Scalable Production of Tunable Cucurbit[n]urils for Advanced Supramolecular Applications
The chemical landscape of supramolecular host molecules has been significantly advanced by the innovations detailed in patent CN100339380C, which introduces a robust methodology for the preparation of Cucurbit[n]urils with tunable ring sizes ranging from n equals 4 to 12. This technological breakthrough addresses a long-standing limitation in the field where traditional synthesis methods, such as the Behrend reaction, were largely restricted to producing the n equals 6 homolog with minimal control over oligomer distribution. By leveraging a sophisticated template-directed synthesis approach, this patent enables the precise manipulation of macrocyclic architecture through the strategic addition of ionic or organic template compounds during the acid-catalyzed condensation of glycoluril and formaldehyde. For R&D Directors and Procurement Managers seeking reliable cucurbituril supplier partnerships, this process represents a paradigm shift towards customizable host molecules that can be tailored for specific guest encapsulation applications in pharmaceuticals and advanced materials. The ability to access a broader spectrum of cucurbituril homologs opens new avenues for drug delivery systems, catalysis, and environmental remediation technologies that require specific cavity dimensions for optimal performance. Furthermore, the process operates under relatively mild thermal conditions compared to prior art, reducing energy consumption and enhancing the safety profile of the manufacturing workflow. This report analyzes the technical merits and commercial implications of adopting this template-controlled synthesis for the commercial scale-up of complex macrocycles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cucurbiturils has been plagued by a lack of selectivity, often resulting in complex mixtures where the Cucurbit[6]uril homolog dominates the product distribution regardless of the desired application specifications. Traditional protocols typically require harsh reaction conditions, including prolonged heating at high temperatures and the use of concentrated mineral acids without effective means to steer the oligomerization pathway towards specific ring sizes. This lack of control necessitates extensive and costly downstream purification processes, such as repeated recrystallization or chromatographic separation, to isolate minor homologs like Cucurbit[5]uril or Cucurbit[7]uril which are often needed for specific host-guest chemistry applications. The inefficiency of these conventional methods leads to significant material waste and extended production lead times, creating bottlenecks for supply chain heads who require consistent availability of high-purity host molecules. Moreover, the inability to tune the cavity size limits the functional utility of the final product, forcing formulators to adapt their designs to the available chemistry rather than optimizing the chemistry for their design. These structural and process limitations have historically hindered the widespread adoption of cucurbiturils in cost-sensitive industrial applications where consistency and scalability are paramount.
The Novel Approach
The novel approach described in the patent data overcomes these historical constraints by introducing a template effect that actively directs the formation of methylene bridges between glycoluril units to favor specific oligomer sizes. By incorporating specific salts, such as alkali metal halides, or organic compounds into the reaction mixture, the synthesis can be tuned to produce Cucurbit[n]urils where n varies from 4 to 12 with significantly improved selectivity. ![General structure of Cucurbit[n]uril showing variable ring size n](/insights/img/cucurbituril-synthesis-template-supplier-20260315040543-02.png) This method allows for the stabilization of transition states that correspond to specific ring dimensions, effectively lowering the activation energy for the formation of non-standard homologs like n equals 5 or n equals 7. The process utilizes readily available starting materials including glycoluril and formaldehyde precursors, ensuring that cost reduction in supramolecular chemistry manufacturing is achievable without compromising on molecular complexity. Operating temperatures are optimized to range between 60 and 100 degrees Celsius, which is milder than the extreme conditions often required in older methodologies, thereby enhancing process safety and equipment longevity. This flexibility enables manufacturers to respond dynamically to market demands for different cucurbituril species without retooling entire production lines, offering a strategic advantage in reducing lead time for high-purity host molecules.
This method allows for the stabilization of transition states that correspond to specific ring dimensions, effectively lowering the activation energy for the formation of non-standard homologs like n equals 5 or n equals 7. The process utilizes readily available starting materials including glycoluril and formaldehyde precursors, ensuring that cost reduction in supramolecular chemistry manufacturing is achievable without compromising on molecular complexity. Operating temperatures are optimized to range between 60 and 100 degrees Celsius, which is milder than the extreme conditions often required in older methodologies, thereby enhancing process safety and equipment longevity. This flexibility enables manufacturers to respond dynamically to market demands for different cucurbituril species without retooling entire production lines, offering a strategic advantage in reducing lead time for high-purity host molecules.
Mechanistic Insights into Template-Directed Cyclization
The core mechanism driving this synthesis involves the acid-catalyzed condensation of glycoluril units, where the carbonyl groups of the glycoluril react with formaldehyde to form methylene bridges that link the monomers into a cyclic oligomer. 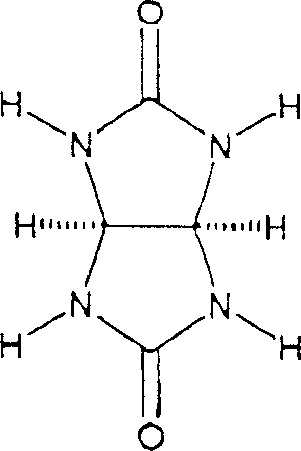 Crucially, the presence of template compounds introduces an ionic templating effect where cations or anions interact with the carbonyl portals of the forming macrocycle, stabilizing specific intermediate geometries through ion-dipole interactions or hydrogen bonding. For instance, smaller cations like lithium may favor smaller ring sizes due to steric fit within the cavity, while larger cations like cesium stabilize larger homologs, allowing for precise control over the final product distribution. This interaction is not merely passive; it actively shifts the thermodynamic equilibrium of the reversible reaction steps towards the desired oligomer, minimizing the formation of unwanted byproducts and polymeric tars. Understanding this mechanistic nuance is vital for R&D teams aiming to replicate the process, as the choice of acid anion and template cation must be matched to the target ring size to achieve optimal yields. The patent data suggests that the proton from the acid also acts as a template, competing with added cations, which adds a layer of complexity that can be leveraged to fine-tune the reaction outcome. This deep mechanistic control ensures that the resulting cucurbiturils possess the structural integrity required for demanding applications in drug stabilization and molecular recognition.
Crucially, the presence of template compounds introduces an ionic templating effect where cations or anions interact with the carbonyl portals of the forming macrocycle, stabilizing specific intermediate geometries through ion-dipole interactions or hydrogen bonding. For instance, smaller cations like lithium may favor smaller ring sizes due to steric fit within the cavity, while larger cations like cesium stabilize larger homologs, allowing for precise control over the final product distribution. This interaction is not merely passive; it actively shifts the thermodynamic equilibrium of the reversible reaction steps towards the desired oligomer, minimizing the formation of unwanted byproducts and polymeric tars. Understanding this mechanistic nuance is vital for R&D teams aiming to replicate the process, as the choice of acid anion and template cation must be matched to the target ring size to achieve optimal yields. The patent data suggests that the proton from the acid also acts as a template, competing with added cations, which adds a layer of complexity that can be leveraged to fine-tune the reaction outcome. This deep mechanistic control ensures that the resulting cucurbiturils possess the structural integrity required for demanding applications in drug stabilization and molecular recognition.
Impurity control is inherently built into this mechanism through the selective precipitation and solubility differences of the various cucurbituril-template complexes formed during the reaction. Different homologs exhibit distinct solubility profiles in the presence of specific salt concentrations, allowing for the separation of Cucurbit[6]uril from Cucurbit[5]uril or Cucurbit[7]uril through controlled crystallization or selective dissolution steps. 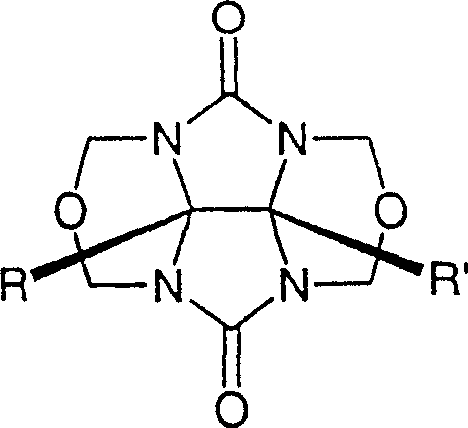 The patent highlights that certain template compounds can induce the precipitation of specific complexes, effectively pulling the equilibrium towards the desired product and simplifying the purification workflow. This reduces the reliance on resource-intensive chromatographic techniques, which are often difficult to scale for industrial production of bulk chemicals. By manipulating the ionic strength and composition of the reaction medium, manufacturers can achieve high-purity specifications without excessive solvent usage or waste generation. This aspect of the process is particularly attractive for supply chain heads focused on environmental compliance and operational efficiency, as it streamlines the path from crude reaction mixture to finished high-purity cucurbituril. The ability to predict and control impurity profiles based on template selection ensures consistent quality across batches, a critical factor for regulatory compliance in pharmaceutical intermediate manufacturing.
The patent highlights that certain template compounds can induce the precipitation of specific complexes, effectively pulling the equilibrium towards the desired product and simplifying the purification workflow. This reduces the reliance on resource-intensive chromatographic techniques, which are often difficult to scale for industrial production of bulk chemicals. By manipulating the ionic strength and composition of the reaction medium, manufacturers can achieve high-purity specifications without excessive solvent usage or waste generation. This aspect of the process is particularly attractive for supply chain heads focused on environmental compliance and operational efficiency, as it streamlines the path from crude reaction mixture to finished high-purity cucurbituril. The ability to predict and control impurity profiles based on template selection ensures consistent quality across batches, a critical factor for regulatory compliance in pharmaceutical intermediate manufacturing.
How to Synthesize Cucurbit[n]urils Efficiently
The synthesis protocol outlined in the patent provides a clear pathway for producing tunable cucurbiturils by combining glycoluril derivatives with formaldehyde sources in the presence of strong acids and specific templating agents. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during scale-up operations.
- Mix substituted or unsubstituted glycolurils with a strong acid and a methylene bridge source like formaldehyde.
- Introduce specific template compounds such as alkali metal salts to control the ring size distribution.
- Heat the mixture to 60-100°C and isolate the product via selective precipitation or chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this template-directed synthesis methodology offers substantial commercial advantages by fundamentally altering the cost structure and reliability of cucurbituril supply chains. The use of commodity chemicals such as glycoluril, formaldehyde, and common inorganic salts as raw materials ensures that cost reduction in supramolecular chemistry manufacturing is driven by process efficiency rather than expensive proprietary reagents. Cost Reduction in Manufacturing: The elimination of complex multi-step sequences required in older methods significantly lowers operational expenditures, as the reaction proceeds directly to the macrocyclic product with high atom economy. By avoiding the need for exotic catalysts or extreme high-pressure equipment, capital investment requirements are minimized, allowing for faster ROI on production assets. The milder reaction temperatures also translate to lower energy consumption per kilogram of product, contributing to a more sustainable and cost-effective manufacturing footprint. Furthermore, the improved selectivity reduces the volume of waste solvents and byproducts that require disposal, aligning with modern green chemistry principles and reducing environmental compliance costs. These factors combine to create a robust economic model that supports competitive pricing for high-value supramolecular hosts.
- Cost Reduction in Manufacturing: The process leverages the template effect to minimize the formation of unwanted oligomers, thereby reducing the load on downstream purification units and increasing the overall yield of the target species. This efficiency gain means that less raw material is wasted on off-spec products, directly improving the gross margin for each production batch. Additionally, the ability to use standard stainless steel reactors instead of specialized lined vessels further decreases maintenance and replacement costs over the lifecycle of the plant. The simplified workflow allows for shorter batch cycles, increasing the throughput capacity of existing facilities without the need for physical expansion. All these elements contribute to a significant optimization of the cost of goods sold, making advanced macrocycles accessible for broader industrial applications.
- Enhanced Supply Chain Reliability: Sourcing reliability is greatly improved because the key raw materials are globally available commodity chemicals with stable supply lines, mitigating the risk of production stoppages due to material shortages. The flexibility to produce different ring sizes using the same base equipment allows manufacturers to pivot quickly in response to shifting customer demand without long lead times for retooling. This agility ensures that reducing lead time for high-purity host molecules becomes a tangible reality, enabling just-in-time delivery models for critical pharmaceutical or electronic chemical clients. The robustness of the reaction conditions also means that production is less susceptible to minor variations in utility supply, such as steam pressure or cooling water temperature, ensuring consistent output. Consequently, supply chain heads can forecast inventory levels with greater confidence, reducing the need for excessive safety stock and freeing up working capital.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by the patent examples which range from gram-scale laboratory experiments to multi-kilogram preparations without loss of control over product distribution. The use of aqueous acid systems and the potential for recycling template salts minimizes the generation of hazardous organic waste, simplifying wastewater treatment and regulatory reporting. Scalability is further supported by the fact that the reaction does not rely on sensitive air- or moisture-free conditions, allowing for standard industrial handling procedures that reduce operational complexity. This ease of scale-up facilitates the commercial scale-up of complex macrocycles from pilot plant to full industrial production, meeting the growing demand for functional host molecules. Environmental compliance is enhanced by the reduced solvent intensity and the potential for closed-loop systems, positioning the manufacturer as a responsible partner in sustainable chemistry initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis technology in an industrial setting.
Q: How does the template effect control cucurbituril ring size?
A: The template effect utilizes specific cations or anions that fit within the forming macrocyclic cavity, stabilizing intermediates of specific sizes (n=4 to 12) during the acid-catalyzed condensation of glycoluril and formaldehyde.
Q: What are the advantages of this synthesis over traditional methods?
A: Unlike traditional methods that predominantly yield Cucurbit[6]uril, this patented process allows for the selective production of various ring sizes (n=5, 7, 8, etc.) under milder temperature conditions, improving process flexibility.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process uses readily available raw materials like glycoluril and formaldehyde and operates at moderate temperatures, making it highly scalable for industrial production of high-purity supramolecular hosts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cucurbituril Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating this advanced patent technology into commercial reality, offering extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team possesses the expertise to navigate the nuances of template-directed synthesis, ensuring stringent purity specifications are met for every batch of cucurbituril delivered. We operate rigorous QC labs equipped with advanced analytical instrumentation to verify ring size distribution and impurity profiles, guaranteeing that our high-purity cucurbit[n]urils meet the exacting standards of the global pharmaceutical and specialty chemical industries. Our commitment to quality is matched by our dedication to process safety and environmental stewardship, making us a preferred partner for companies seeking sustainable supply chain solutions. By leveraging our deep understanding of the underlying chemistry, we can customize production parameters to optimize yield and cost for specific customer requirements.
We invite you to engage with our technical procurement team to discuss how our capabilities can support your specific project needs and drive value through our Customized Cost-Saving Analysis. We encourage potential partners to request specific COA data and route feasibility assessments to validate the suitability of our materials for your applications. Our goal is to establish long-term collaborations that foster innovation and efficiency in the development of next-generation supramolecular systems. Contact us today to explore how our scalable manufacturing solutions can accelerate your product development timelines.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
