citral hydrogenation without prior purification, handling byproducts like iso-nerols effectively within the continuous flow system.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Linalool Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of advanced process technology in delivering high-value chemical intermediates to the global market. Our expertise extends beyond simple manufacturing; we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory concepts are successfully translated into robust industrial realities. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch against the highest industry standards. We understand that the transition to continuous processing requires a partner with deep technical knowledge and the infrastructure to support complex chemical transformations safely and efficiently.
We invite you to collaborate with us to optimize your supply chain for linalool and related terpene alcohols. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to reach out to request specific COA data and route feasibility assessments to determine how our advanced isomerization capabilities can enhance your product portfolio. By partnering with us, you gain access to a reliable supply of high-purity materials produced through cutting-edge technology that prioritizes both economic efficiency and environmental responsibility.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
