Scalable Synthesis of High-Efficiency Antibacterial Triazole Intermediates for Pharmaceutical Applications
The pharmaceutical and agrochemical industries are constantly seeking novel scaffolds to combat rising antibiotic resistance and improve therapeutic efficacy. Patent CN102206190A introduces a significant advancement in this domain by disclosing a series of high-efficiency antibacterial triazole compounds. These compounds, characterized by a 1-substituted phenyl-4-substituted phenyl-5-substituted salicylaldimine-1,2,3-triazole core, represent a strategic evolution in antimicrobial drug design. The patent details a robust synthetic methodology that leverages diazotization, azidation, and cyclization reactions to construct this complex heterocyclic system with remarkable precision. For R&D Directors and Procurement Managers, this technology offers a viable pathway to develop next-generation antifungal and antibacterial agents that overcome the limitations of existing treatments like fluconazole. The structural novelty of these intermediates provides a strong foundation for intellectual property expansion and the development of differentiated products in a crowded market. Furthermore, the synthesis route is designed with scalability in mind, utilizing common industrial reagents that facilitate a smoother transition from laboratory bench to commercial manufacturing plants. This report analyzes the technical merits and commercial implications of this patented technology, highlighting its potential to serve as a reliable pharmaceutical intermediate supplier solution for global supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for synthesizing triazole-based antimicrobial agents often rely on multi-step sequences that involve expensive transition metal catalysts or harsh reaction conditions. These conventional pathways frequently suffer from low atom economy, generating significant amounts of hazardous waste that complicate disposal and increase environmental compliance costs. Moreover, the use of heavy metal catalysts necessitates rigorous purification steps to ensure that residual metal levels meet stringent regulatory standards for pharmaceutical ingredients, which can drastically reduce overall process yield and extend production timelines. In many cases, the starting materials required for these older methods are not readily available or are subject to volatile pricing, creating supply chain vulnerabilities for manufacturers. The structural rigidity of some conventional triazole derivatives also limits their ability to interact effectively with resistant microbial strains, leading to diminished therapeutic outcomes in clinical settings. Consequently, there is a pressing need for synthetic routes that are not only chemically efficient but also economically sustainable and environmentally benign. The reliance on complex purification protocols in traditional methods often becomes a bottleneck, hindering the ability to achieve cost reduction in pharmaceutical intermediates manufacturing at a commercial scale.
The Novel Approach
The methodology outlined in CN102206190A presents a transformative alternative by employing a streamlined four-step synthesis that begins with readily available functionalized anilines. This approach eliminates the need for precious metal catalysts, instead utilizing common bases and acids to drive the reaction sequence, which significantly simplifies the downstream processing requirements. The key innovation lies in the strategic combination of o-hydroxyphenyl and 1,2,3-triazole molecular fragments, which synergistically enhance the biological activity of the final compound against a broad spectrum of pathogens. By optimizing reaction conditions such as temperature and pH during the condensation phase, the process achieves high conversion rates while minimizing the formation of unwanted by-products. This efficiency translates directly into operational advantages, as fewer purification steps are required to isolate the target molecule with high purity. The use of standard solvents like ethanol and toluene further enhances the practicality of this method, allowing for easier solvent recovery and recycling. For supply chain leaders, this novel approach represents a more resilient manufacturing strategy that reduces dependency on scarce reagents and mitigates the risks associated with complex synthetic routes. The ability to produce high-purity antibacterial triazole intermediates with reduced environmental impact aligns perfectly with modern green chemistry principles and corporate sustainability goals.
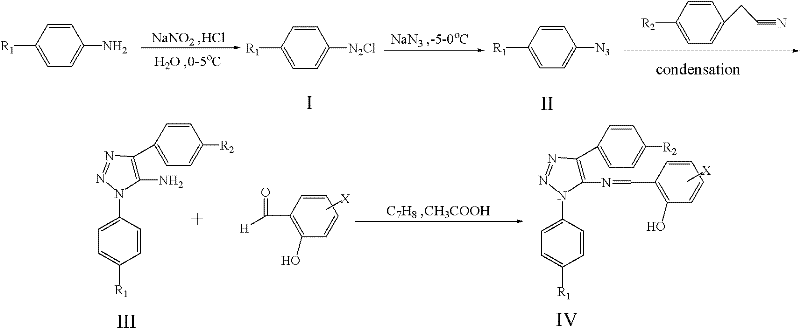
Mechanistic Insights into Diazotization and Cyclization Reactions
The core of this synthetic strategy involves a carefully orchestrated sequence of chemical transformations that build the triazole ring system with high fidelity. The process initiates with the diazotization of substituted anilines, where the amino group is converted into a diazonium salt using sodium nitrite under acidic conditions at low temperatures (0-5°C). This step is critical for controlling the reactivity of the intermediate and preventing premature decomposition. Subsequently, the diazonium salt undergoes azidation upon treatment with sodium azide, forming a substituted phenyl azide which serves as the 1,3-dipole in the subsequent cycloaddition reaction. The cyclization step involves the reaction of this azide with a substituted phenylacetonitrile in the presence of a base catalyst, leading to the formation of the 1,2,3-triazole core through a [3+2] cycloaddition mechanism. This ring-closing reaction is highly regioselective, ensuring that the desired isomer is produced predominantly, which simplifies the purification process and improves the overall yield of the intermediate. The final condensation with substituted salicylaldehyde introduces the Schiff base moiety, which is essential for the compound's antimicrobial activity. This step is typically carried out under reflux conditions in a solvent like toluene, with acid catalysis to facilitate the dehydration process. The mechanistic clarity of this route allows for precise control over reaction parameters, enabling manufacturers to optimize the process for maximum efficiency and consistency.
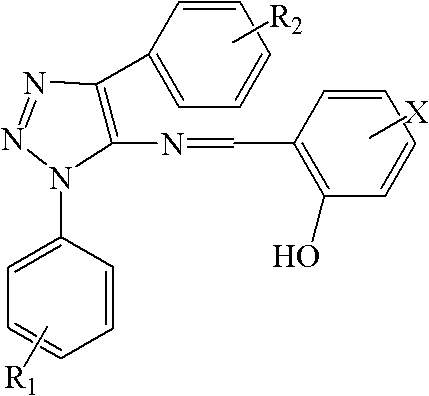
Impurity control is a paramount concern in the synthesis of pharmaceutical intermediates, and this patented route offers inherent advantages in this regard. The use of stoichiometric reagents and the avoidance of transition metals minimize the introduction of inorganic impurities that are difficult to remove. Furthermore, the reaction conditions are mild enough to prevent the degradation of sensitive functional groups on the aromatic rings, preserving the structural integrity of the molecule throughout the synthesis. The crystallization steps described in the patent, utilizing solvents such as ethanol, are highly effective in removing organic impurities and isomers, resulting in a final product with a sharp melting point and high chemical purity. The ability to tune the substituents on the phenyl rings (R1, R2, X) allows for the optimization of physicochemical properties, such as solubility and stability, without compromising the synthetic efficiency. This flexibility is crucial for R&D teams aiming to develop a portfolio of related compounds with tailored biological profiles. By understanding the mechanistic nuances of each step, process chemists can identify potential failure points and implement robust control strategies to ensure consistent quality across different production batches. This level of control is essential for meeting the rigorous quality standards required by regulatory agencies and end-users in the pharmaceutical industry.
How to Synthesize 1-Phenyl-4-Phenyl-5-Salicylaldimine-1,2,3-Triazole Efficiently
The synthesis of this specific triazole derivative serves as a representative example of the broader platform technology described in the patent. The process begins with the preparation of the diazonium salt from aniline, followed by conversion to phenyl azide, which is then reacted with phenylacetonitrile to form the triazole amine intermediate. This intermediate is subsequently condensed with salicylaldehyde to yield the final target compound. The detailed standardized synthesis steps involve precise control of temperature, stoichiometry, and reaction time to ensure optimal yields and purity. For technical teams looking to implement this route, it is essential to adhere to the specified reaction conditions, particularly during the diazotization and azidation steps where safety and stability are critical. The use of TLC monitoring is recommended to track the progress of the reaction and determine the optimal endpoint for each step.
- Perform diazotization of functionalized aniline using sodium nitrite and acid at 0-5°C to form diazonium salt.
- Conduct azidation by reacting the diazonium salt with sodium azide at -10°C to 0°C to yield substituted phenyl azide.
- Execute ring closure reaction with phenylacetonitrile and catalyst at 20-78°C to form the triazole core.
- Complete condensation with substituted salicylaldehyde under reflux to finalize the target antibacterial compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers substantial benefits for procurement and supply chain operations. The elimination of expensive transition metal catalysts removes a significant cost driver from the manufacturing process, leading to direct savings in raw material expenses. Additionally, the simplified purification workflow reduces the consumption of solvents and energy, further contributing to cost reduction in pharmaceutical intermediates manufacturing. The use of commodity chemicals as starting materials ensures a stable and reliable supply chain, minimizing the risk of production delays due to raw material shortages. This reliability is crucial for maintaining continuous production schedules and meeting delivery commitments to downstream customers. The high yields reported in the patent examples indicate a material-efficient process that maximizes the output from each batch, enhancing overall production capacity without the need for significant capital investment in new equipment. Furthermore, the environmental benefits of this green chemistry approach can lead to lower waste disposal costs and improved regulatory compliance, which are increasingly important factors in supplier selection criteria. For supply chain heads, this technology represents a strategic opportunity to enhance supply chain reliability and reduce lead time for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The process avoids the use of precious metal catalysts, which are often subject to price volatility and require complex removal steps. By utilizing common bases and acids, the manufacturing cost structure is significantly optimized, allowing for more competitive pricing in the market. The high atom economy of the reaction sequence ensures that a larger proportion of the raw materials are converted into the final product, reducing waste and maximizing resource utilization. This efficiency translates into lower variable costs per unit, providing a strong margin advantage for manufacturers. The simplified downstream processing also reduces labor and utility costs associated with purification, further enhancing the economic viability of the process. Overall, the streamlined nature of this synthesis supports substantial cost savings without compromising on product quality or performance.
- Enhanced Supply Chain Reliability: The reliance on readily available aniline derivatives and standard solvents ensures that the supply chain is robust and resilient to disruptions. Unlike processes that depend on specialized or scarce reagents, this route can be easily sourced from multiple suppliers, reducing the risk of single-source dependency. The mild reaction conditions also mean that the process can be implemented in a wide range of manufacturing facilities, increasing the flexibility of production planning. This flexibility allows for better responsiveness to changes in market demand, ensuring that supply can be scaled up or down as needed. The consistent quality of the raw materials and the robustness of the reaction contribute to a stable production output, which is essential for maintaining long-term partnerships with key customers. This reliability is a key differentiator in a competitive market where supply continuity is often as important as price.
- Scalability and Environmental Compliance: The synthetic route is designed with scalability in mind, using unit operations that are standard in the fine chemical industry. This makes the commercial scale-up of complex pharmaceutical intermediates straightforward and low-risk. The absence of hazardous reagents and the generation of minimal waste align with strict environmental regulations, reducing the compliance burden on manufacturers. The use of recyclable solvents and the potential for solvent recovery further enhance the sustainability profile of the process. This environmental friendliness is increasingly valued by customers and regulators, potentially opening up new market opportunities where green credentials are a prerequisite. The ability to produce large quantities of the intermediate with a low environmental footprint positions this technology as a preferred choice for sustainable manufacturing initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this triazole synthesis technology. These answers are derived from the detailed data and examples provided in the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these aspects is crucial for evaluating the feasibility of integrating this route into existing production portfolios. The information covers key areas such as process safety, quality control, and regulatory considerations, providing a comprehensive overview for stakeholders.
Q: What are the key advantages of this triazole synthesis route over conventional methods?
A: The patented route utilizes readily available aniline derivatives and avoids complex transition metal catalysts, significantly simplifying purification and reducing environmental impact while maintaining high yields.
Q: How does this compound address antibiotic resistance issues?
A: The novel structure combines o-hydroxyphenyl and 1,2,3-triazole fragments, demonstrating strong broad-spectrum activity against Candida albicans and Escherichia coli, even where traditional fluconazole shows resistance.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the synthesis uses standard industrial reagents and mild conditions (0-150°C), making it highly scalable for commercial scale-up of complex pharmaceutical intermediates without requiring specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triazole Compound Supplier
The technical potential of this antibacterial triazole synthesis route is immense, offering a pathway to high-value intermediates with broad applications in drug development. NINGBO INNO PHARMCHEM, as a seasoned CDMO expert, possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this technology to fruition. Our facilities are equipped with stringent purity specifications and rigorous QC labs to ensure that every batch meets the highest industry standards. We understand the critical nature of supply chain continuity and are committed to providing a reliable pharmaceutical intermediate supplier partnership that supports your long-term growth. Our team of process chemists is ready to optimize this route for your specific needs, ensuring maximum efficiency and cost-effectiveness.
We invite you to initiate a conversation about how this technology can enhance your product portfolio and drive value for your organization. Contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate the viability of this synthesis for your projects. Let us help you navigate the complexities of chemical manufacturing and secure a competitive advantage in the market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
