Advanced Solid-Phase Synthesis of Buserelin for Scalable Pharmaceutical Manufacturing
The pharmaceutical landscape for Luteinizing Hormone-Releasing Hormone (LHRH) analogues has seen a paradigm shift towards more efficient manufacturing protocols, as evidenced by the innovative techniques disclosed in patent CN101935339A. This pivotal intellectual property outlines a robust solid-phase preparation method for Buserelin, a critical peptide therapeutic used in treating hormone-dependent conditions such as prostate cancer and endometriosis. Traditional liquid-phase syntheses have long been plagued by operational complexities and low industrial viability, but this new methodology leverages specific resin technologies to overcome these historical bottlenecks. By utilizing an HMPB-MBHA resin with a precisely controlled degree of substitution, the process achieves a remarkable balance between reaction kinetics and final product integrity. For global procurement teams and R&D directors, this represents a significant opportunity to secure a reliable peptide intermediate supplier capable of delivering high-purity materials with reduced environmental footprints. The technical breakthrough lies not just in the sequence of amino acid addition, but in the fundamental optimization of the solid support itself, ensuring that the delicate peptide chain remains intact throughout the rigorous synthesis cycle.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of complex polypeptides like Buserelin relied heavily on liquid-phase synthesis strategies, which introduced substantial inefficiencies into the supply chain. These conventional methods often necessitated multiple isolation and purification steps after every single amino acid coupling, leading to exponential losses in overall yield and drastically increasing the cost of goods sold. Furthermore, the harsh chemical conditions frequently required for deprotection in liquid phases could compromise the stereochemical integrity of sensitive residues, resulting in difficult-to-remove impurities that jeopardize regulatory compliance. The accumulation of solvent waste and the need for extensive downstream processing made these legacy routes environmentally unsustainable and economically unviable for large-scale cost reduction in API manufacturing. Consequently, manufacturers faced significant challenges in scaling up production to meet the growing global demand for LHRH analogues without incurring prohibitive operational expenses.
The Novel Approach
In stark contrast, the novel solid-phase synthesis route described in the patent introduces a streamlined workflow that eliminates the need for intermediate purifications, thereby enhancing overall process efficiency. The core of this innovation is the strategic use of HMPB-MBHA resin, which allows for the cleavage of the fully protected peptide under mild acidic conditions, preserving the structural fidelity of the molecule. This approach facilitates a continuous assembly line where amino acids are coupled sequentially on the solid support, significantly reducing the handling time and solvent consumption associated with traditional methods. The implementation of advanced coupling reagents ensures that each step proceeds to near completion, minimizing the formation of deletion sequences that complicate final purification. As illustrated in the comprehensive reaction scheme below, this method transforms a fragmented multi-step process into a cohesive, scalable operation suitable for modern GMP facilities.
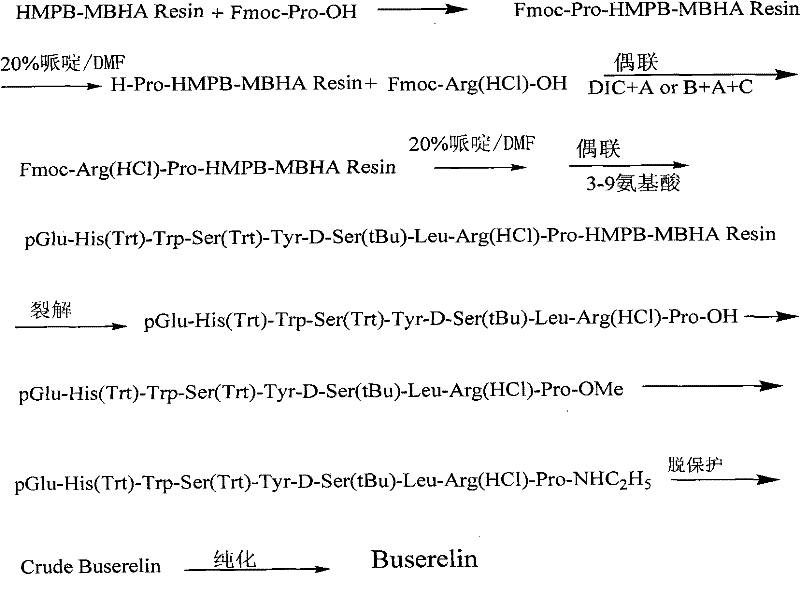
Mechanistic Insights into HMPB-MBHA Resin Loading and Coupling
The success of this solid-phase strategy hinges on the precise control of the resin's substitution value, which acts as the foundation for the entire peptide assembly. Experimental data within the patent reveals that maintaining the HMPB-MBHA resin substitution between 0.2 mmol/g and 0.9 mmol/g is critical, with an optimal value of 0.60 mmol/g identified for maximizing both yield and purity. If the loading is too high, steric hindrance prevents complete coupling of subsequent amino acids, while loading that is too low results in inefficient use of the solid support and increased production costs. The initial anchoring of Fmoc-Pro-OH to the resin is catalyzed by highly efficient reagents such as HATU and HOAt, which activate the carboxyl group rapidly while suppressing racemization. This specific activation mechanism is crucial for the first residue, as any stereochemical error at the C-terminus would propagate through the entire synthesis, rendering the final batch useless.
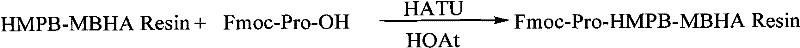
Following the initial loading, the sequential coupling of the remaining protected amino acids employs a versatile activation system involving DIC combined with additives like HOBt or HOAt. The inclusion of organic bases such as DIPEA or TMP plays a dual role: they neutralize the acid generated during coupling and actively prevent the racemization of activated amino acid species. This careful selection of reagents ensures that the peptide chain elongates with high fidelity, maintaining the specific sequence required for biological activity. The use of Fmoc protection chemistry allows for mild deprotection steps using piperidine, which is compatible with the acid-labile linkers on the resin. This orthogonality between the temporary N-terminal protection and the permanent resin linker is what enables the final cleavage step to release the fully protected peptide without damaging side-chain protecting groups, a feat that is often difficult to achieve in less optimized systems.
How to Synthesize Buserelin Efficiently
Implementing this synthesis route requires strict adherence to the optimized parameters defined in the patent to ensure consistent quality and yield. The process begins with the meticulous preparation of the loaded resin, followed by the iterative cycle of deprotection and coupling that builds the decapeptide chain. Operators must utilize real-time monitoring techniques, such as the Kaiser test, to verify the completion of each coupling step before proceeding, thereby preventing the accumulation of incomplete sequences. While the general workflow is straightforward, the specific choice of solvents, reaction times, and reagent ratios dictates the success of the commercial scale-up of complex peptide intermediates. For detailed operational instructions, please refer to the standardized guide below which outlines the critical control points for each stage of the synthesis.
- Prepare Fmoc-Pro-HMPB-MBHA resin using HATU and HOAt to achieve a substitution value of approximately 0.60 mmol/g.
- Sequentially couple protected amino acids using DIC-based activation systems while monitoring with Kaiser tests.
- Cleave the peptide from the resin using TFA/DCM, followed by ethylamination, deprotection, and HPLC purification.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement managers, the transition to this solid-phase methodology offers tangible benefits that extend far beyond simple technical metrics. By eliminating the numerous intermediate purification steps inherent in liquid-phase synthesis, the process drastically reduces the consumption of solvents and chromatography media, leading to a leaner and more cost-effective manufacturing profile. The ability to produce the fully protected peptide with high purity directly from the cleavage step means that downstream processing is simplified, allowing for faster turnaround times and improved responsiveness to market demands. This efficiency translates directly into a more resilient supply chain, where production bottlenecks are minimized, and the risk of batch failure due to cumulative yield losses is significantly mitigated. Furthermore, the mild reaction conditions reduce the burden on waste treatment facilities, aligning production practices with increasingly stringent environmental regulations.
- Cost Reduction in Manufacturing: The elimination of intermediate isolation and purification steps fundamentally alters the cost structure of Buserelin production. By avoiding the loss of material associated with multiple work-ups and the high cost of preparative HPLC at every stage, the overall cost of goods is substantially lowered. Additionally, the high efficiency of the coupling reagents ensures that expensive protected amino acids are utilized effectively, minimizing raw material waste. This economic advantage allows manufacturers to offer more competitive pricing structures while maintaining healthy margins, making it an attractive option for generic drug developers seeking cost reduction in API manufacturing.
- Enhanced Supply Chain Reliability: The robustness of the solid-phase protocol ensures consistent batch-to-batch quality, which is paramount for maintaining uninterrupted supply to pharmaceutical clients. The use of commercially available Fmoc-amino acids and standard coupling reagents reduces dependency on exotic or hard-to-source catalysts, thereby stabilizing the raw material supply chain. Moreover, the scalability of the resin-based process means that production volumes can be ramped up quickly to meet surges in demand without requiring significant capital investment in new equipment. This flexibility is crucial for reducing lead time for high-purity peptide intermediates in a volatile global market.
- Scalability and Environmental Compliance: The process is inherently designed for industrial scale-up, with reaction conditions that are easily managed in large-scale reactors. The reduction in solvent usage and the avoidance of heavy metal catalysts simplify waste management and disposal, ensuring compliance with green chemistry principles. This environmental stewardship not only reduces regulatory risks but also enhances the corporate sustainability profile of the manufacturer. The ability to handle large quantities of resin safely and efficiently demonstrates a mature process capability that is ready for immediate commercial scale-up of complex peptide intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the solid-phase synthesis of Buserelin, derived directly from the patented methodology. Understanding these nuances is essential for partners looking to integrate this technology into their existing manufacturing portfolios. The answers provided reflect the specific advantages of the HMPB-MBHA resin system and the optimized coupling protocols that define this process. We encourage potential collaborators to review these details to fully appreciate the value proposition of this advanced synthetic route.
Q: What is the optimal substitution value for the HMPB-MBHA resin in this synthesis?
A: The patent specifies an optimal substitution value range between 0.2 mmol/g and 0.9 mmol/g, with experimental data indicating that 0.60 mmol/g yields the highest purity and product recovery.
Q: How does this solid-phase method compare to traditional liquid-phase synthesis?
A: Unlike liquid-phase synthesis which requires complex intermediate purifications, this solid-phase approach utilizes mild acid cleavage conditions that preserve the fully protected peptide structure, significantly simplifying the workflow and reducing environmental pollution.
Q: Which coupling agents are recommended to prevent racemization?
A: The process recommends using organic bases such as DIPEA or TMP in conjunction with coupling systems like DIC/HOBt or HATU/HOAt to ensure rapid reaction kinetics while minimizing the risk of amino acid racemization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Buserelin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthesis technologies to maintain a competitive edge in the pharmaceutical sector. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practical manufacturing environments. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of Buserelin intermediate adheres to the highest international standards. Our infrastructure is specifically designed to handle the complexities of solid-phase peptide synthesis, providing a seamless bridge between laboratory innovation and industrial reality.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can enhance your supply chain efficiency. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the specific economic benefits tailored to your production volume. We are prepared to provide specific COA data and route feasibility assessments to demonstrate our capability as a trusted partner. Let us collaborate to drive down costs and accelerate the availability of this vital therapeutic agent to patients worldwide.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
