Scalable Green Synthesis of 1,2,3-Triazoles Using Recyclable Polymer-Copper Catalytic Systems
The pharmaceutical and fine chemical industries are constantly seeking robust, environmentally benign methodologies for constructing nitrogen-containing heterocycles, particularly 1,2,3-triazoles, which serve as critical scaffolds in drug discovery and material science. Patent CN103772302A introduces a groundbreaking approach to this challenge by detailing a method for the catalytic synthesis of 1,2,3-triazole compounds utilizing a carboxymethylated pullulan and cuprous oxide catalytic system. This innovation represents a significant departure from traditional protocols that often rely on toxic organic solvents and non-recyclable heavy metal catalysts. By leveraging a biopolymer-based support, the technology enables the reaction to proceed efficiently in water, a universally safe and inexpensive solvent, while maintaining high catalytic activity and selectivity. For R&D directors and process chemists, this patent offers a compelling pathway to streamline synthetic routes for complex intermediates, ensuring that the final products meet stringent purity specifications required for active pharmaceutical ingredients (APIs) without the burden of extensive heavy metal removal processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,2,3-triazoles via the Huisgen 1,3-dipolar cycloaddition has faced substantial hurdles regarding operational safety and environmental impact. Conventional methods typically employ monovalent copper salts or oxides that require additional ligands or bases to enhance catalytic activity, often necessitating the use of volatile organic compounds (VOCs) as solvents. Furthermore, when bulk metal copper or simple cuprous oxide powders are used directly, the reactions frequently suffer from incomplete conversion, low yields, and excessively long reaction times. A critical bottleneck in these traditional workflows is the inability to recover and reuse the catalyst; once the reaction is complete, the copper species often remain dispersed in the mixture, requiring costly and complex purification steps to remove trace metal residues from the final product. This not only inflates the cost of goods sold (COGS) but also generates significant hazardous waste, posing challenges for supply chain heads tasked with maintaining environmental compliance and sustainability goals.
The Novel Approach
The methodology disclosed in the patent overcomes these deficiencies by introducing carboxymethylated pullulan as a functional additive that activates cuprous oxide within an aqueous medium. This novel catalytic system operates effectively under mild conditions, ranging from room temperature to 100°C, eliminating the need for energy-intensive heating or high-pressure equipment. The carboxymethylated pullulan serves a dual purpose: it stabilizes the cuprous oxide nanoparticles to prevent agglomeration and enhances their catalytic efficiency through specific interactions in the water phase. Crucially, because the polymer is water-soluble yet the product is often extractable into organic phases, the catalytic system remains in the aqueous layer, allowing for straightforward separation and repeated reuse. This shift from homogeneous, single-use catalysis to a quasi-heterogeneous, recyclable system fundamentally alters the economic and ecological profile of triazole manufacturing, offering a reliable pharmaceutical intermediate supplier a distinct competitive advantage in green chemistry capabilities.
Mechanistic Insights into Carboxymethylated Pullulan/Cu2O Catalysis
The core of this technological advancement lies in the synergistic interaction between the biopolymer and the metal oxide. In standard aqueous environments, cuprous oxide (Cu2O) often exhibits limited solubility and catalytic activity for click chemistry reactions. However, the introduction of carboxymethyl groups onto the pullulan backbone creates a microenvironment that facilitates the activation of the copper species. The carboxyl groups likely coordinate with the copper centers, increasing their Lewis acidity and accessibility to the substrates, thereby accelerating the cycloaddition kinetics. This mechanism allows the reaction to proceed with high atom economy, converting terminal alkynes and organic azides directly into the desired triazole rings with minimal byproduct formation. The stability provided by the polymer matrix prevents the oxidation of Cu(I) to less active Cu(II) species, ensuring consistent performance throughout the reaction lifecycle.
From an impurity control perspective, this mechanism offers profound benefits for quality assurance teams. Traditional copper-catalyzed reactions often leave behind difficult-to-remove metal complexes that can catalyze degradation pathways in the final API. By utilizing a polymer-supported system that stays in the aqueous phase, the partition coefficient favors the migration of the organic triazole product into the extraction solvent, leaving the bulk of the copper and polymer behind. This inherent separation capability drastically reduces the metal load in the crude product, simplifying downstream purification. Moreover, the mild reaction conditions minimize thermal degradation of sensitive functional groups on the substrates, such as esters or nitro groups, ensuring a cleaner impurity profile. This level of control is essential for producing high-purity pharmaceutical intermediates that comply with international regulatory standards.
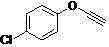

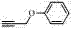
How to Synthesize 1,2,3-Triazole Compounds Efficiently
Implementing this synthesis route requires precise preparation of the catalytic components followed by controlled reaction conditions to maximize yield and recyclability. The process begins with the modification of pullulan to introduce carboxymethyl groups, followed by the mixing of this polymer with cuprous oxide in water. Once the catalytic system is established, the terminal alkyne and organic azide substrates are introduced, and the mixture is stirred under the specified thermal conditions. The detailed standardized synthesis steps, including specific molar ratios, stirring speeds, and workup procedures necessary for reproducibility, are outlined in the guide below.
- Prepare the carboxymethylated pullulan polymer by reacting pullulan with sodium chloroacetate in an alkaline aqueous solution.
- Mix the terminal alkyne compound and organic azide compound in water with cuprous oxide and the prepared carboxymethylated pullulan.
- Stir the reaction mixture at temperatures ranging from room temperature to 100°C until the conversion is complete, then extract the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this carboxymethylated pullulan/cuprous oxide system translates into tangible operational efficiencies and risk mitigation. The primary value driver is the elimination of expensive and toxic organic solvents in favor of water, which significantly reduces raw material costs and waste disposal fees. Additionally, the recyclability of the catalyst system means that the consumption of copper reagents is drastically lowered over multiple production batches, leading to substantial cost savings in manufacturing inputs. The simplicity of the operation, which does not require inert gas protection or specialized high-pressure reactors, further lowers the barrier to entry for scale-up, allowing for faster deployment of production capacity.
- Cost Reduction in Manufacturing: The transition to a water-based solvent system eliminates the need for purchasing, storing, and disposing of large volumes of volatile organic solvents, which are subject to fluctuating market prices and strict environmental regulations. Furthermore, the ability to recycle the catalytic system multiple times without significant loss of activity reduces the per-batch cost of the copper catalyst and the polymer support. This cumulative effect results in a leaner cost structure for the production of triazole derivatives, enhancing profit margins without compromising on quality.
- Enhanced Supply Chain Reliability: Relying on water as the primary solvent mitigates supply chain risks associated with solvent shortages or logistics disruptions, as water is universally available and inexpensive. The robustness of the catalytic system, which tolerates a wide range of substrates including those with electron-withdrawing or donating groups, ensures consistent output even when feedstock quality varies slightly. This reliability is crucial for maintaining continuous production schedules and meeting delivery commitments to downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The green nature of this process aligns perfectly with increasingly stringent global environmental standards, reducing the regulatory burden on manufacturing facilities. The absence of toxic heavy metal residues in the final product simplifies the validation process for regulatory filings. Moreover, the mild reaction conditions allow for easy scale-up from laboratory to pilot and commercial scales using standard stainless steel reactors, facilitating rapid expansion of production capacity to meet market demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for decision-makers evaluating this process for integration into their supply chains.
Q: What is the primary advantage of using carboxymethylated pullulan in this catalytic system?
A: Carboxymethylated pullulan acts as a non-toxic, water-soluble support that activates cuprous oxide, allowing the catalyst to be recycled multiple times without significant loss of activity, unlike traditional homogeneous copper salts.
Q: Can this synthesis method be performed in organic solvents?
A: While possible, the patent specifically highlights water as the preferred solvent to maximize the green chemistry benefits, reduce toxicity, and facilitate the recycling of the polymer-copper catalytic system.
Q: Is the catalyst system reusable for industrial scale-up?
A: Yes, experimental data indicates the catalytic system can be reused for at least 10 cycles with maintained high yields, making it highly suitable for cost-effective commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2,3-Triazole Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of green catalytic technologies like the carboxymethylated pullulan/cuprous oxide system in modernizing chemical manufacturing. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 1,2,3-triazole intermediates meets the exacting standards required by the global pharmaceutical industry.
We invite procurement leaders and technical directors to collaborate with us to optimize their supply chains for triazole-based APIs. By leveraging our expertise in green chemistry and process optimization, we can help you achieve significant efficiency gains. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your next development milestone.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
