Optimizing Rilpivirine Production: Advanced Intermediate Synthesis for Commercial Scale
Optimizing Rilpivirine Production: Advanced Intermediate Synthesis for Commercial Scale
The global demand for second-generation non-nucleoside reverse transcriptase inhibitors (NNRTIs) continues to surge, driven by the critical need for effective HIV-1 treatment regimens with improved safety profiles. At the forefront of this therapeutic class is Rilpivirine, a molecule whose complex architecture demands precise synthetic strategies to ensure both economic viability and pharmaceutical grade quality. Patent CN103183642A introduces a groundbreaking approach to synthesizing key intermediates for Rilpivirine, addressing long-standing bottlenecks in yield and purification that have plagued previous methodologies. This technical insight report analyzes the proprietary pathway disclosed in the patent, highlighting its potential to redefine supply chain standards for reliable pharmaceutical intermediate suppliers. By leveraging a novel diamine intermediate structure, manufacturers can bypass tedious silica gel chromatography, achieving final product purity exceeding 99% through simple recrystallization.
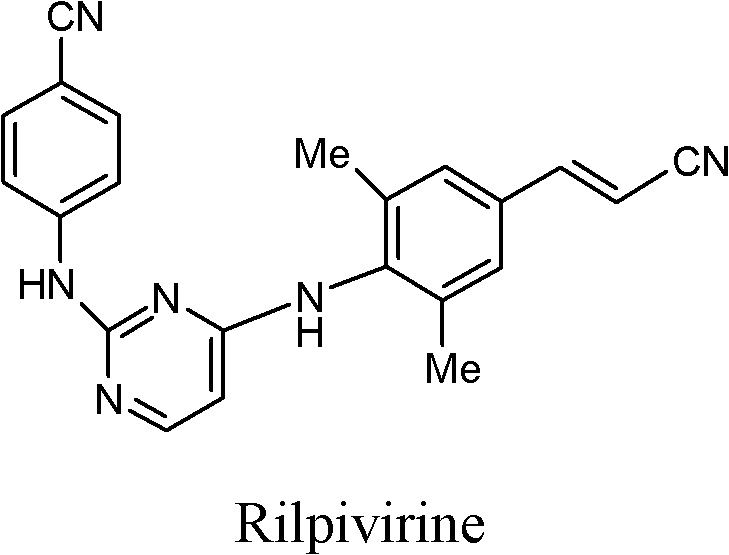
The structural integrity of Rilpivirine relies heavily on the precise coupling of its aromatic components, a challenge that this new intermediate technology resolves with elegance. The disclosed method not only streamlines the synthetic route but also ensures that the critical stereochemical and purity specifications required by regulatory bodies are met consistently. For R&D directors and procurement leaders, understanding the nuances of this pathway is essential for securing a competitive edge in the antiretroviral market. The following analysis dissects the technical merits of this innovation, contrasting it with legacy methods to demonstrate its superiority in cost reduction in API manufacturing and operational efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Rilpivirine has been hindered by inefficient routes that rely on expensive starting materials and generate significant amounts of difficult-to-separate impurities. Prior art, such as the routes reported in J. Med. Chem. 2005 and patent WO2004016581A1, often necessitates the use of costly reagents like 4-(4-chloro-pyrimidinyl-2-ylamino)-benzonitrile, which drives up the overall cost of goods. Furthermore, these conventional pathways frequently result in the formation of cis/trans isomer mixtures during the olefination steps, creating a nightmare for downstream purification teams who must resort to preparative silica gel column chromatography. This reliance on chromatography is not only labor-intensive and time-consuming but also introduces significant batch-to-batch variability and solvent waste, making it unsuitable for commercial scale-up of complex polymer additives or pharmaceutical intermediates alike.
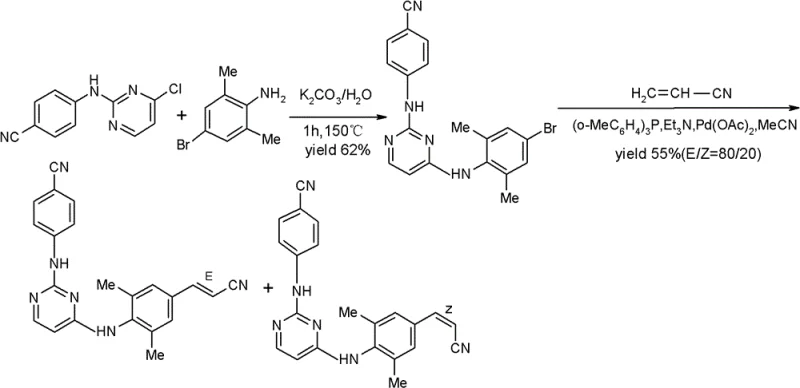
The economic implications of these legacy methods are profound, as the low overall yields and high purification costs directly erode profit margins for generic manufacturers. In many documented cases, the final step yields are suboptimal, often hovering around 55% to 62%, which necessitates larger reactor volumes and increased raw material consumption to meet production targets. Additionally, the harsh reaction conditions employed in some older routes can lead to the degradation of sensitive functional groups, further complicating the impurity profile. For supply chain heads, these factors translate into longer lead times and higher risks of supply disruption, as the process lacks the robustness required for continuous, high-volume manufacturing environments.
The Novel Approach
In stark contrast to the cumbersome legacy processes, the methodology outlined in CN103183642A offers a streamlined, high-efficiency alternative that prioritizes both yield and purity. The core innovation lies in the design of a specific halogenated intermediate, typically a bromo-substituted pyrimidine diamine, which serves as a versatile building block for the final coupling reactions. This new route eliminates the need for column chromatography entirely, relying instead on a straightforward acetonitrile recrystallization step to achieve HPLC purity levels greater than 99%. Such a simplification in downstream processing represents a paradigm shift in high-purity OLED material and pharmaceutical manufacturing, where purification costs often account for a substantial portion of the total production expense.
Moreover, the reaction conditions employed in this novel approach are notably mild, utilizing readily available and inexpensive raw materials that enhance the overall economic feasibility of the process. The patent data indicates that each synthetic step achieves a molar yield exceeding 85%, culminating in a total molar yield that significantly outperforms traditional methods. This improvement in efficiency not only reduces the environmental footprint by minimizing waste generation but also enhances the scalability of the process, making it ideal for reducing lead time for high-purity pharmaceutical intermediates. By adopting this route, manufacturers can secure a more resilient supply chain capable of meeting the rigorous demands of the global HIV treatment market.
Mechanistic Insights into Pyrimidine-Amine Coupling
The success of this synthetic strategy hinges on the precise control of the nucleophilic substitution reactions that construct the central scaffold of the molecule. The process begins with the protection of the amino group on the pyrimidine ring, typically using a benzyloxycarbonyl (Cbz) or tert-butoxycarbonyl (Boc) group, which prevents unwanted side reactions during the subsequent coupling steps. This protection strategy is crucial for maintaining the regioselectivity of the reaction, ensuring that the coupling occurs exclusively at the desired position on the pyrimidine ring. The use of potassium carbonate as a base in a dioxane solvent system facilitates the displacement of the halogen atom on the aniline derivative, forming the critical C-N bond with high fidelity.
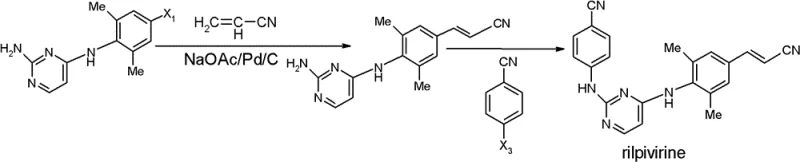
Following the coupling reaction, the removal of the protecting group is achieved under acidic conditions, such as treatment with hydrobromic acid, which cleanly reveals the free amine functionality required for the final assembly of Rilpivirine. The mechanistic elegance of this sequence lies in its ability to suppress the formation of common impurities, such as bis-coupled byproducts or hydrolysis products, which are often prevalent in less optimized routes. The resulting intermediate possesses a high degree of chemical homogeneity, which is essential for the subsequent palladium-catalyzed Heck coupling or amidation reactions. This level of control over the impurity profile is a key differentiator for R&D teams seeking to minimize validation burdens and accelerate time-to-market for new drug formulations.
Furthermore, the final conversion of the intermediate to Rilpivirine involves a dehydration or cyclization step using phosphorus oxychloride, which must be carefully managed to avoid over-chlorination or degradation. The patent specifies that this step can be conducted with high efficiency, yielding the final active pharmaceutical ingredient with minimal residual solvents or heavy metals. For quality assurance professionals, the robustness of this mechanism provides confidence in the consistency of the final product, ensuring that every batch meets the stringent specifications required for clinical use. The integration of these mechanistic insights into the manufacturing process underscores the technical sophistication of the proposed method.
How to Synthesize Rilpivirine Intermediate Efficiently
Implementing this advanced synthesis route requires a clear understanding of the operational parameters and safety considerations associated with each step. The process is designed to be compatible with standard glass-lined or stainless steel reactors commonly found in multipurpose pharmaceutical facilities, eliminating the need for specialized equipment. Operators must adhere to strict temperature controls during the reflux stages to maximize conversion rates while preventing thermal decomposition of the sensitive intermediates. The detailed standardized synthesis steps provided below outline the precise stoichiometry and workup procedures necessary to replicate the high yields reported in the patent literature.
- Protect the amino group of 4-chloro-2-amino-pyrimidine using benzyloxycarbonyl chloride under reflux conditions.
- Couple the protected pyrimidine with 4-bromo-2,6-dimethyl-aniline in dioxane with potassium carbonate.
- Remove the protecting group using hydrobromic acid to yield the final diamine intermediate with over 98% purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis route offers transformative benefits for procurement managers and supply chain leaders tasked with optimizing cost structures and ensuring supply continuity. The elimination of silica gel chromatography alone represents a significant reduction in operational expenditures, as it removes the need for large quantities of adsorbents and organic solvents, thereby lowering waste disposal costs. Additionally, the use of cheap and easily obtainable raw materials mitigates the risk of price volatility associated with exotic reagents, providing a more stable cost baseline for long-term production planning. These factors collectively contribute to a leaner, more agile manufacturing operation capable of responding swiftly to market demands.
- Cost Reduction in Manufacturing: The streamlined process significantly lowers the cost of goods sold by improving overall yield and reducing purification complexity. By avoiding expensive transition metal catalysts in certain steps and utilizing simple recrystallization for purification, the method minimizes material loss and solvent consumption. This efficiency translates directly into improved profit margins, allowing companies to offer competitive pricing without compromising on quality standards.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals and standard reaction conditions enhances the robustness of the supply chain, reducing the likelihood of disruptions caused by raw material shortages. The high yield and purity of the intermediate ensure consistent output, enabling manufacturers to maintain reliable inventory levels and meet delivery commitments to downstream API producers. This stability is crucial for maintaining trust with global pharmaceutical partners.
- Scalability and Environmental Compliance: The process is inherently scalable, having been designed with industrial production in mind from the outset. The reduction in solvent usage and waste generation aligns with modern green chemistry principles, facilitating easier compliance with increasingly stringent environmental regulations. This sustainability aspect not only reduces regulatory risk but also enhances the corporate social responsibility profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a factual basis for decision-making. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this route into their existing production portfolios.
Q: What is the primary advantage of this new intermediate synthesis route?
A: The primary advantage is the elimination of column chromatography. The process relies on simple acetonitrile recrystallization to achieve HPLC purity exceeding 99%, significantly reducing processing time and solvent waste compared to prior art.
Q: How does this method improve cost efficiency for API manufacturers?
A: By utilizing cheap and easily available raw materials and achieving a total molar yield of over 85%, the method drastically lowers the cost of goods sold (COGS). The mild reaction conditions also reduce energy consumption and equipment wear.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the patent explicitly states the method meets the requirements for large-scale industrial production. The avoidance of difficult separations and the use of standard reagents make it highly scalable for commercial metric-ton manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rilpivirine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes in the competitive landscape of antiretroviral drug manufacturing. Our team of expert chemists has extensively evaluated the pathway described in CN103183642A and confirmed its viability for large-scale production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of intermediate we deliver meets the highest industry standards.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs through the adoption of this superior synthesis technology. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes and requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can drive value for your organization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
